 |
 |
 |
| |
Open-Label, Drug-Drug Interaction Study between HIV-1 Attachment Inhibitor BMS-663068 and Maraviroc in Healthy Subjects
|
| |
| |
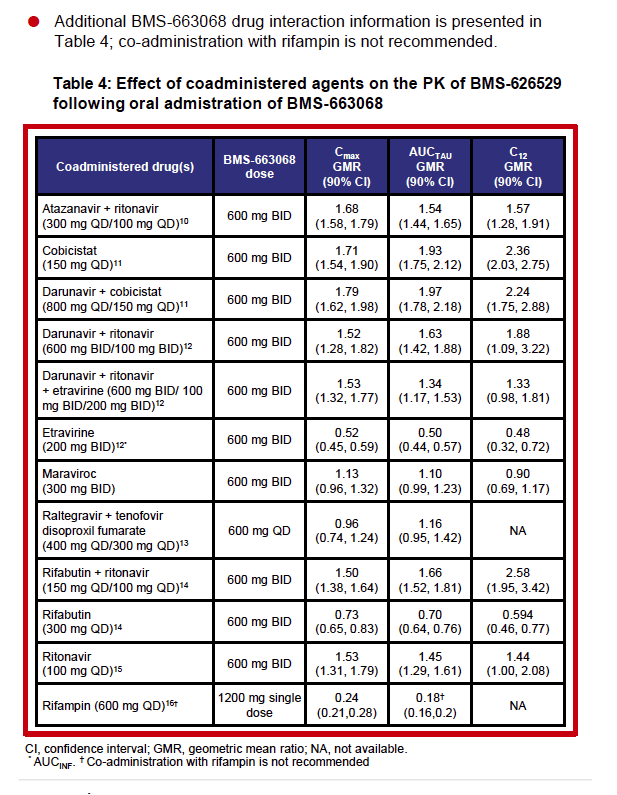
"A Phase III trial (NCT02362503) evaluating BMS-663068 for use in heavily treatment-experienced adults who have two or fewer active antiretroviral (ARV) classes remaining due to resistance, intolerabilities and/or contraindications, is ongoing. .....These data suggest that coadministration of BMS-663068 and maraviroc requires no dose modification for either agent."
Reported by Jules Levin
17th International Workshop on Clinical Pharmacology of HIV & Hepatitis Therapy; June 8-10, 2016; Washington, DC
M Chang,1 E Myers,1 P Ravindran,2 J Wang,2 L Reynolds,3 V Fishman,4 E DeNoia,5 S Lubin,1 T Eley2
1Bristol-Myers Squibb, Research and Development, Pennington, NJ, USA; 2Bristol-Myers Squibb, Research and Development, Princeton, NJ, USA; 3ICON Development Solutions, Gaithersburg, MD, USA; 4ICON Clinical Solutions, Warrington, PA, USA; 5ICON Development Solutions, San Antonio, TX, USA


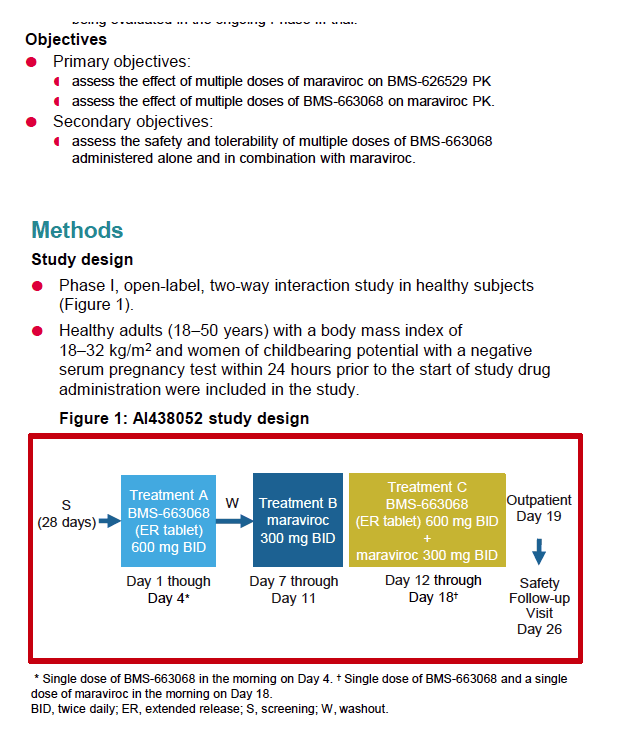


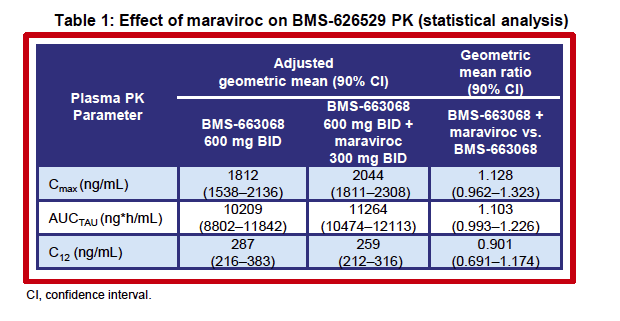
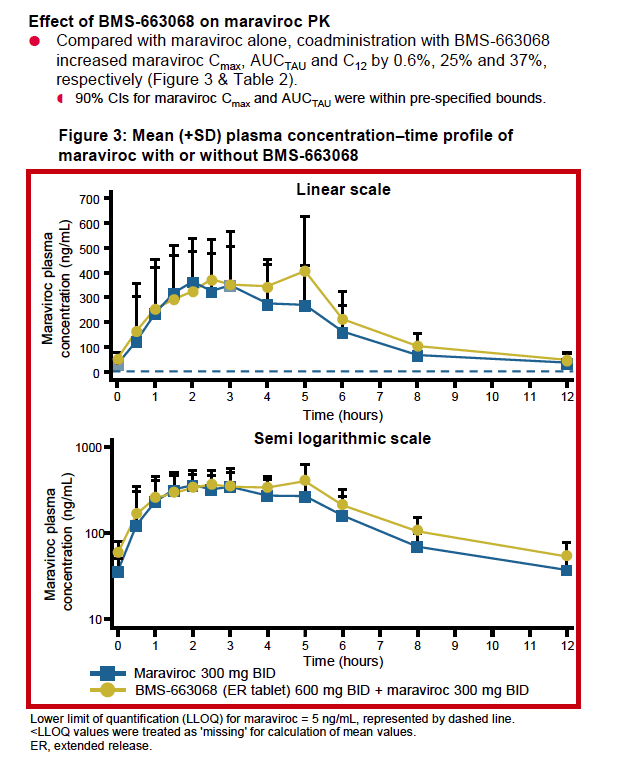

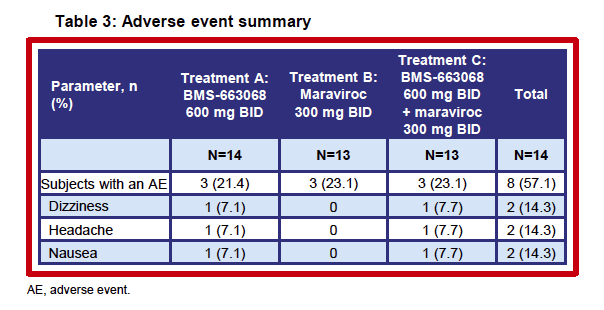

|
| |
|
 |
 |
|
|