 |
 |
 |
| |
Prospective, Randomized Assessment of HBV-associated and Other Clinical Outcome Events During Long-Term Therapy With Entecavir or Other HBV Nucleos(t)ide Analogues in Patients With Chronic HBV Infection
|
| |
| |
Reported by Jules Levin
AASLD Annual Meeting 2017, Washington, DC, USA, October 20-24
J-L Hou,1W Zhao,2C-H Lee,3H-W Hann,4C-Y Peng,5T Tanwandee,6V Morozov,7CP Berg,8JD Sollano,9A Streinu-Cercel,10H Cheinquer,11A Thiry,12EL Cooney,13S-G Lim14
1Nanfang Hospital, Guangzhou, China; 22nd Hospital Nanjing, Nanjing, China; 3Daegu Catholic University Hospital, Daegu, Republic of Korea; 4Thomas Jefferson University Hospital, Philadelphia, PA; 5China Medical University Hospital, Taichung, Taiwan; 6Faculty of Medicine SirirajHospital, Bangkok, Thailand; 7LLC Medical Company " Hepatolog", Samara, Russian Federation; 8Universitaetsklinikum Tuebingen, Tuebingen, Germany; 9Cardinal Santos Medical Center, Manila, Philippines; 10Carol Davila University of Medicine and Pharmacy, National Institute for Infectious Diseases "Prof.Dr. MateiBals", Bucharest, Romania; 11Hospital de Clinicasde Porto Alegre, Porto Alegre, Brazil; 12Bristol-Myers Squibb Inc., Princeton, NJ; 13Bristol-Myers Squibb Inc., Wallingford, CT; 14National University Health System, National University of Singapore, Singapore.


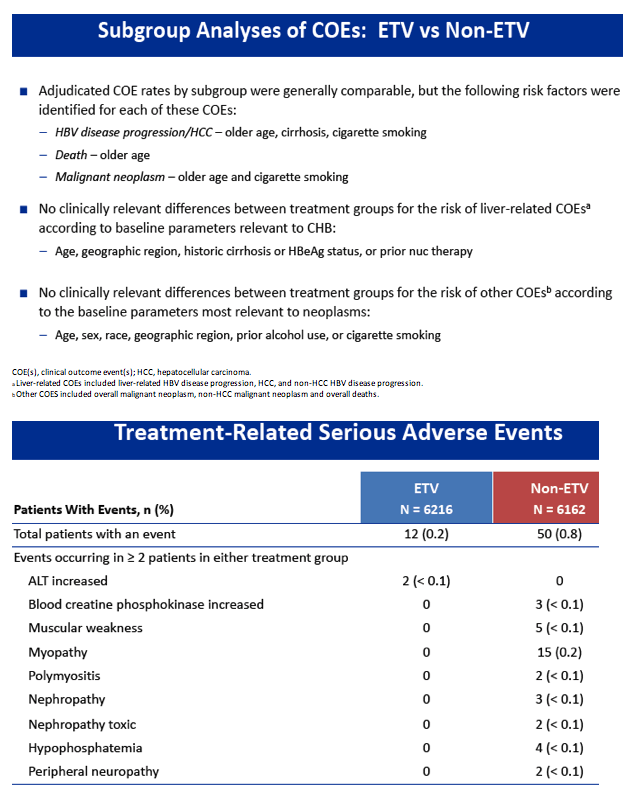
|
| |
|
 |
 |
|
|