 |
 |
 |
| |
Clinical, biological and Pharmacological relevance of Ledipasvir/Sofosbuvir drug interaction with TDF containing Regimen in HCV/HIV co-infected patients.
|
| |
| |
Reported by Jules Levin
17th International Workshop on Clinical Pharmacology of HIV & Hepatitis Therapy
June 8-10, 2016, Washington, DC, USA
Blondel L1, L. MP1, 8, Gervais A2, Larrouy L3, Salmon D4, Todesco E5, Valantin MA6, Lopez-Zaragoza JL7, Dominguez S,7Yazdanpanah Y2, 8, Peytavin G1, 8
1APHP, Bichat-Claude Bernard Hospital, Pharmaco-Toxicology Department , 2APHP, Bichat-Claude Bernard Hospital, Infectious Diseases & Tropical Department, 3APHP, Bichat-Claude Bernard Hospital, Virology Department, 4APHP, Cochin Hospital, Infectious Diseases & Tropical Department, 5 APHP , Pitie salpetriere Hospital, Virology Department6APHP , Pitie salpetriere Hospital, Infectious Diseases & Tropical Department, 7Henri Mondor hospital, clinical immunology department, 8IAME, UMR 1137,
INSERM, Paris, France


from Program Abstract
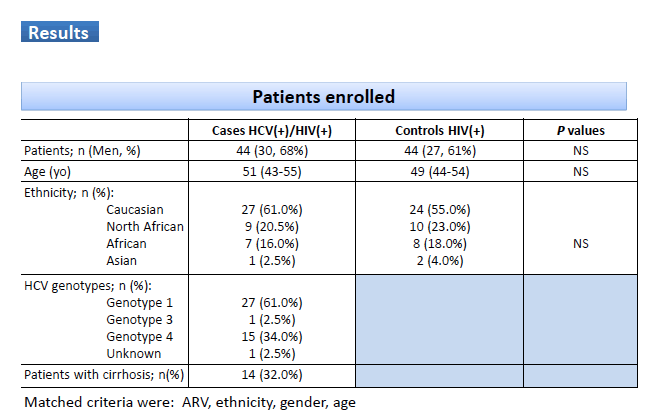
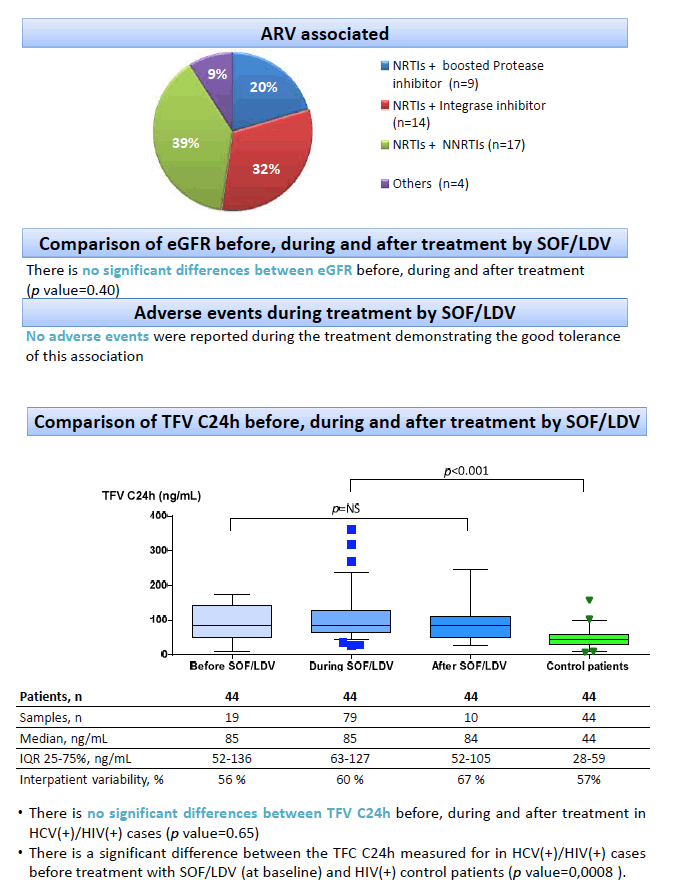
Results: Overall, 44 patients were recruited (68% men, age 51yo (43-55)). HCV genotypes: 1 (61%); 3 (2.5%); 4 (34%); unknown (2.5%). DAA duration was: 8 weeks (3 patients); 12 weeks (37 patients); 24 weeks (4 patients). Thirty four patients without cirrhosis (77%), 8 with cirrhosis (18%), 2 unknown (5%). Antiretroviral associated with TDF were: emtricitabine (42 patients) and abacavir (1 patient); etravirine (3 patients), rilpivirine (10 patients) and efavirenz (8 patients); darunavir/r (7 patients), atazanavir/r (3 patients); raltegravir (11 patients), dolutegravir (1 patient) and elvitegravir/cobi (4 patients). Median time between the 2 set points during and after LDV was 23 weeks (22-26). TFV C24h before LDV was 85ng/mL (52-136; n=15 samples; n=15 patients, CV 56%); during LDV was 85ng/mL (63-127; n=75 samples; n=44 patients, CV 60%); after LDV was 86 ng/mL (76-90; n=10 samples; n=9 patients, CV 67%) (p value=0.65). LDV C24h was 171ng/mL (95-290; n=75 samples; n=44 patients, CV 107%). SOF007 C24h was 390 ng/mL (255-622, n=75 samples; n=44 patients, CV 82%). eGFR before LDV was 85 mL/min (76-90; n=15 samples; n=15 patients, CV 15%); during LDV was 90 mL/min (78-90; n=75 samples; n=44 patients, CV 18%) and after LDV was 90 mL/min (84-90; n=9 samples; 9 patients, CV 14%) (p value=0.40). No adverse events were reported during treatment.
Conclusion: In a population of HCV/HIV co-infected patients chronically receiving TDF containing regimen, TDF C24h before DAA initiation were as expected in hepatic impairment, slightly higher compared to mono-HIV-infected patients with normal liver function (22-66 ng/mL). In these conditions, the drug-drug interaction between SOF/LDV and TFV appeared limited. Consequently, no impact on kidney function or safety considerations was observed during the short 8 to 24 weeks DAA treatment duration. Nevertheless, therapeutic drug monitoring should still be recommended in patients with pre-existent renal dysfunction.

|
| |
|
 |
 |
|
|