 |
 |
 |
| |
Earlier [cd4 350-500] Versus Delayed Antiretroviral
Therapy Initiation and Risk of Cancer
|
| |
| |
--------------------
Cancer Risk Almost Halved With Earlier Antiretroviral Therapy Start
Conference on Retroviruses and Opportunistic Infections (CROI), February 13-16, 2017, Seattle
Mark Mascolini
Starting antiretroviral therapy (ART) within 6 months of reaching a 350-500 CD4 count rather than later almost halved the risk of any cancer, according to a 10,000-person analysis in the North American AIDS Cohort Collaboration on Research and Design (NA-ACCORD) [1]. The analysis also linked earlier ART to a lower risk of virus-unrelated non-AIDS cancer or AIDS-defining cancer.
US and international guidelines recommend starting ART immediately after HIV diagnosis, regardless of CD4 count. As more clinicians adopt this practice, incidence of AIDS and non-AIDS diseases may change. NA-ACCORD researchers conducted this study to assess the impact of early ART (within 6 months of a 350-500 CD4 count) and delayed ART (no ART or starting after 6 months).
The NA-ACCORD population consists of initially antiretroviral-naive, AIDS-free adults. Follow-up in this analysis continued from the time cohort members reached a CD4 count of 350-500 until a new cancer diagnosis, death, loss to follow-up, or 2009. The researchers confirmed new cancer diagnoses by chart review or cancer registry linkage. They considered four groups of cancers: (1) any cancer, (2) virus-related non-AIDS cancer (anal, liver, Hodgkin lymphoma, or HPV-related oral/pharyngeal, penile, vaginal, or vulvar cancer), (3) AIDS-defining cancer (Kaposi sarcoma, non-Hodgkin lymphoma, cervical cancer), and (4) virus-unrelated non-AIDS cancers (all others).
The researchers used marginal structural modeling based on inverse probability weighting to compare the impact of early ART versus later ART on cancer incidence. Primary results emulated an intention-to-treat randomized trial or a per-protocol trial analysis. Variables contributing to weights were baseline age, sex, cohort, entry year, race/ethnicity, HIV risk, ever smoking, ever HBV or HCV positive, among others.
The study group included 10,434 HIV-positive people, 84% of them men, with a median age of 39 years. Similar proportions were white (46%) or black (43%), while 7% were Hispanic. Almost two thirds, 65%, ever smoked, 18% had been exposed to HCV and 3% to HBV. Median baseline CD4 count stood at 431.
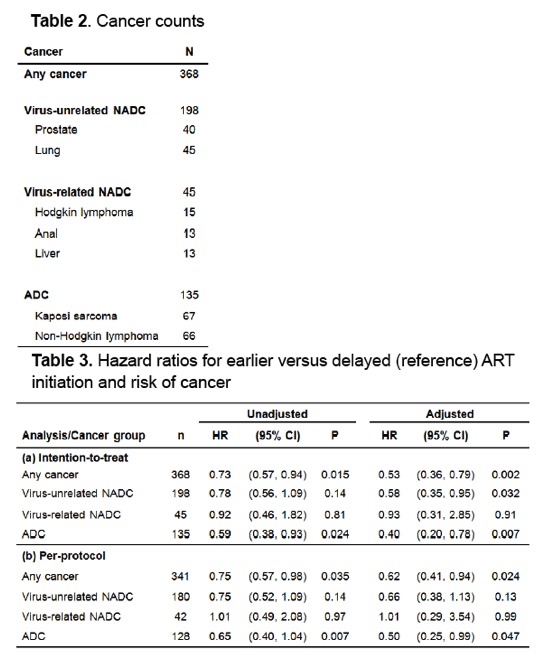
During follow-up cancers developed in 368 people, including 198 virus-unrelated non-AIDS cancers (45 lung, 40 prostate), 45 virus-related non-AIDS cancers (15 Hodgkin, 13 anal, 13 liver), and 135 AIDS-defining cancers (67 Kaposi, 66 non-Hodgkin).
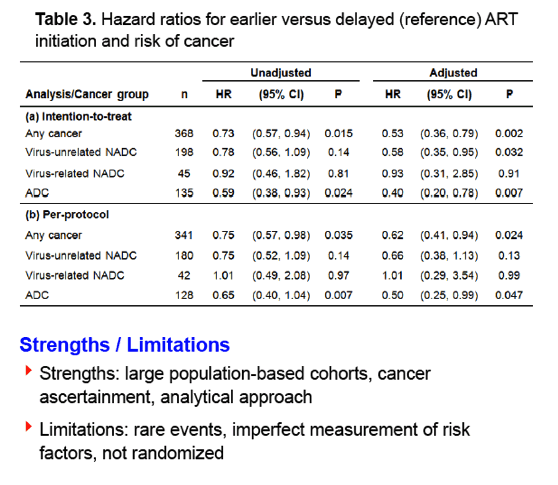
The adjusted intention-to-treat analysis linked early ART to lower risk of three cancer groups at the following hazard ratios (HR) and 95% confidence intervals (CI):
-- Any cancer: HR 0.53, 95% CI 0.36 to 0.79, P = 0.002
-- Virus-unrelated non-AIDS cancer: HR 0.58, 95% CI 0.35 to 0.95, P = 0.032
-- AIDS-defining cancer: HR 0.40, 95% CI 0.20 to 0.78, P = 0.007
The adjusted per-protocol analysis tied early ART to lower risk of two cancer groups:
-- Any cancer: HR 0.62, 95% CI 0.41 to 0.94, P = 0.024
-- AIDS-defining cancer: HR 0.50, 95% CI 0.25 to 0.99, P = 0.047
The researchers caution that their analysis is limited by the low number of cancer diagnoses, imperfect measurement of risk factors, and lack of randomization. With those limitations in mind, they propose their findings "suggest that the burden of certain cancer groups may be substantially reduced as more HIV providers and patients adopt ART guidelines for immediate initiation."
Reference
1. Silverberg MJ, Leyden W, Lin H, et al. Earlier versus delayed antiretroviral therapy initiation and risk of cancer. Conference on Retroviruses and Opportunistic Infections (CROI), February 13-16, 2017, Seattle. Abstract 598.
--------------------
Earlier Versus Delayed Antiretroviral Therapy Initiation and Risk of Cancer
Michael J Silverberg1, Wendy Leyden1, Haiqun Lin2, Li Qin3, Chad J Achenbach4, Gypsyamber D'Souza5, Eric A Engels6, Charles Rabkin6, Keri N Althoff5, Romain Neugebauer1
1-Kaiser Permanente, Oakland, CA; 2-Yale School of Public Health, New Haven, CT; 3-Yale School of Medicine, New Haven, CT; 4-Northwestern University Feinberg School of Medicine, Chicago, IL; 5-Johns Hopkins Bloomberg School of Public Health, Baltimore, MD; 6-National Cancer Institute, Rockville, MD


|
| |
|
 |
 |
|
|