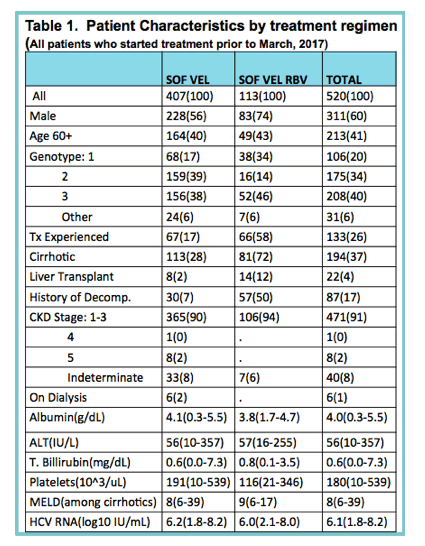
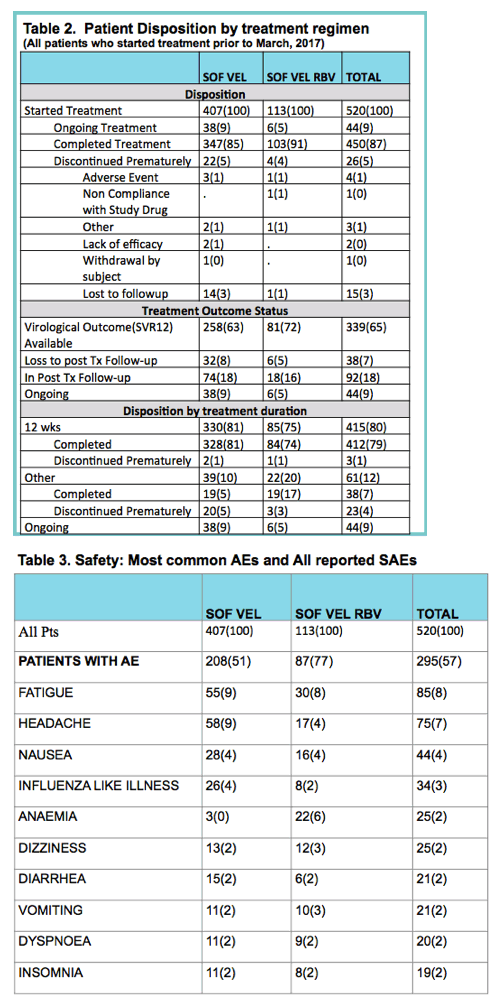
 |
 |
 |
| |
Safety and Efficacy of Velpatasvir and Sofosbuvir-based Regimens for the Treatment
of HCV Genotype 1-6: Results of the HCV-TARGET Study
|
| |
| |
Reported by Jules Levin
AASLD Annual Meeting 2017, Washington, DC, USA, October 20-24
Landis, CS; Sulkowski, MS; Reau, N; Lutchman, GA; Vainorius, M; Welzel, T; Zeuzem, S; Khalili, M; Nelson, DR; Lok, AS; Fried, MW; Dong, M; Terrault, N
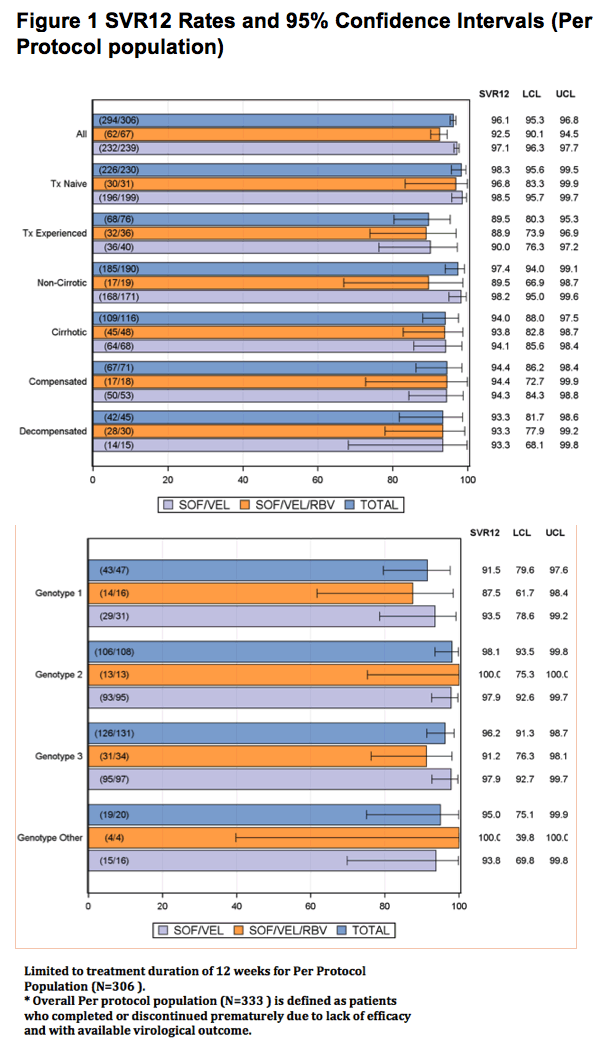
Limited to treatment duration of 12 weeks for Per Protocol Population (N=306 ).
* Overall Per protocol population (N=333 ) is defined as patients who completed or discontinued prematurely due to lack of efficacy and with available virological outcome.





|
| |
|
 |
 |
|
|