 |
 |
 |
| |
TLR7 Agonist Treatment of SIV+ Monkeys on ART Can Lead to Complete Viral Remission
|
| |
| |
Reported by Jules Levin
CROI 2017 Feb 14-16 Seattle, WA
So-Yon Lim1, Christa E. Osuna1, Joseph Hesselgesser2, Alison L. Hill3, Michael D. Miller2, Tomas Cihlar2, Romas Geleziunas2, William A. Lee 2 and James B. Whitney1,4
1Center for Virology and Vaccine Research, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, MA 02215, USA; 2 Gilead Sciences, Inc. Foster City, CA 94404; 3Program for Evolutionary Dynamics, Harvard University, Cambridge, MA 02138 USA; 4Ragon Institute of MGH, MIT, and Harvard, Cambridge, MA 02139, USA


Abstract Body:
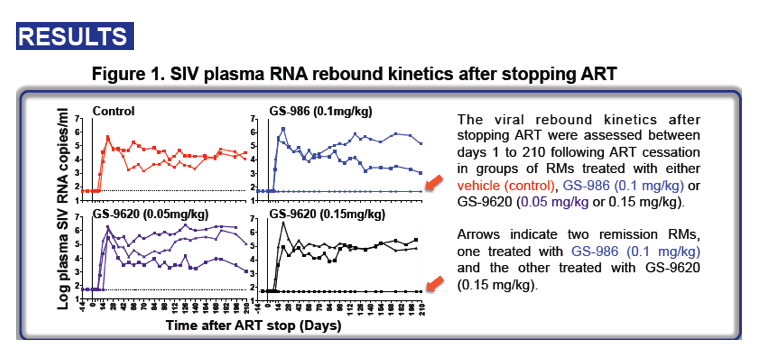
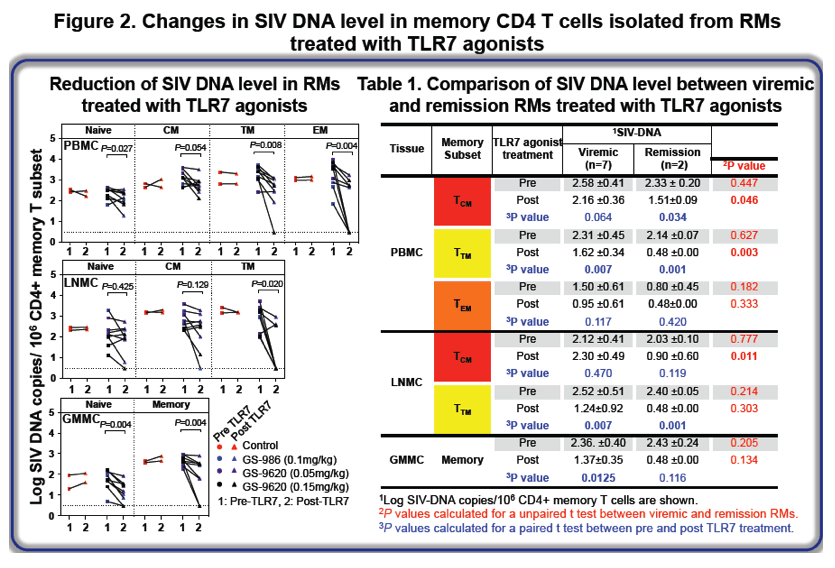
The persistence of an HIV-1 reservoir in patients on antiretroviral therapy (ART) represents the major obstacle to HIV remission. We have previously reported that oral TLR7 agonists (GS-9620 and GS-986) can induce transient viremia in SIV-infected, ART-treated rhesus monkeys (RMs). After ART release of nine TLR7 agonist-treated RMs, complete remission was noted in two RMs, and two other RMs have exhibited significant control of recrudescent viremia.
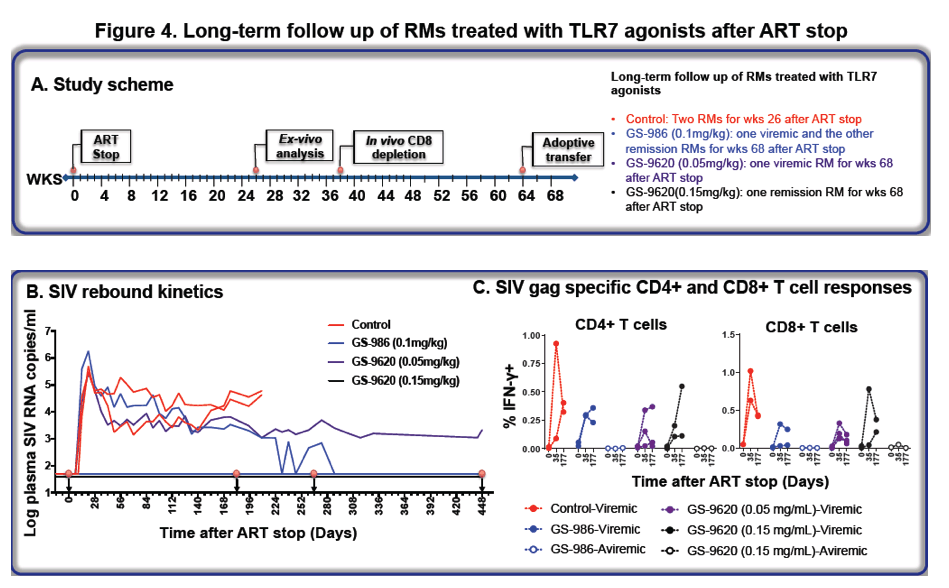
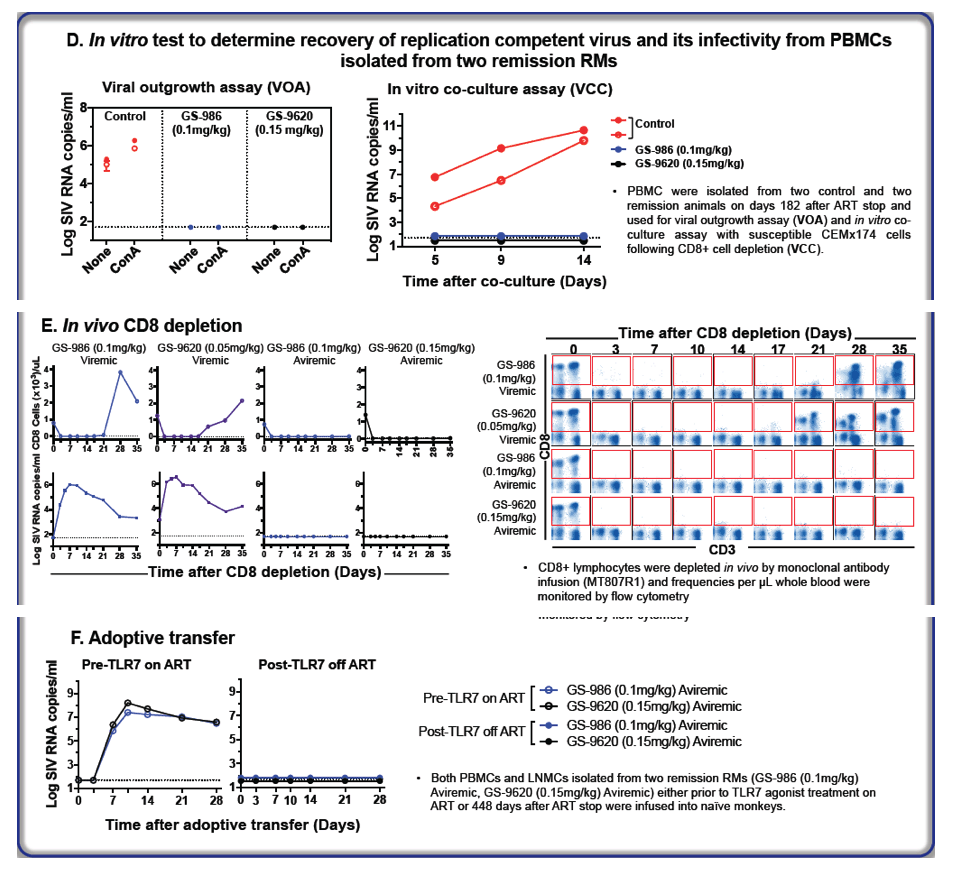
After repeated GS-9620 or GS-986 adminsitration and ART stop, we have conducted a long-term follow up of two 'remission' RMs. We also monitored 2 RMs that had rebound viremia after ART stop, as controls. We assessed multiple endpoints including: viral outgrowth (VOA) and viral co-culture (VCC) using lymph node mononuclear cells (LNMC) and PBMC. We assessed SIV-specific T cell responses at multiple time points. We also conducted in vivo CD8+ T cell depletion in remission and viremic control RMs. Finally, we adoptively transferred PBMC and LNMC cells into SIV naïve RMs, from remission RMs prior to TLR7 agonist treatment. In parallel, we also transferred PBMC and LNMC, into SIV naïve RMs, isolated from remisson RMs after GS-9620 or GS-986 treatment and ART stop.
To date, both TLR7 agonist-treated RMs have remained aviremic for more than 1 year after ART cessation. Longitudinal assesment of VOA or VCC in the two remission RMs was uniformly negative, whereas viremic RMs scored consistently postive. Longitudal SIV-specific immune monitoring revealed sustained responses in viremic RMs, but no detectable SIV-specific T cell responses in remission RMs. In vivo CD8+ T cell depletion did not induce rebound viremia in remission RMs, but viremic control RMs exhibited significant increases in SIV RNA levels that later waned as CD8+ T cells recovered. Finally, the adoptive transfer of PBMC and LNMC samples (prior to TLR7 agonist treamtent) induced a persistent SIV infection into naïve RMs. Adoptive transfer of PBMC and LMNC cells from remission RMs did not induce SIV infection in naïve recipients.
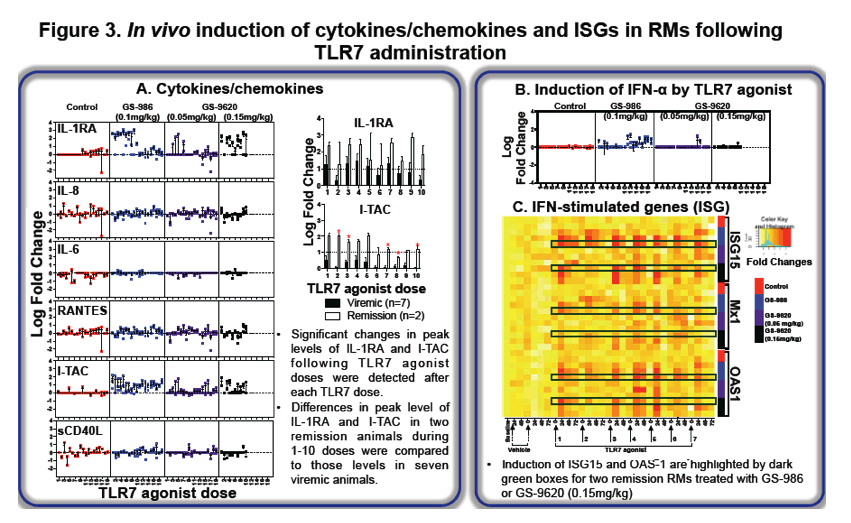
Administration of GS-9620 or GS-986 to SIV+ ART-suppressed RM is safe, induces transient viremia and impacts SIV DNA levels. GS-986 or GS-9620 treatment can delay viral rebound or induce durable long-term remisison after ART cessation in some RMs. These novel findings highlight a possible mechanism of SIV remission after ART cessation and underscore the need for continued investigation of GS-9620 in HIV-1 infected patients on ART.


|
| |
|
 |
 |
|
|