 |
 |
 |
| |
Phase 1b Clinical trial of TG1050, a novel HBV-targeted immunotherapy, in NUC suppressed chronic hepatitis B patients: safety and early immunological data following single administration
|
| |
| |
Reported by Jules Levin
AASLD Oct 2017

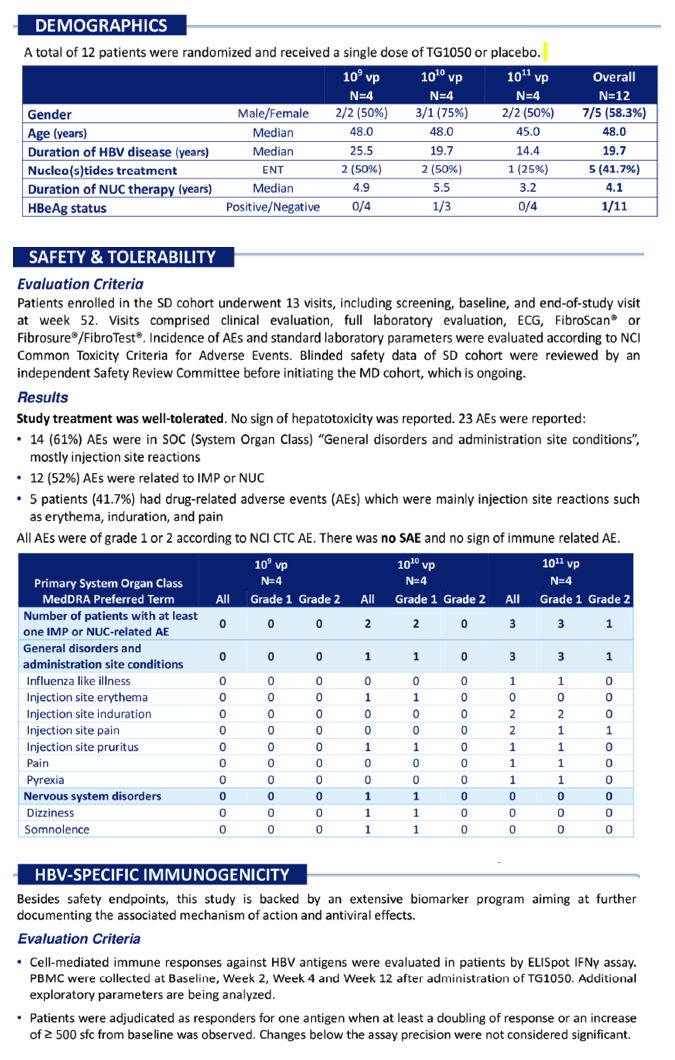
Safety: A subcutaneous SD of TG1050 in NUC suppressed CHB patients was safe over 3 DLs and did not have any negative impact on disease control. This observation, in a particularly sensitive population(no pre-immunity against the adenoviral vector), demonstrated a robust safety profile of TG1050.
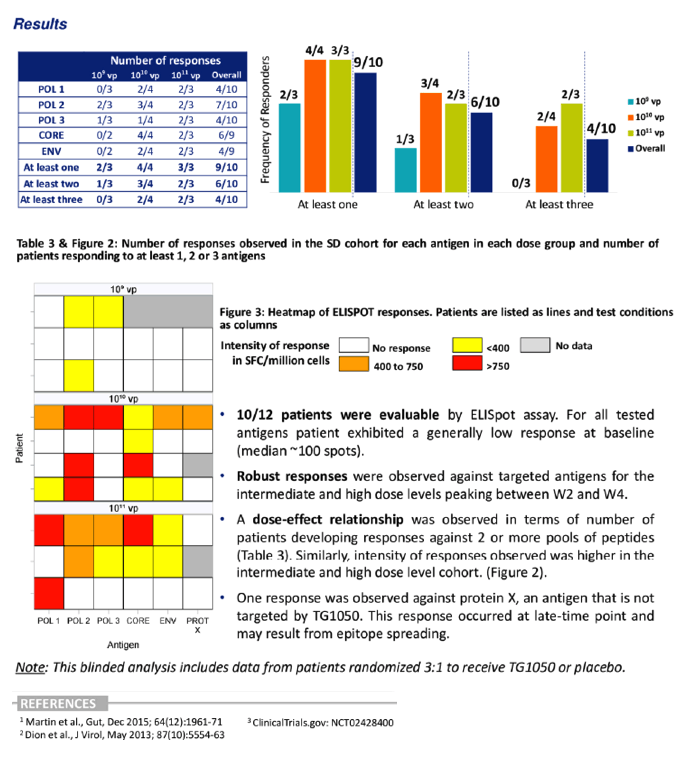
Immunogenecity: Early analysis indicated that TG1050 induced a robust cell-mediated response against the HBV polymerase, the CORE protein and the Envelope protein. A dose-effect relationship was observed across the 3 DLs in terms of number of responders.
Perspective: Additional immune and HBV related markers are being assessed and will be published after unblinding.
Recruitment has been completed in the ongoing MD cohort. 36 CHB patients were randomized regardless of anti-Adenovirus serology status: 27 patients received 3 doses of TG1050 at the 3 DLs and 9 patients received matching placebo.

|
| |
|
 |
 |
|
|