 |
 |
 |
| |
In Vitro -In Vivo Evaluation of a Biodegradable Implant Containing TAF for HIV PrEP
|
| |
| |
Reported by Jules Levin
CROI 2017 Feb 14-17 Seattle, WA
Phillip G Durham1, Greg Gatto1, Leah Johnson1, Mark A Marzinke2, Stephanie Swarner1, Ginger Rothrock1, TejalDesai3,
Ian McGowan4, Ida Washington5, Ariane van der Straten6
1RTI International, Durham, NC, USA, 2Johns Hopkins University School of Medicine, Baltimore, MD, USA, 3University of California, San Francisco, San Francisco, CA, USA, 4University of Pittsburgh, Pittsburgh, PA, USA, 5Magee-Womens Research Institute, Pittsburgh, PA, USA, 6RTI International, San Francisco, CA, USA

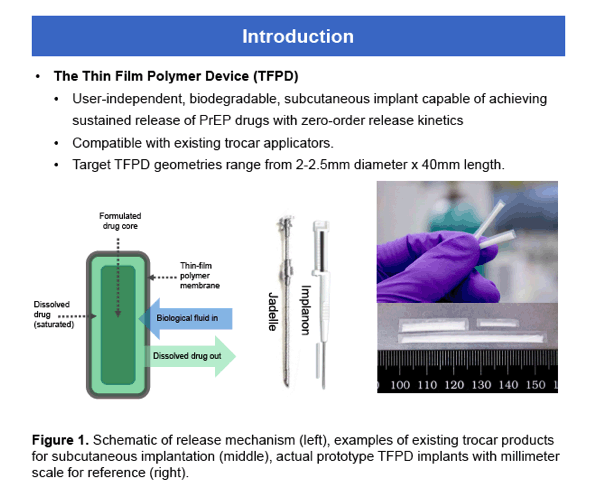

Figure 3. Cumulative in vitro release of TAF from devices measured by UV-Vis absorbance in PBS over 30 days (left). Cumulative release as a percentage of the total release from devices over 30 days (right).
The in vitro devices released approximately 50% faster than intended. The actual in vitro release was quantified over the linearrelease period, determined as having an R2value > 0.95. These actual values were used for determining the in vitro/in vivo correlation, and are reflected in Table 1.
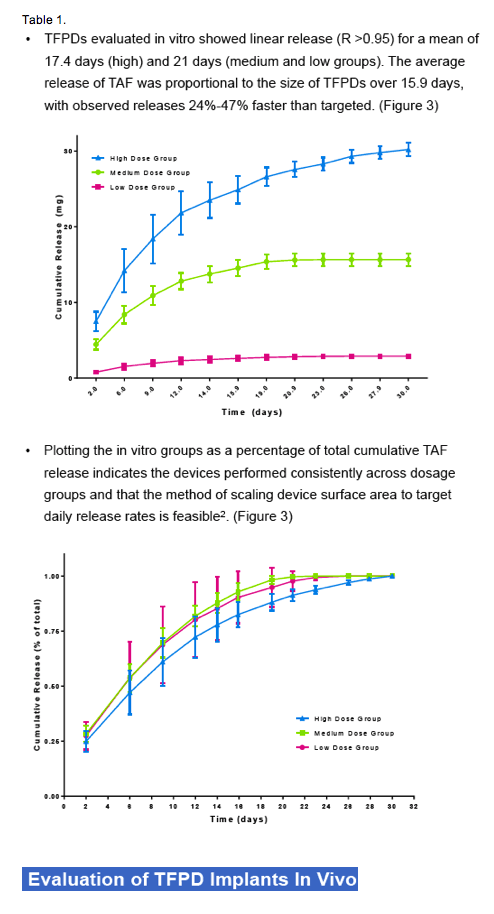
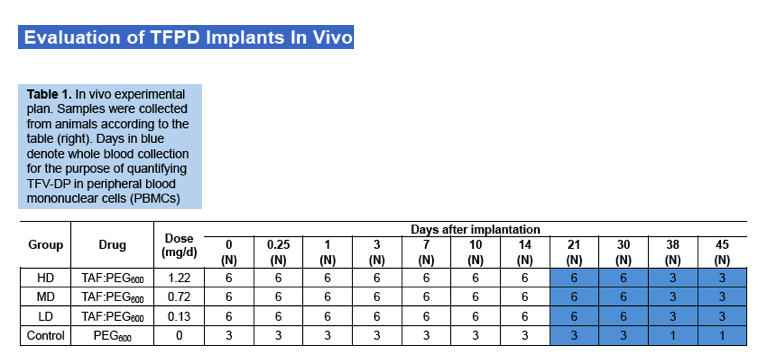
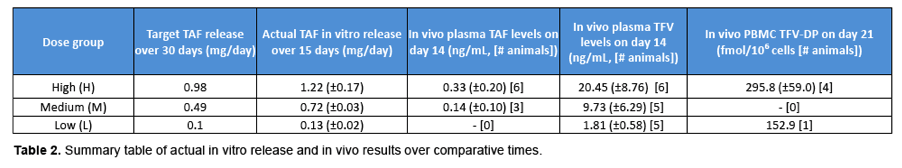
Pharmacokinetic results show sustained TAF and TFV levels for a period of time after implantation. (Figure 4) For the high dosage group, TAF was detectable as early as 6 hours after implantation and plasma concentrations rose initially through day 1 and remained fairly constant through day 14, at approximately 0.3 ng/mL.TFV levels in this dosage group were sustained over the same period, at a concentration of approximately 20 ng/mL.In the medium dose group, a similar trend was observed, with slightly higher variability. TAF and TFV concentrations for this group were maintained at approximately 0.15 ng/mL and 13 ng/mL, respectively, through day 14. The low dosage group resulted in the lowest TAF and TFV levels as expected. Many of the TAF samples were below the limit of detection, but corresponding TFV levels were measurable and appear to be sustained for a period of 14 days (5 animals) at a plasma concentration of approximately 3 ng/mL.TFV-DP in PBMCs was quantifiable on day 21in 4 animals of the high dosage group with an average concentration of 296 ±59 fmol/106cells (1483 ±295 nM). (Table 2)
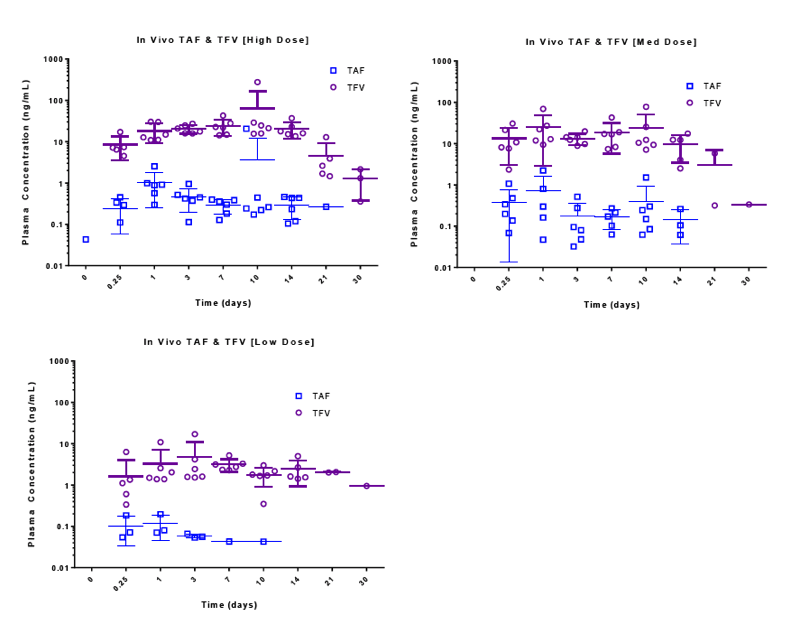
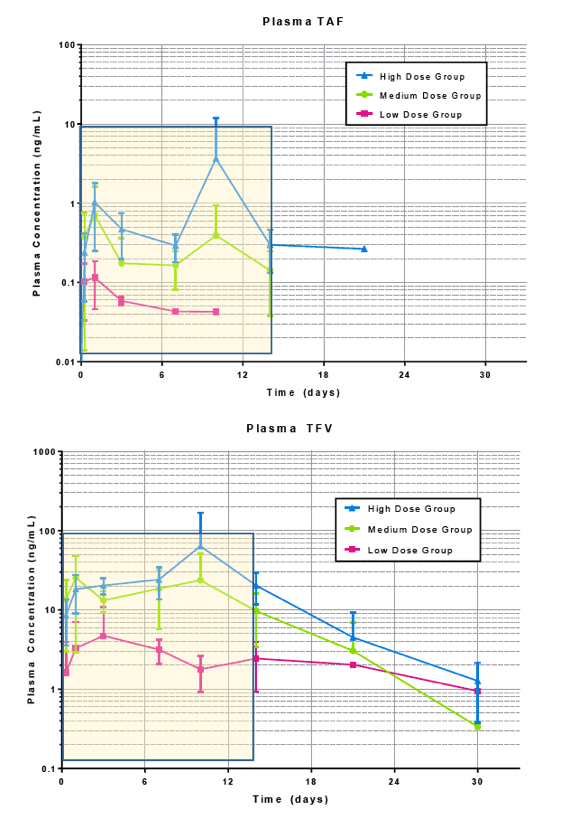
Figure 4. In vivo plasma concentrations of TAF and TFV (top) for each dosing group (high, medium and low, left, middle and right respectively) over a period of 30 days and average data presented on a linear time scale (bottom) for TAF (left) and TFV (right). Highlighted box indicates plasma data used for the correlation
Following the sustained period of release, plasma TAF concentrations decreased in all dosage groups to below the limit of detection in the measurement window, with only one animal quantifiable in high dose group at day 21, TAF was undetectable in the medium dose group at day 21, and undetectable in the low dose group at day 14. TFV concentrations remained detectable in some animals for each dosing group through day 30.
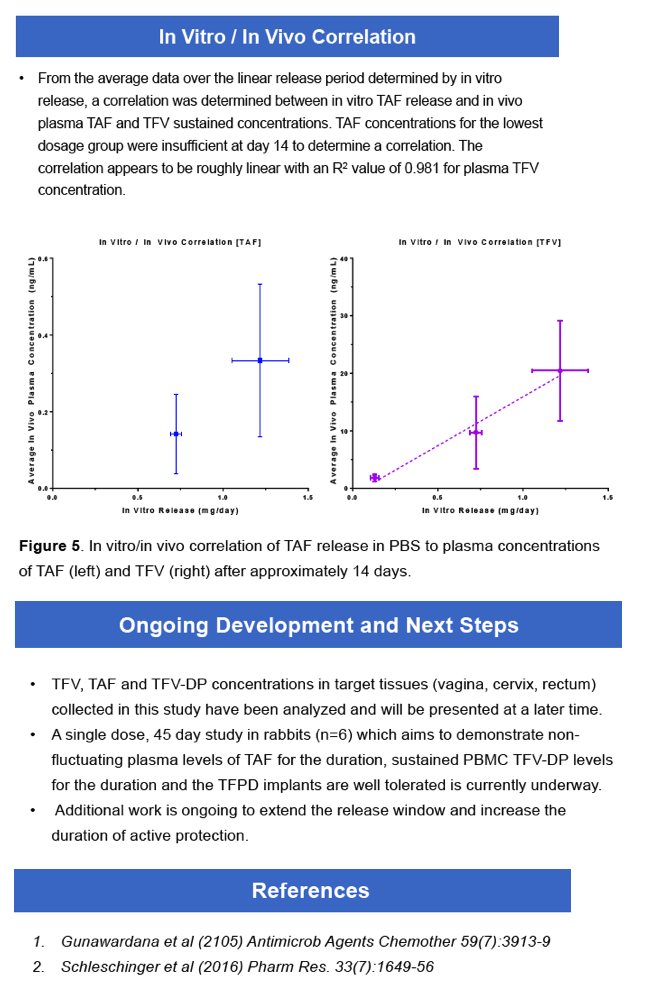

|
| |
|
 |
 |
|
|