 |
 |
 |
| |
Development of Dolutegravir Combination Nanoparticle Fabrications for HIV Prophylaxis
|
| |
| |
Reported by Jules Levin
17th International Workshop on Clinical Pharmacology of HIV and Hepatitis Therapy, 8-10 June 2016, Washington, DC, USA
Annemarie Shibata, Ph.D.
in collaboration with Dr. Christopher Destache
Creighton University
Omaha, NE

program abstract
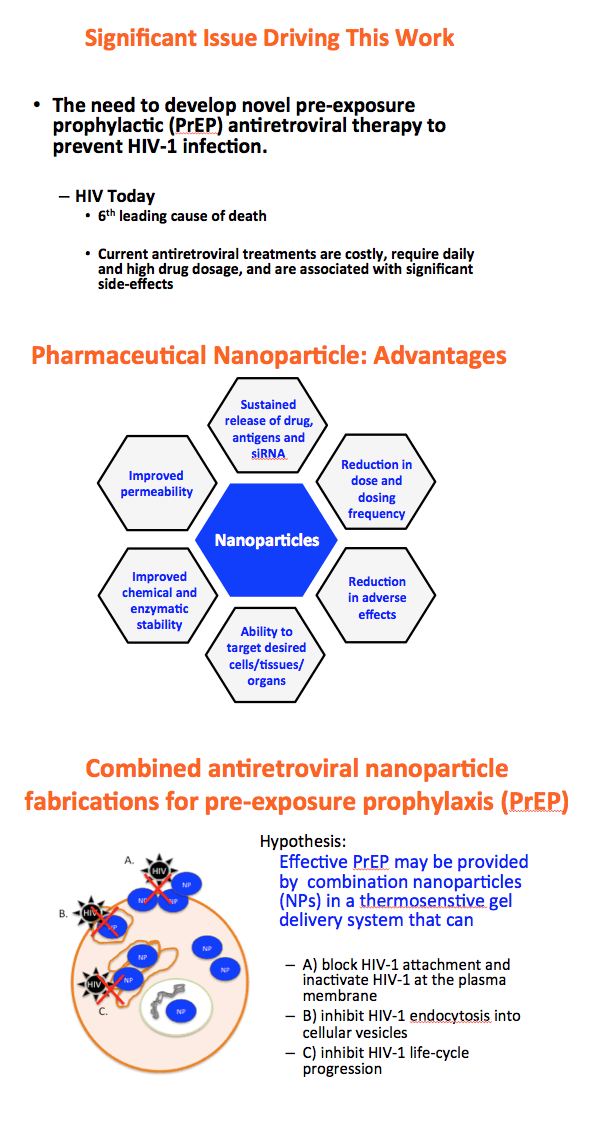
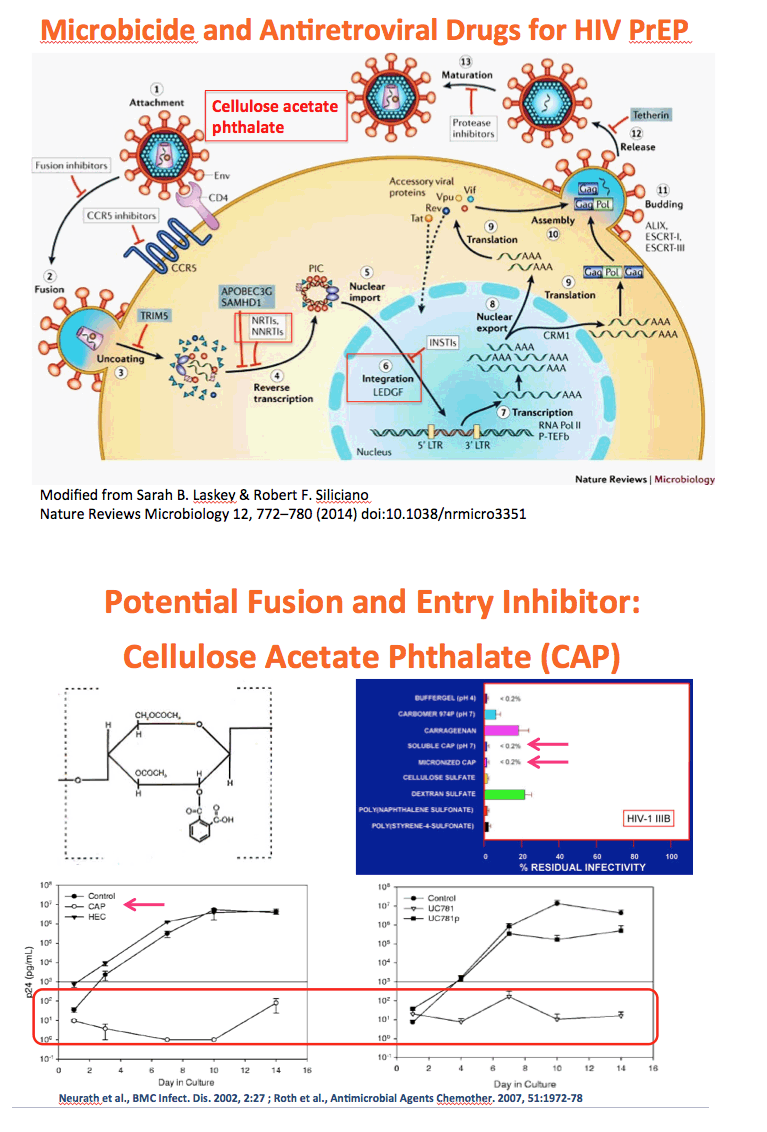
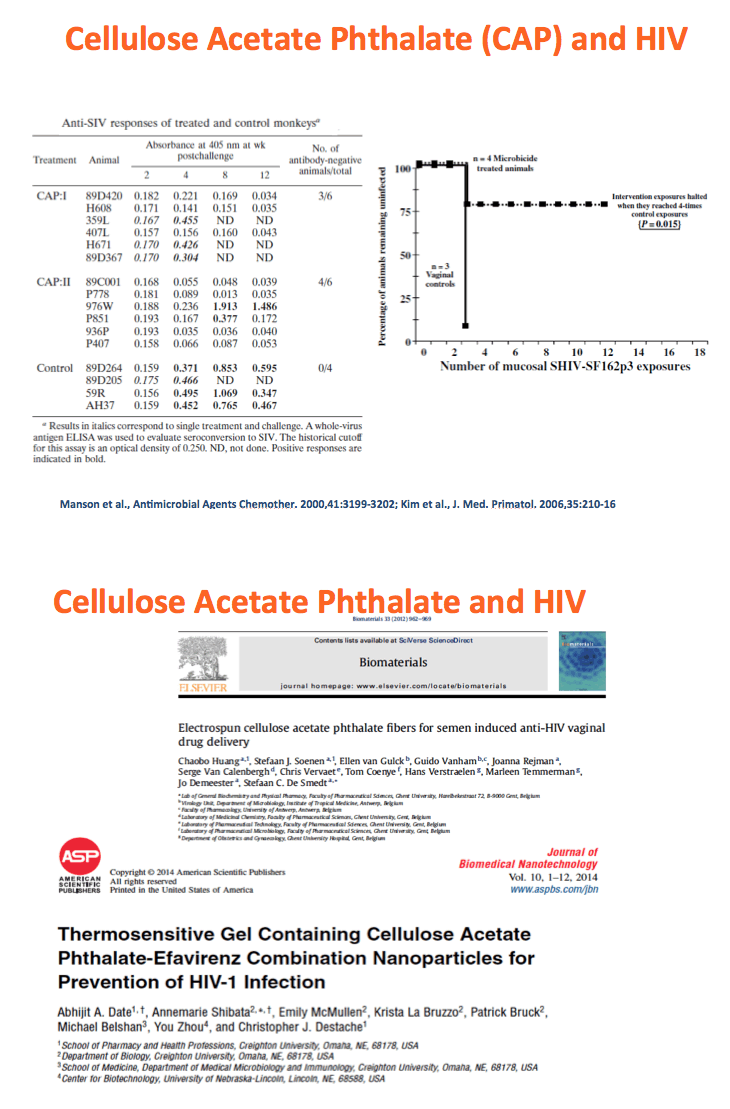
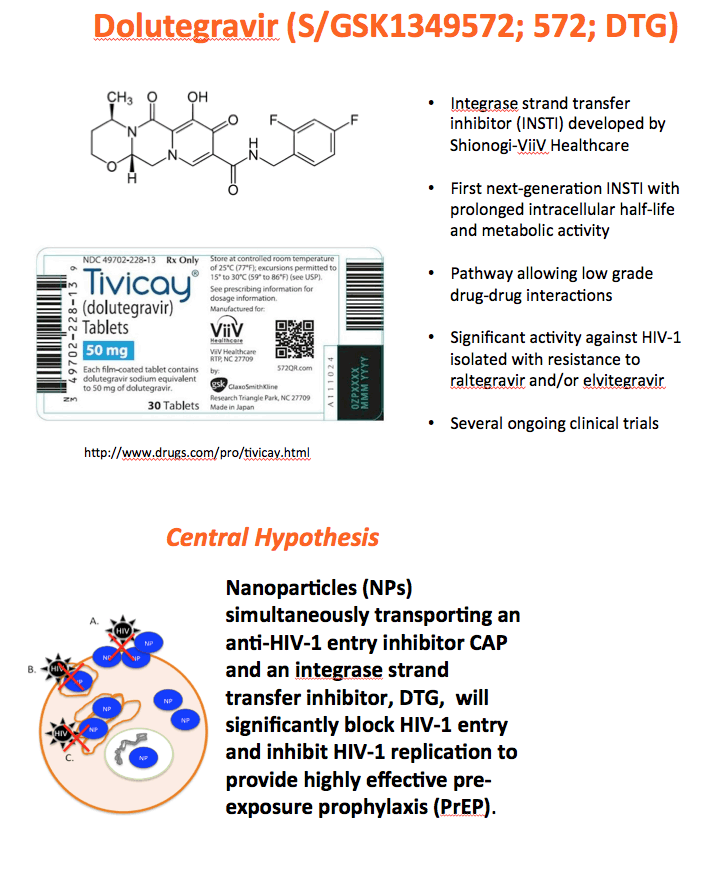
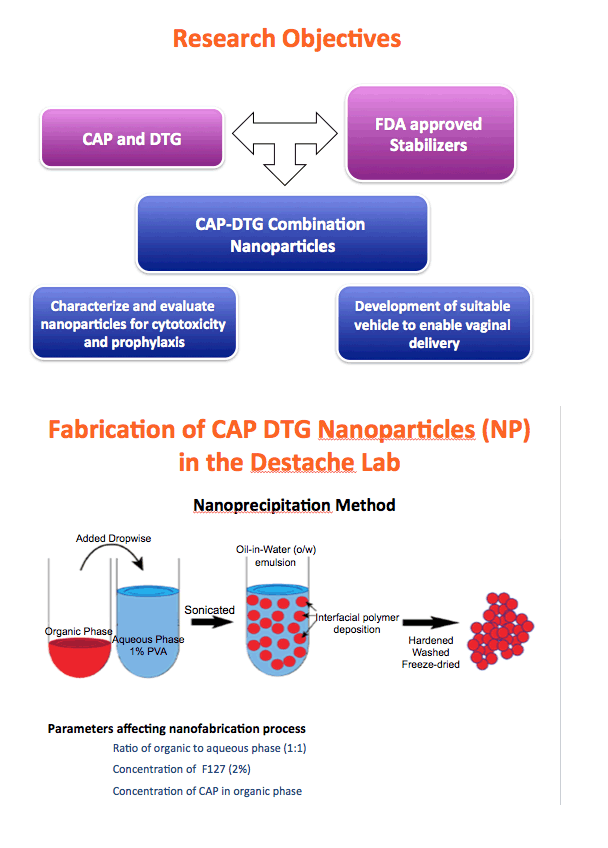
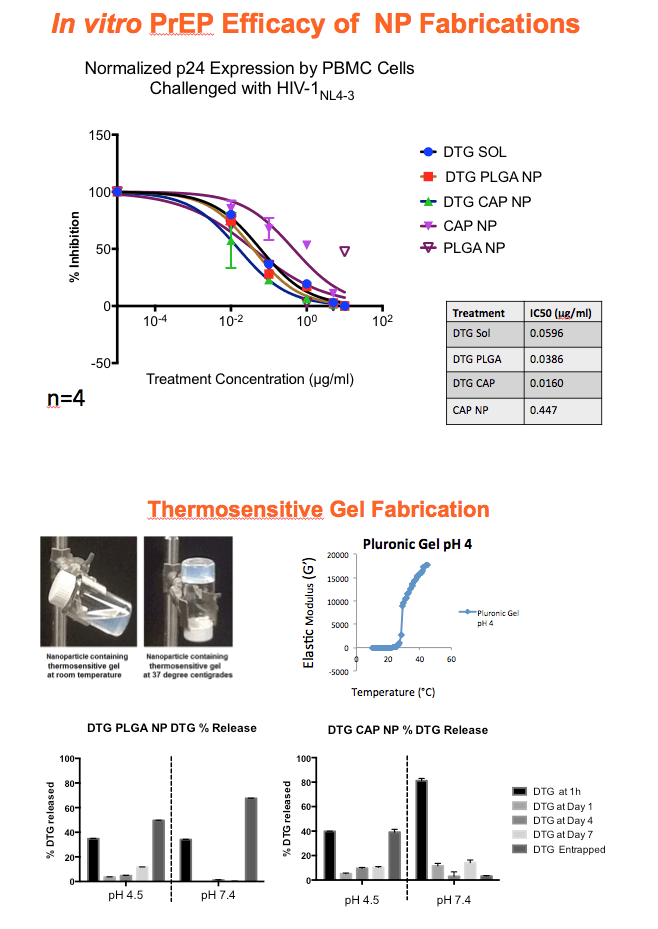
Background: The numbers of individuals infected with HIV could be reduced with highly effective pre-exposure prophylaxis (PreP). Dolutegravir (DTG) is an integrase strand transfer inhibitor with potent anti-HIV activity. Cellulose acetate phthalate (CAP) is a natural polymer with HIV-1 entry inhibitory properties. Development of DTG-loaded CAP nanoparticles (DTG-CAP-NP) could serve as a novel combination for PreP.
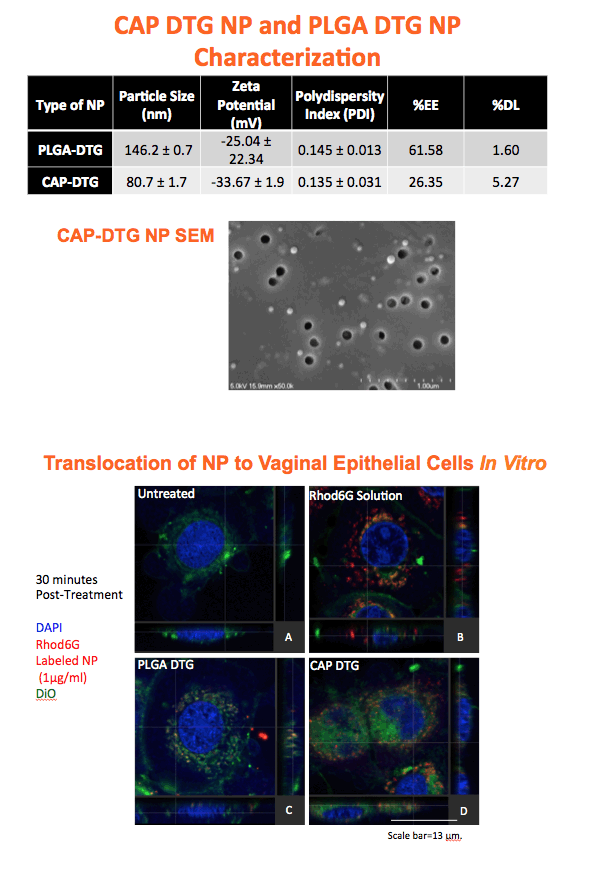
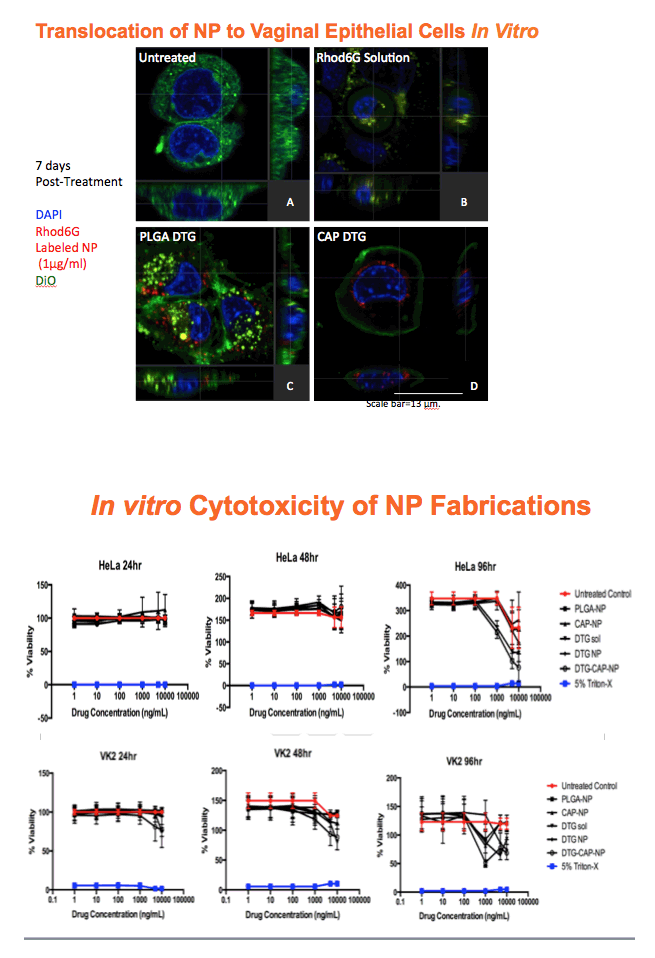
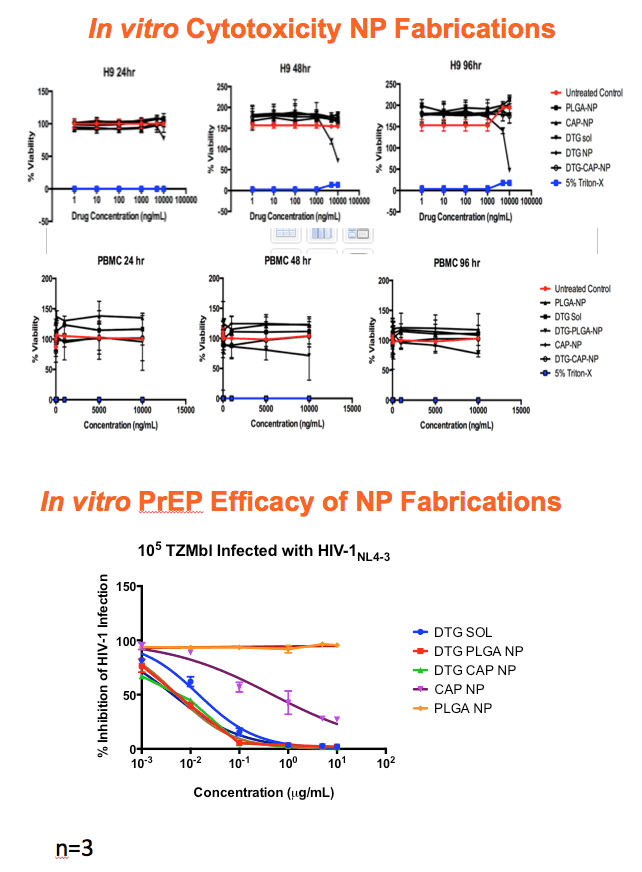
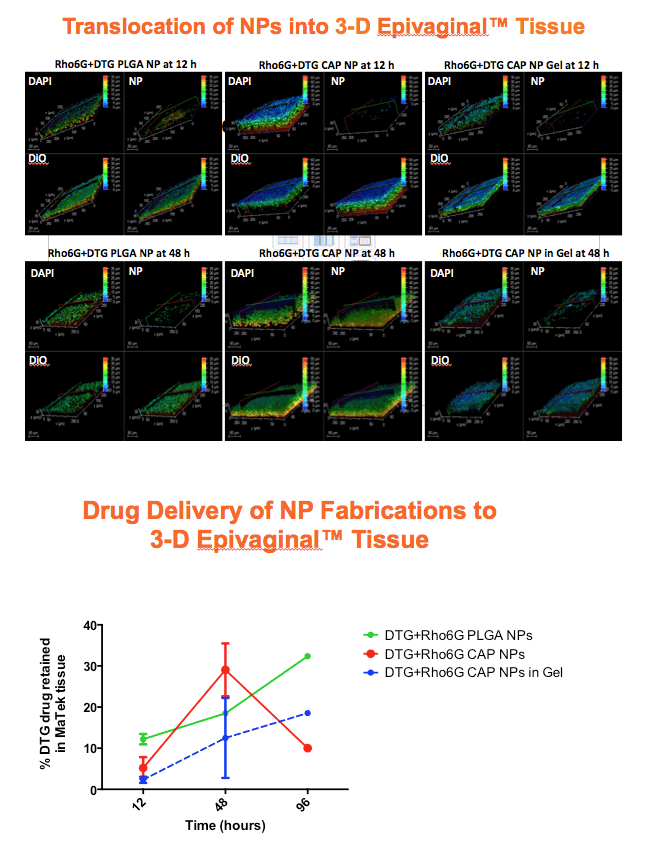
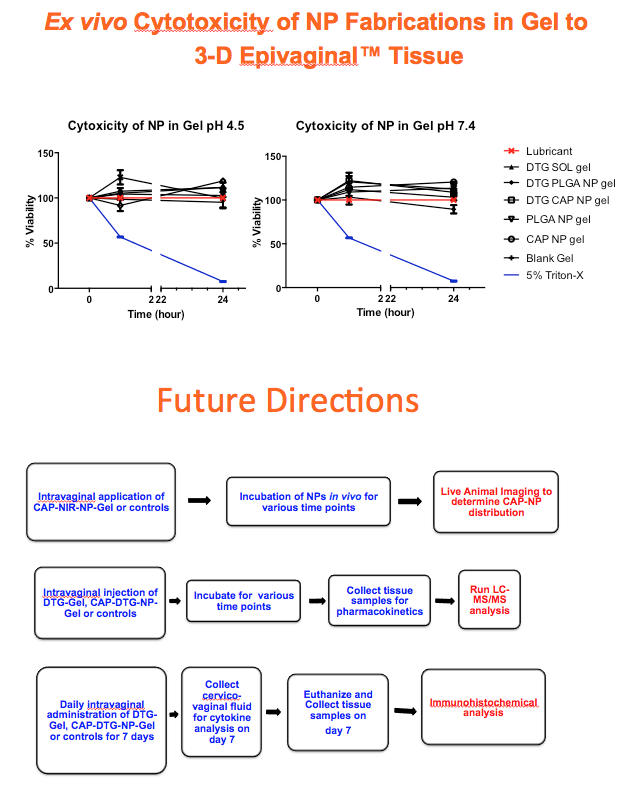
Methods: Anoil-in-water homogenization was used to fabricate DTG-CAP-NP and CAP-NP. Pluronic F127 (2%) and organic to aqueous phase ratio of 1:1 were used for DTG-CAP-NP and CAP-NP. Encapsulation efficiency (EE) of DTG in DTG-CAP-NP was validated by HPLC. Cytotoxicity of DTG-CAP-NP was evaluated and compared to CAP-NP and DTG solution in cell lines and human Epivaginal™ tissues. Intracellular delivery of DTG to cell lines was determined by pretreatment with DTG-CAP-NP or DTG solution and cell lysates were analyzed by HPLC. To evaluate antiviral activity, TZM-bl cells were treated with DTG-CAP-NP and DTG solution for 1 or 3 days then infected with HIV-1NL-4-3. Luminescence was measured after 48 h.
Results: Mean (+ SD) size < 80.7 ± 1.7 nm and surface charge -33.67 ± 1.9 mV for DTG-CAP-NP. EE of DTG was > 26.3% in CAP-DTG-NP. DTG-CAP-NP were significantly less toxic to cell lines as compared to DTG solution and non-toxic to endocervical tissues. CAP-NP were not toxic to cell lines or Epivaginal™ tissues at concentrations as high as 100 μg/mL. DTG-CAP-NP showed complete inhibition of HIV-1 infection at a concentration equivalent to 5 ng of DTG whereas DTG solution and CAP-NP showed significantly less inhibition at this concentration (p<0.05). These data indicate synergy between DTG and CAP as well as the advantage of nanoscale fabrication.
Conclusions: DTG-CAP-NP would be a novel vaginal microbicide with greater potency than DTG in solution and may offer a sustained release PrEP option for HIV. This is the first report of DTG fabricated into a NP formulation for PReP for treatment.

|
| |
|
 |
 |
|
|