 |
 |
 |
| |
IFN-FREE THERAPY IS EFFECTIVE AND SAFE FOR HCV RECURRENCE IN LT HCV/HIV CO-INFECTION
|
| |
| |
Reported by Jules Levin
CROI 2017 Feb 14-16 Seattle WA
C. Manzardo1, M.C. Londono1, S. del Campo2, L. Castells3, V. Aguilera4, J.R. Fernandez5, J.Calvo-Pulido6, J.Penafiel1, A. Rimola1, J.M. Miro1, FIPSE Investigators
1Hospital Clinic-IDIBAPS, University of Barcelona, Barcelona, Spain; 2 Hospital Universitario Ramon y Cajal, Madrid, Spain; 3 Hospital Universitario Vall d'Hebron, Barcelona, Spain; 4 Hospital Universitario La Fe, Valencia, Spain; 5
Hospital Universitario de Cruces, Bilbao, Spain; 6 Hospital Doce de Octubre, Madrid, Spain

Abstract Body:
Interferon (IFN)-based therapy against hepatitis C virus (HCV) recurrence after liver transplantation (LT) has poor effectiveness and tolerability both in HCV-mono-infected (≈30% of sustained virological response [SVR]) and HIV-HCV co-infected LT recipients (≈20% of SVR). Only small case series have reported on the use of direct antiviral agents (DAAs) in LT HCV/HIV co-infected recipients. The aim of this study is to report the effectiveness and safety of IFN-free regimens in a nationwide cohort of HIV HCV co-infected individuals having undergone LT.
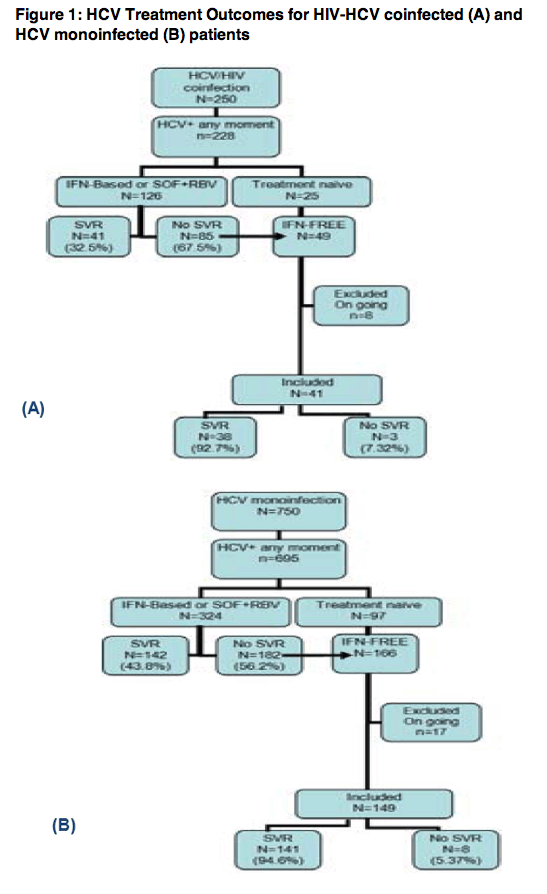
A prospective, multicenter cohort study, including HCV/HIV co-infected LT patients who received IFN-free treatment for recurrent hepatitis C with two or more DAAs (patients receiving DAAs with IFN or Sofosbuvir (SOF) plus ribavirine (RBV) were excluded). For comparison, we included a matched cohort of HCV mono-infected patients who received similar treatment for recurrent HCV. Only patients reaching a follow-up of at least 12 weeks after the end of treatment were analyzed.
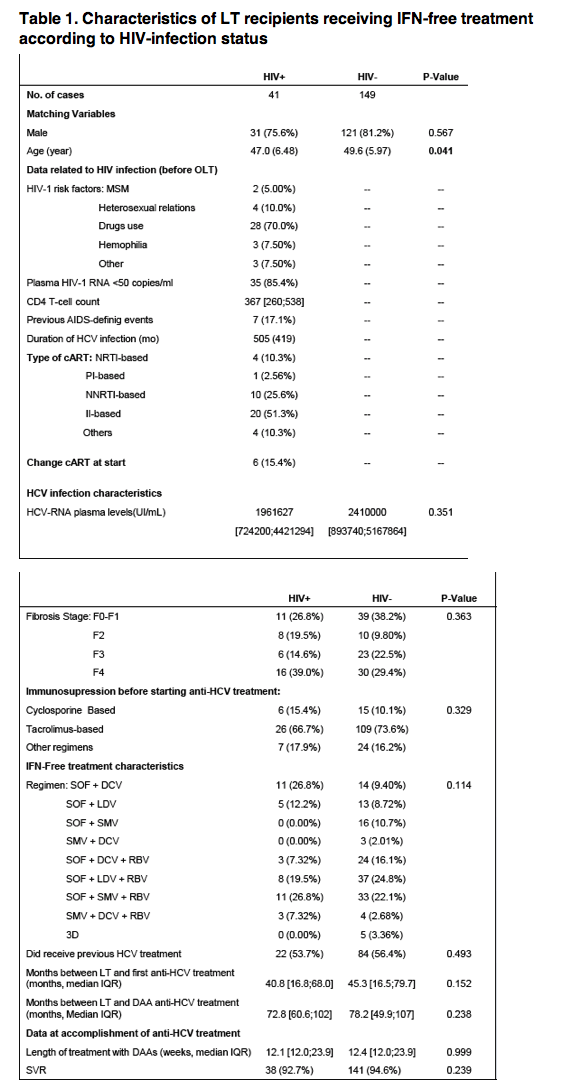
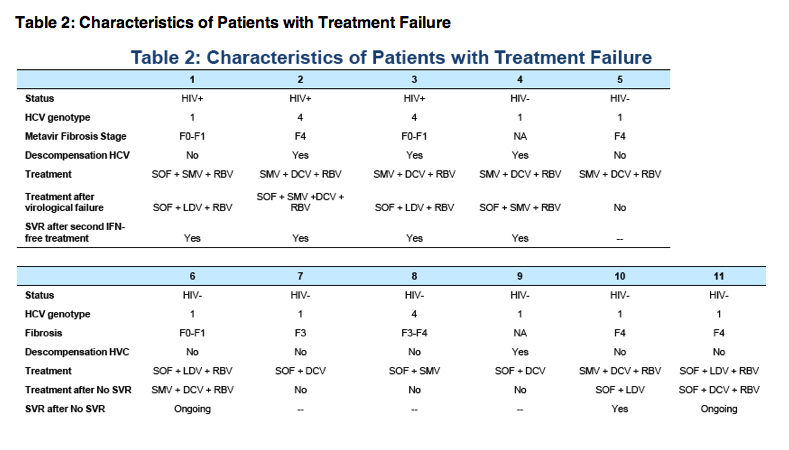
Among patients with post-LT HCV recurrence in the FIPSE cohort, 39/228 (17%) of HIV+ and 118/693 (17%) of HCV mono-infected patients received IFN-free regimens containing at least 2 DAAs +/- RBV after a median (IQR) of 42 (16-72) months after LT. No differences in demographics and pre- or peri-transplant characteristics were observed. For HIV-infected individuals, median (IQR) CD4 T-cell count was 367 (200-465) cells/μL. All patients received antiretroviral treatment (ART) and 33 (85%) had a plasma HIV-RNA <50 copies/mL; 19 (48%) were receiving ART based on a non-boosted integrase inhibitor. SVR rates were high (95%) and similar in the HIV-infected and uninfected cohorts (Table). Of note, two failures in HIV+ patients were observed for genotype 4 (SVR 75% vs. 100% among other genotypes, p=0.038). No significant differences in SVR rates among genotypes were observed for HCV mono-infected individuals. Treatment was well tolerated. Only one patient in the mono-infected cohort died because treatment was started in the advanced decompensated cirrhosis stage.
IFN-free regimens for post LT HCV recurrence in HIV infected individuals of our national cohort were highly effective and well tolerated, with results comparable to HCV mono-infected patients. Newer treatment options will probably improve efficacy for genotype 4 in co-infected LT recipients.
|
| |
|
 |
 |
|
|