 |
 |
 |
| |
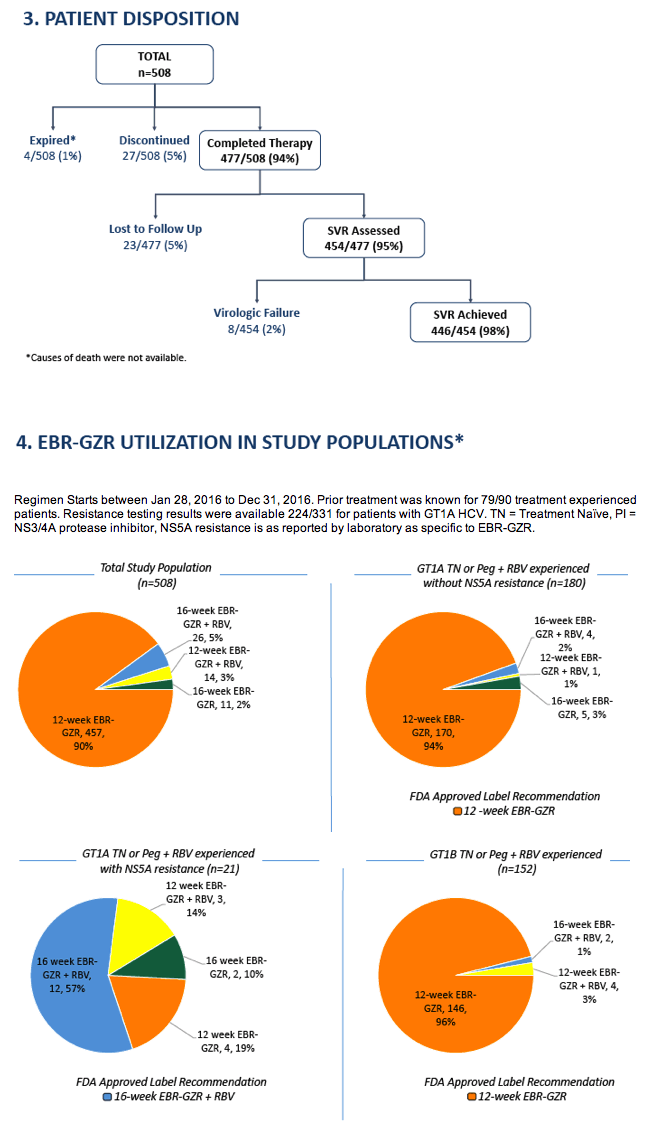
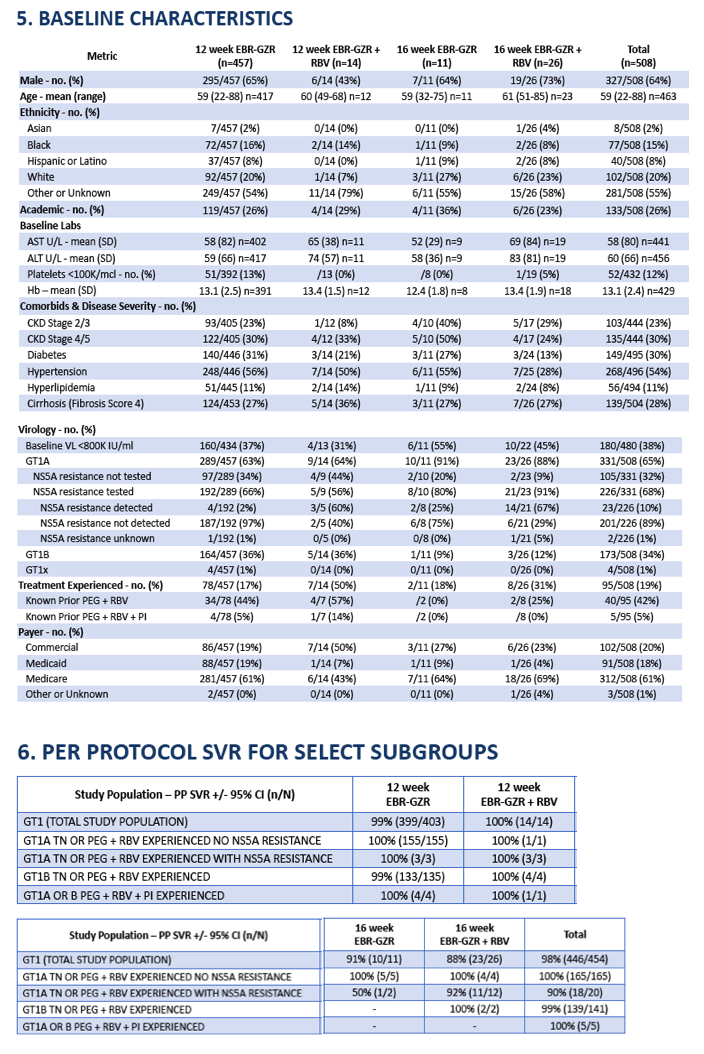
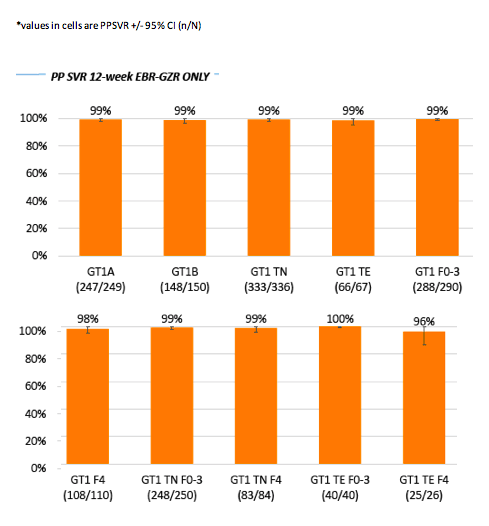
Impact of treatment duration and ribavirin (RBV) addition on real-world effectiveness of elbasvir/grazoprevir (EBR-GZR) in select patient subgroups with Genotype 1 (GT1) Chronic Hepatitis C (HCV): Retrospective Data Analyses from the Trio Network.
|
| |
| |
Reported by Jules Levin
AASLD: The Liver Meeting 2017, October 20-24, 2017, Washington, DC
C. NWANKWO1, B. BACON2, M. CURRY3, D. DIETERICH4, S.L. FLAMM5, K. KOWDLEY6, S. MILLIGAN7, N. TSAI8, Z. YOUNOSSI9, AND N. AFDHAL3
1Merck & Co., Inc., Kenilworth, NJ USA, 2Saint Louis University School of Medicine, St. Louis, MO , 3Beth Israel Deaconess Medical Center, Boston, MA , 4Icahn School of Medicine at Mount Sinai, New York, NY , 5Northwestern University Feinberg School of Medicine, Chicago IL , 6Liver Care Network, Swedish Medical Center, Seattle, WA , 7Trio Health Analytics, La Jolla, CA , 8Queens Medical Center, University of Hawaii, Honolulu, HI , and 9Center for Liver Diseases, Department of Medicine, Inova Fairfax Hospital, Falls Church, VA

|
| |
|
 |
 |
|
|