 |
 |
 |
| |
An HIV pre-exposure prophylaxis (PrEP) demonstration project and
safety study for adolescent MSM ages 15-17 in the United States (ATN 113)
|
| |
| |
Reported by Jules Levin
Durban 2016 July 18-22
Presenter
Sybil Hosek
Authors
S. Hosek1, R. Landovitz2, B. Rudy3, B. Kapogiannis4, G. Siberry4, B. Rutledge5, N. Liu5, J. Brothers1, J. Rooney6, C.M. Wilson7, The Adolescent Trials Network for HIV/AIDS Interventions (ATN)
Institutions
1Stroger Hospital of Cook County, Psychiatry, Chicago, United States, 2University of California Los Angeles, Los Angeles, United States, 3New York University Medical Center, New York, United States, 4NICHD/MPIDB, Bethesda, United States, 5Westat, Rockville, United States, 6Gilead Sciences, Foster City, United States, 7University of Alabama at Birmingham, Birmingham, United States

IAC: Changes in Bone Mass after Discontinuation of PrEP with Tenofovir Disoproxil Fumarate/Emtricitabine (TDF/FTC) in Young Men Who Have Sex with Men (YMSM): Extension Phase Results of Adolescent Trials Network (ATN) Study 110 - (07/20/14)


Abstract
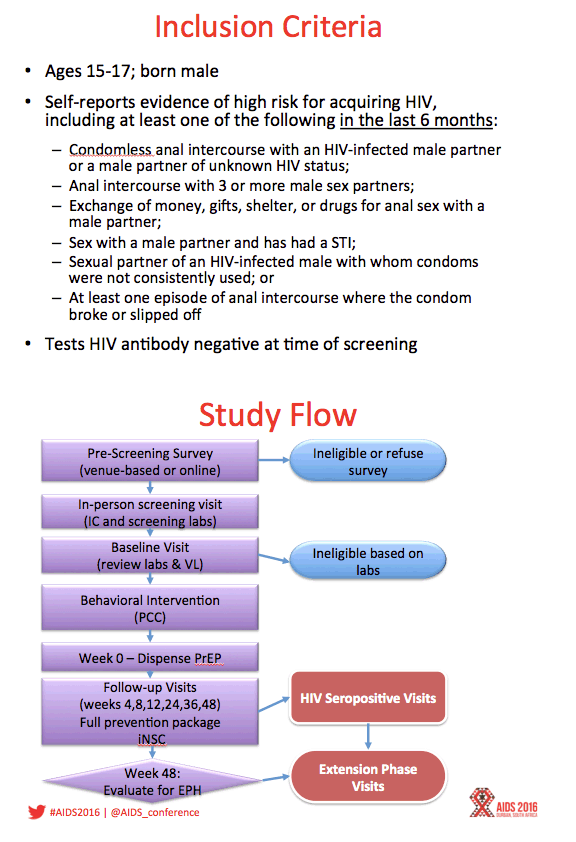
Background: Adolescents represent a key population for implementation of PrEP interventions worldwide, yet TDF/FTC PrEP is not currently licensed under age 18. This open-label PrEP study examined safety of and adherence to PrEP along with changes in sexual risk behavior among adolescent MSM in 6 US cities.
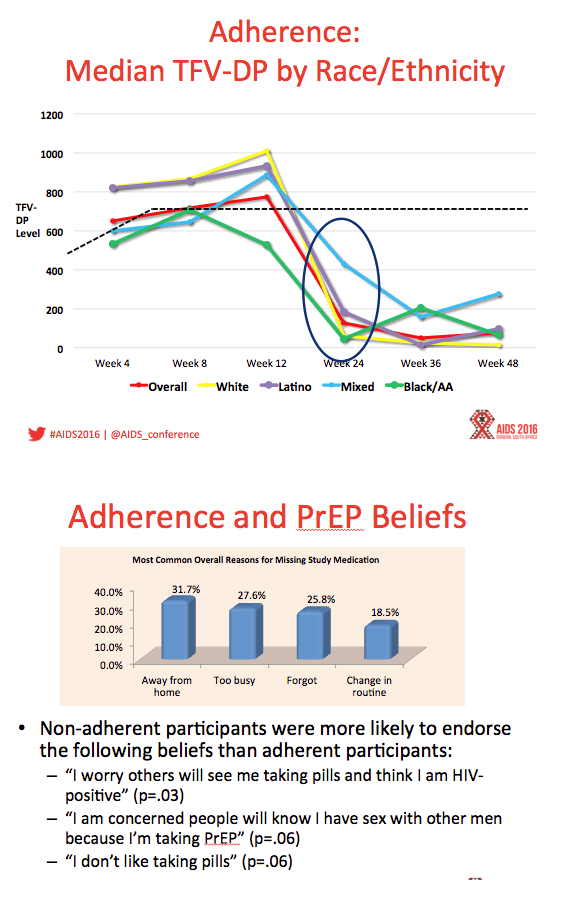
Methods: ATN 113 (Project PrEPare) combined PrEP with behavioral risk reduction and adherence support. HIV-uninfected MSM 15-17 years old who reported HIV risk behavior in the past 6 months were eligible. Youth were allowed to autonomously consent to study participation. Study visits occurred at baseline, monthly through week 12, then quarterly through week 48. Serial tenofovir diphosphate (TFV-DP) levels in dried blood spots (DBS) were used to measure adherence.
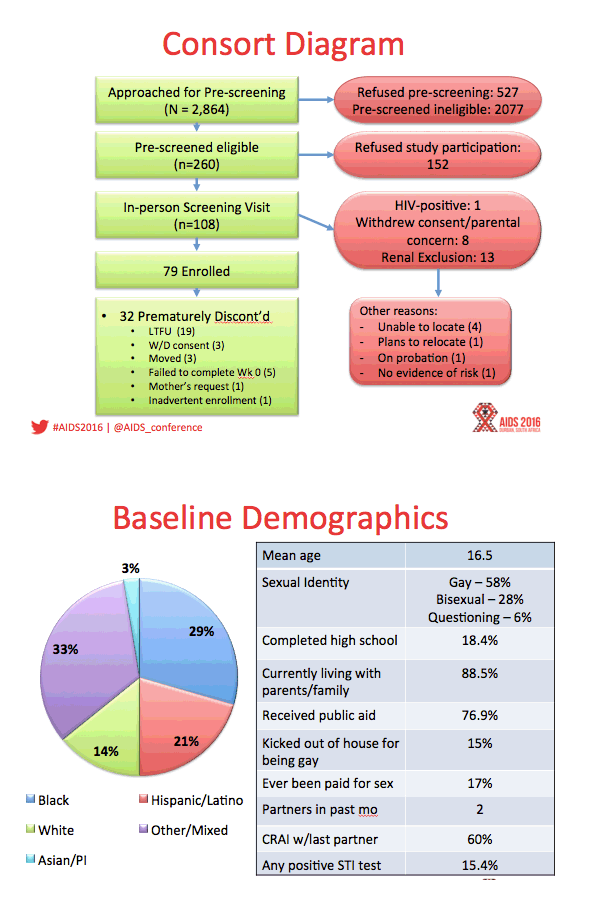
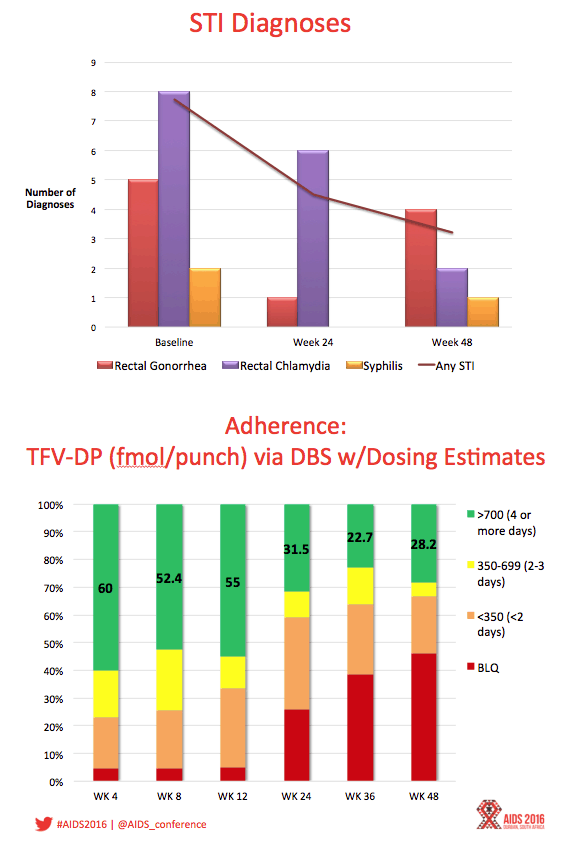
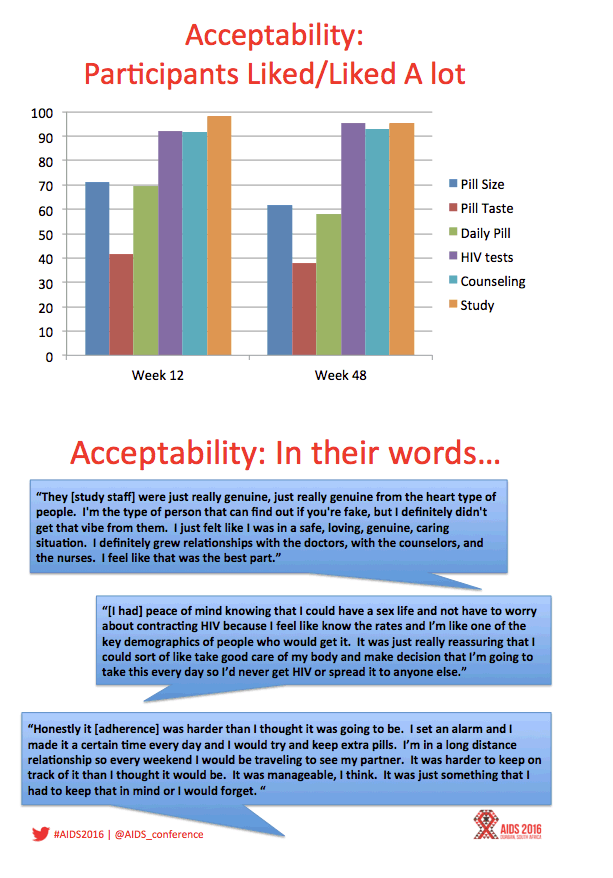
Results: Between August 2013 - September 2014, 2864 individuals were approached, 260 (9%) were preliminarily eligible, and 78 were enrolled (mean age=16.5; 33.3% Mixed race, 29.5% Black, 20.5% Latino). Baseline STIs were diagnosed and treated in 15.4% of participants. Incident STIs were diagnosed in 12.3% of participants at week 24 and 10.6% at week 48. The HIV seroconversion rate per 100 person-years was 6.41 (95% CI: 4.90-25.87). Condomless sex was reported by the majority of participants throughout the study; no significant associations were found between condomless sex and adherence. Figure 1 shows TFV-DP levels. Non-adherent participants were significantly more likely than adherent participants to report worry that others would think they had HIV if they saw their PrEP pills (p=.03).
Conclusions: ATN 113 enrolled a diverse sample of YMSM at risk for HIV who self-consented to study participation. The majority of participants achieved protective drug levels during monthly visits, yet adherence decreased with quarterly visits. HIV incidence was still high despite PrEP provision, suggesting high background incidence. Regulatory approvals for youth under 18 years are required to foster support for youth-friendly settings that will optimize PrEP use.

|
| |
|
 |
 |
|
|