 |
 |
 |
| |
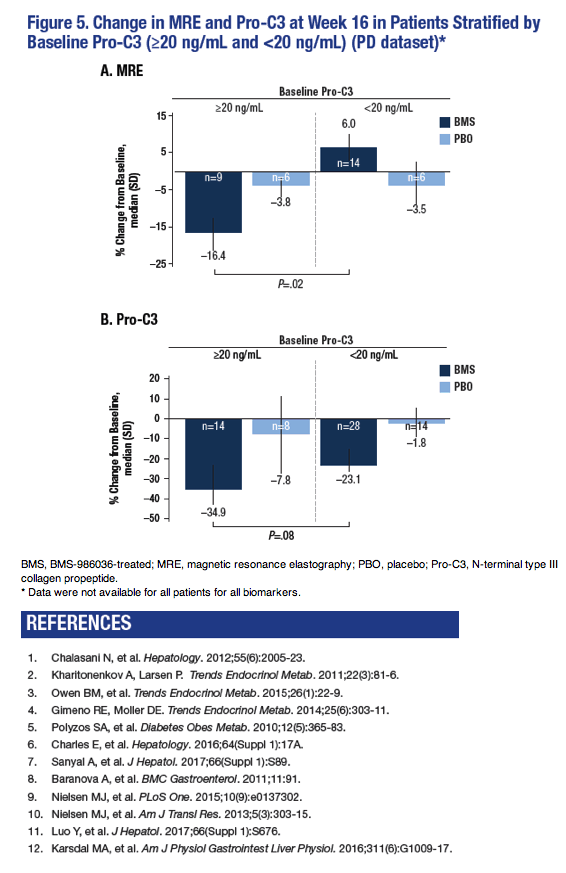
Baseline Serum Pro-C3 Predicts Response to BMS-986036 (PEG-FGF21): A Secondary Analysis of
a Multi-Center Clinical Trial in Non-Alcoholic Steatohepatitis (NASH)
|
| |
| |
AASLD: BMS-986036 (PEGylated FGF21) in Patients with Non-Alcoholic Steatohepatitis: A Phase 2 Study - (10/26/17)
Reported by Jules Levin
American Association for the Study of Liver Diseases (AASLD 2017), Washington, DC, October 20-24, 2017
Manal F. Abdelmalek,1 Edgar D. Charles,2 Brent Neuschwander-Tetri,3 Dina
Halegoua-DeMarzio,4 Arun Sanyal,5 Robert Herring,6 Eric Lawitz,7 Yuping Dong,2 Shuyan Du,2 Robert Gagnon,2 Yi Luo,2 Rose Christian2
1Duke University, Durham, NC, USA; 2Bristol-Myers Squibb, Lawrenceville, NJ, USA; 3Saint Louis University, Saint Louis, MO, USA; 4Thomas Jefferson University, Philadelphia, PA, USA; 5Virginia Commonwealth University, Richmond, VA, USA; 6Quality Medical Research, Nashville, TN, USA; 7Texas Liver Institute, University of Texas Health, San Antonio, TX, USA


|
| |
|
 |
 |
|
|