 |
 |
 |
| |
Drug-Drug Interactions Studies between HCV Antivirals Sofosbuvir/Velpatasvir and HIV Antiretrovirals in Healthy Volunteers-2
|
| |
| |
Anti-HCV Duo Sofosbuvir/Velpatasvir Not Recommended With Efavirenz..... no other clinically meaningful interactions between antiretrovirals and velpatasvir or sofosbuvir.
21st International AIDS Conference (AIDS 2016), July 18-22, 2016, Durban, South Africa
Mark Mascolini
Pharmacokinetic (PK) studies in healthy volunteers indicate that coformulated sofosbuvir/velpatasvir is not recommended with efavirenz because velpatasvir levels drop by 50% when sofosbuvir/velpatasvir is taken with efavirenz plus tenofovir/emtricitabine (TDF/FTC) [1]. The PK studies disclosed no other clinically meaningful interactions between antiretrovirals and velpatasvir or sofosbuvir.
Sofosbuvir, an HCV nucleotide polymerase inhibitor, and velpatasvir, an HCV NS5A inhibitor, are coformulated at doses of 400/100 mg as a once-daily anti-HCV regimen. Phase 3 trials showed high sustained virologic response (SVR) rates with 12 weeks of sofosbuvir/velpatasvir, and the two-in-one formulation is being tested in phase 3 trials in HCV/HIV-coinfected patients [2-4]. In the ASTRAL-5 trial, 12 weeks of sofosbuvir/velpatasvir in coinfected people with HCV genotypes 1, 2, 3, and 4 yielded a 95% SVR12 rate [2].
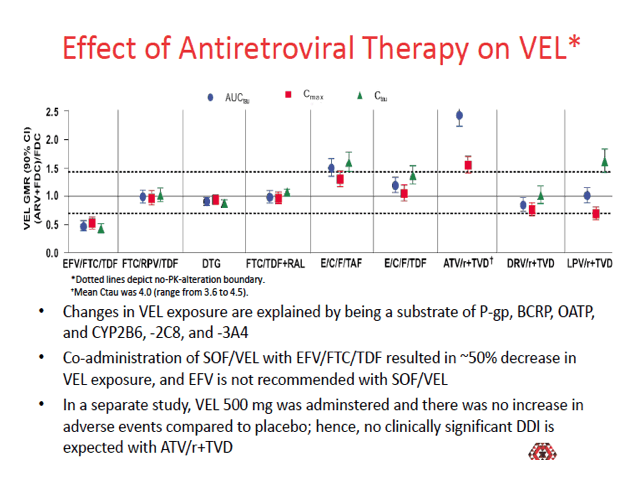
Sofosbuvir and velpatasvir have a medium to high potential for interactions with antiretrovirals. Sofosbuvir is a substrate of the important drug transporters P-glycoprotein (P-gp) and breast cancer resistance protein (BCRP), though it is not a substrate for the critical CYP or UGT-1A1 antiretroviral-metabolizing enzymes. The sofosbuvir metabolite GS-331007 is not a substrate of P-gp, BCRP, or other transporters. Velpatasvir is a substrate of P-gp, BCRP, organic anion transport polypeptide (OATP)-1B, and the important metabolizing enzymes CYP3A4, CYP2B6, and CYP2C8. Velpatasvir inhibits P-gp, BCRP, and OATP1B.
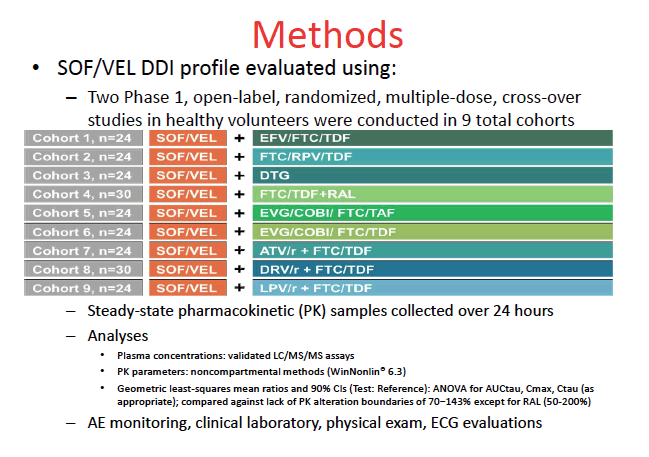
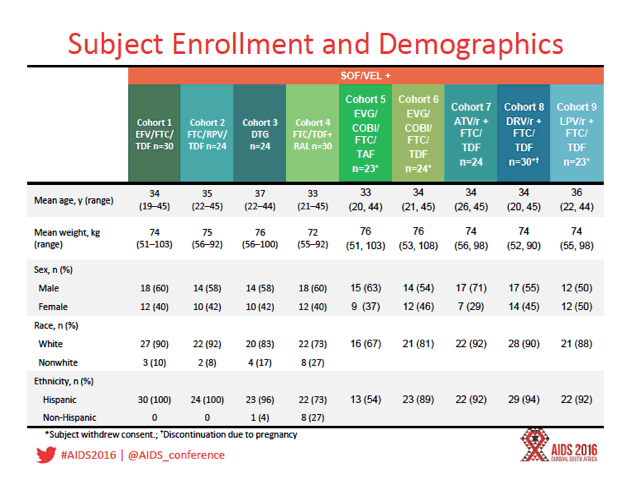
The analysis focused on two phase 1 multiple-dose crossover studies that involved 9 cohorts of healthy volunteers, all of them taking 400/100 mg of sofosbuvir/velpatasvir with one or more antiretrovirals including FTC, TDF, tenofovir alafenamide (TAF), efavirenz, rilpivirine, dolutegravir, raltegravir, elvitegravir/cobicistat, and ritonavir-boosted atazanavir, darunavir, or lopinavir. Participants averaged about 35 years in age, about 40% were women, and two thirds or more in each cohort were white.
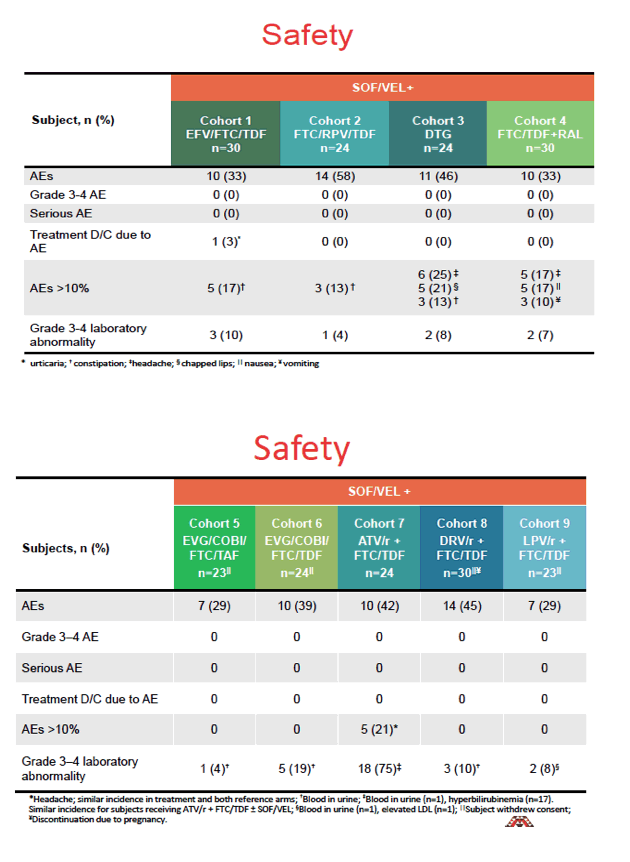
There were no grade 3 or 4 adverse events or serious adverse events in any of the 9 cohorts. Only one participant withdrew because of adverse events. Eighteen of 24 volunteers (75%) in the cohort taking atazanavir/ritonavir had grade 3 or 4 lab abnormalities, 17 of them hyperbilirubinemia. Grade 3 or 4 lab abnormality rates were 19% or lower in the other 8 cohorts.
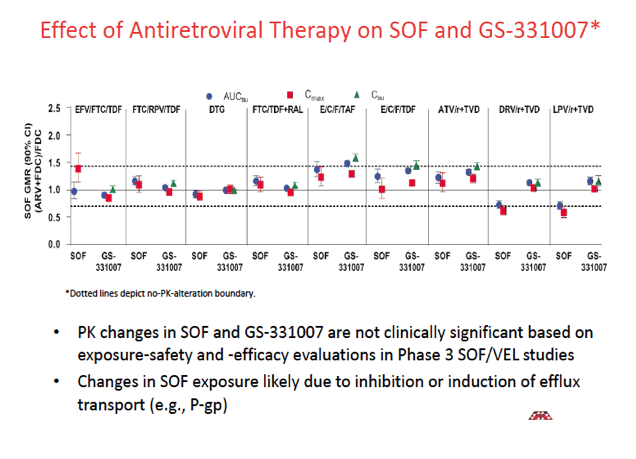
When volunteers took sofosbuvir/velpatasvir with efavirenz/TDF/FTC, velpatasvir exposure fell about 50%. As a result, efavirenz is not recommended with sofosbuvir/velpatasvir. None of the other analyses disclosed a possibly clinically meaningful impact of antiretrovirals on sofosbuvir or velpatasvir.
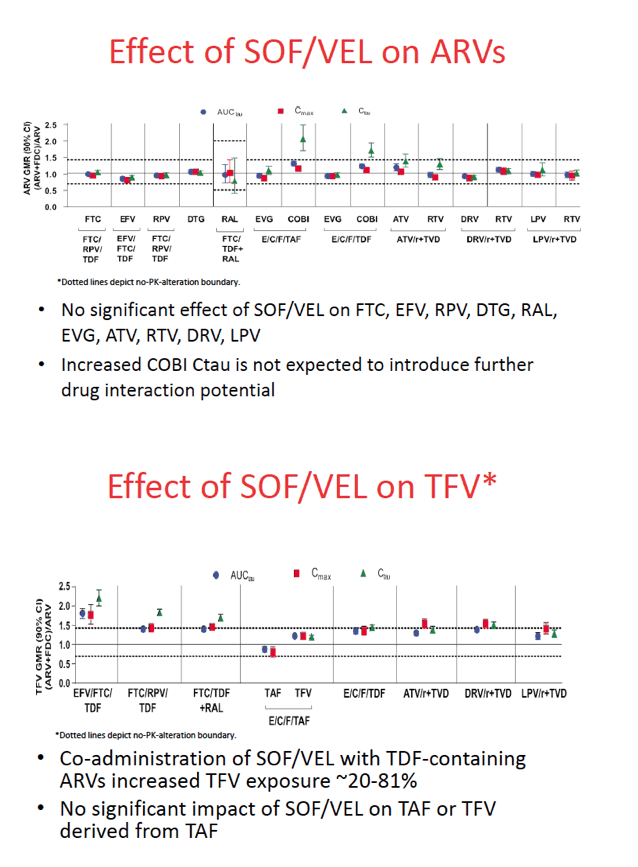
Taking sofosbuvir/velpatasvir with TDF-containing regimens resulted in 20% to 81% higher tenofovir exposure, but sofosbuvir/velpatasvir did not affect concentrations of TAF or tenofovir derived from TAF. The PK studies indicated that sofosbuvir/velpatasvir had no other notable impact on the antiretrovirals studied. Trough concentrations of the booster cobicistat were up to 2-fold higher with than without sofosbuvir/velpatasvir, but a higher cobicistat trough is not expected to affect exposure of coadministered antiretrovirals.
The researchers concluded that these and previous findings support coadministration of sofosbuvir/velpatasvir with the reverse transcriptase inhibitors TDF, TAF, FTC, and rilpivirine; the integrase inhibitors dolutegravir, elvitegravir, and raltegravir; the protease inhibitors atazanavir, darunavir, and lopinavir; and the PK boosters ritonavir and cobicistat.
References
1. Mogalian E, Luetkemeyer A, Naik S, et al. Drug-drug interactions studies between HCV antivirals sofosbuvir and velpatasvir and HIV antiretrovirals. 21st International AIDS Conference (AIDS 2016). July 18-22, 2016. Durban, South Africa. Abstract WEAB0302.
2. Sofosbuvir/velpatasvir for 12 weeks in patients coinfected with HCV and HIV-1: the ASTRAL-5 study. EASL. Barcelona. April 13-17, 2016. http://www.natap.org/2016/EASL/EASL_32.htm
3. ClinicalTrials.gov. Efficacy and safety of sofosbuvir/velpatasvir fixed dose combination for 12 weeks in adults with chronic hepatitis C virus (HCV) and human immunodeficiency virus (HIV)-1 coinfection. ClinicalTrials.gov identifier NCT02480712. https://clinicaltrials.gov/ct2/show/NCT02480712
4. ClinicalTrials.gov. Randomised study of interferon-free treatment for recently acquired hepatitis C in PWID and people with HIV coinfection (REACT). ClinicalTrials.gov identifier NCT02625909. https://clinicaltrials.gov/ct2/show/NCT02625909


|
| |
|
 |
 |
|
|