 |
 |
 |
| |
Rapidly falling costs for new hepatitis C direct-acting
antivirals (DAAs): potential for universal access
|
| |
| |
Reported by Jules Levin
Durban 2016 July 18-22
Dzintars Gotham1, Melissa Barber2, Joseph Fortunak3, Anton Pozniak4, Andrew Hill4
1 Faculty of Medicine, Imperial College London, London, United Kingdom.
2 Centre of Development Studies, Cambridge University, Cambridge, United Kingdom.
3 Chemistry and Pharmaceutical Sciences, Howard University, Washington, DC, USA.
4 St Stephens AIDS Trust, Chelsea and Westminster Hospital, London, UK

Abstract
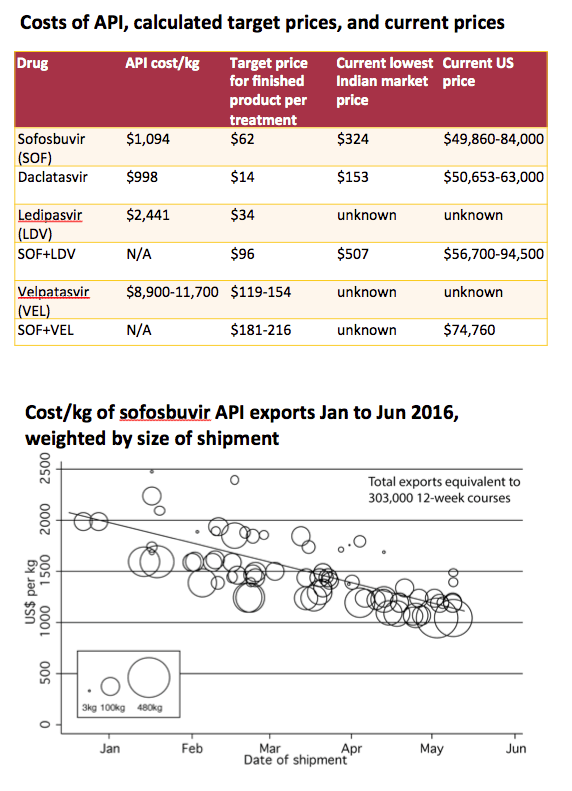
Background: Novel direct-acting antivirals (DAAs) achieve high sustained viral response rates of >90% in chronic hepatitis C (HCV). But access to DAAs is low: ''access'' prices are available in countries covering only 50% of the worldwide epidemic. Production costs depend largely on prices of active pharmaceutical ingredient (API).
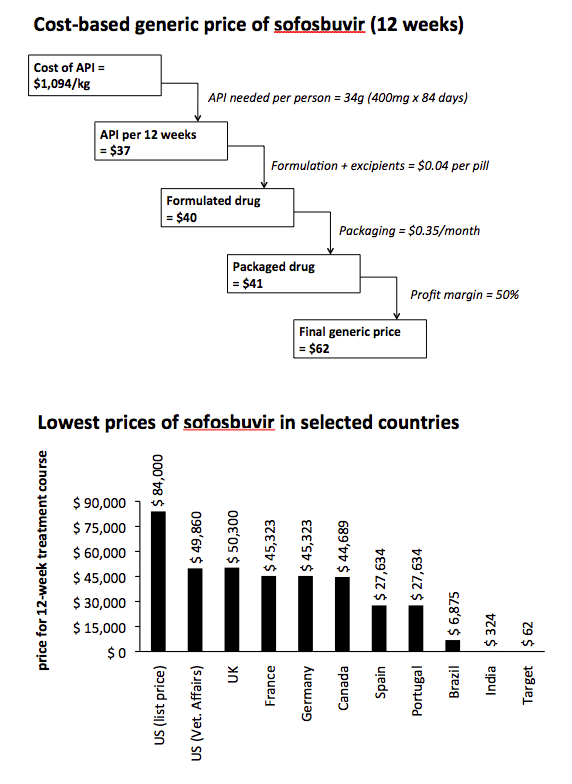
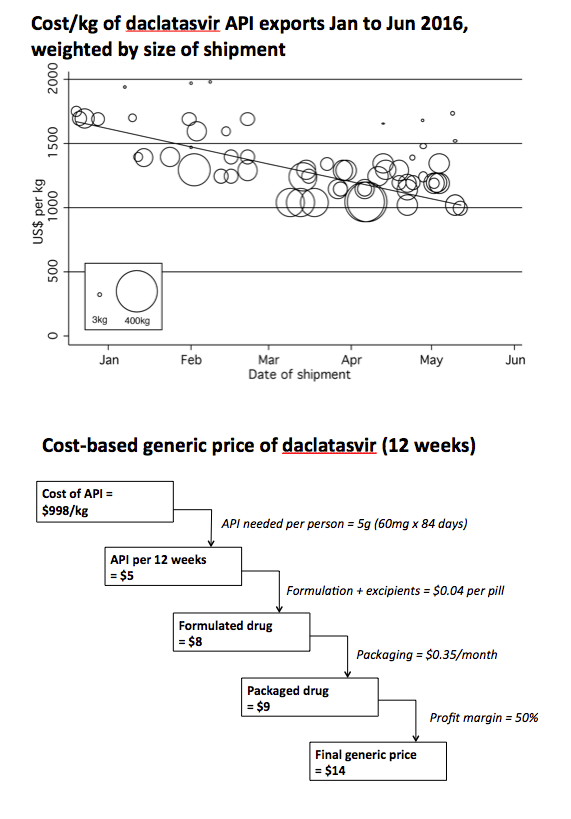
Methods: Data on the per-kilogram prices of exported API, and export volumes, were extracted from an online database (www.infodriveindia.com) for Jan-Dec 2015. Mean end-2015 API costs were calculated using linear regression models weighted by shipment size (figures). For velpatasvir, API cost was calculated by analysing chemical synthesis, costs of raw materials, processes, and yields. Per-pill API costs were calculated based on daily dosage. Estimated costs for formulation and excipients ($0.04/ pill), packaging ($0.35/month), and a profit margin (50%) were added.
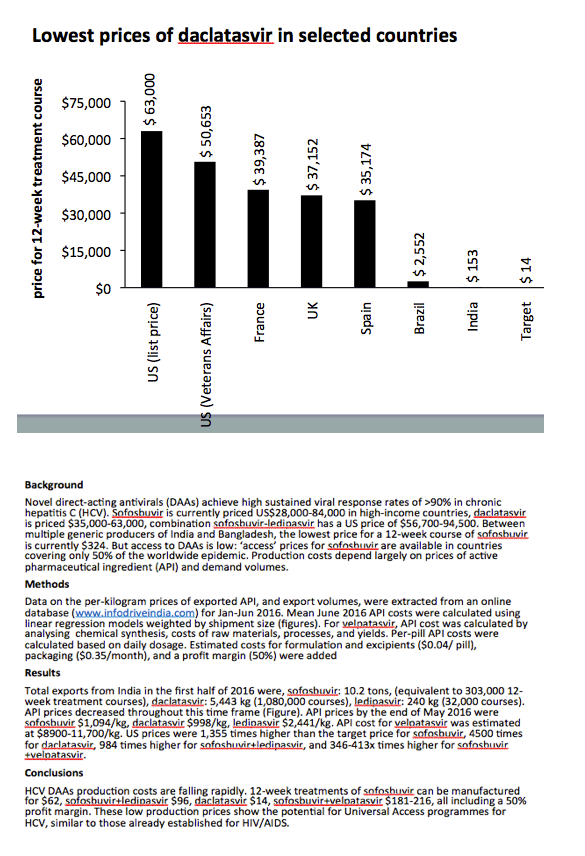
Results: Total exports from India in 2015 were, sofosbuvir: 8.4 tons, (equivalent to 250,000 12-week treatment courses), daclatasvir: 523 kg (104,000 courses), ledipasvir: 56 kg (7300 courses). API prices decreased throughout 2015 (Figure). End-2015 API prices were sofosbuvir $1758/kg, daclatasvir $1432/kg, ledipasvir $11,432/kg. API cost for velpatasvir was estimated at $8900-11,700/kg. US price was 884 times higher than the target price for sofosbuvir, 3706 times for daclatasvir and 409 times higher for sofosbuvir+ledipasvir.
Conclusions: HCV DAAs production costs are falling rapidly. We estimate that 12-week treatments of sofosbuvir can be manufactured for $95, sofosbuvir+ledipasvir $231, daclatasvir $17, velpatasvir $119-154, all including a 50% profit margin. These low production prices show the potential for Universal Access programmes for HCV, similar to those already established for HIV/AIDS.

|
| |
|
 |
 |
|
|