 |
 |
 |
| |
HCV Prevalence in HIV Patients Falls Nearly 50% in Spain in 1 Year
|
| |
| |
16th European AIDS Conference, October 25-27, 2017. Milan
Mark Mascolini
Prevalence of active HCV infection among people with HIV stood at 11.7% in Spain in 2016, nearly a 50% drop from 2015 [1]. A national team of HCV/HIV researchers proposed that HCV can be eliminated in Spain's HIV population if direct-acting antiviral (DAA) therapy becomes available to all coinfected people.
The researchers noted that several factors contribute to changing epidemiology of HCV/HIV infection in Spain, including (1) a drop in injection drug use as an HIV transmission route, (2) emergence of sexually transmitted HCV infection in men who have sex with men, (3) availability of curative DAAs, and (4) higher mortality with HCV/HIV than with HIV alone.
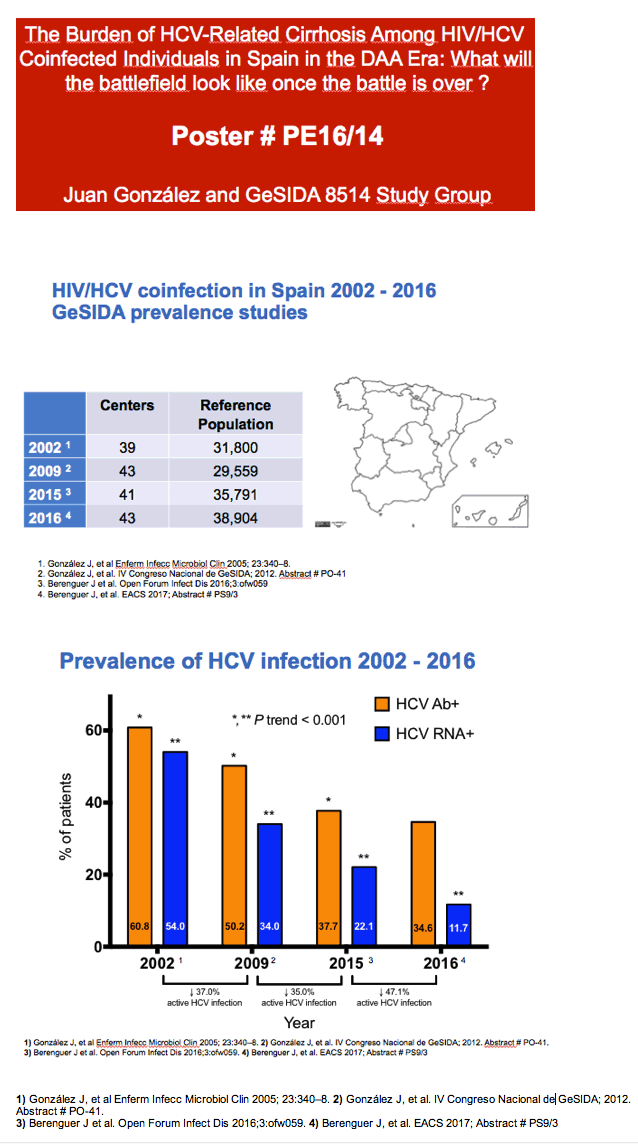
With these variables in mind, the research team conducted this analysis to assess HCV/HIV prevalence in Spain in 2016, to define clinical characteristics of HCV/HIV coinfection, and to compare results with similar coinfection studies done in 2002, 2009, and 2015. This nationwide cross-sectional analysis involved HIV-positive people in active follow-up at participating centers in October and November 2016. The researchers used simple random sampling to select participants from each center.
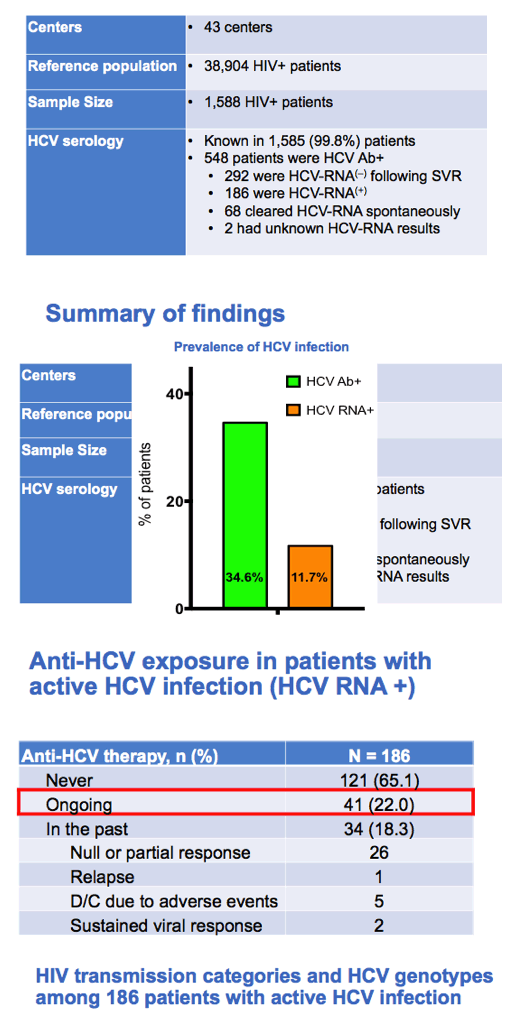
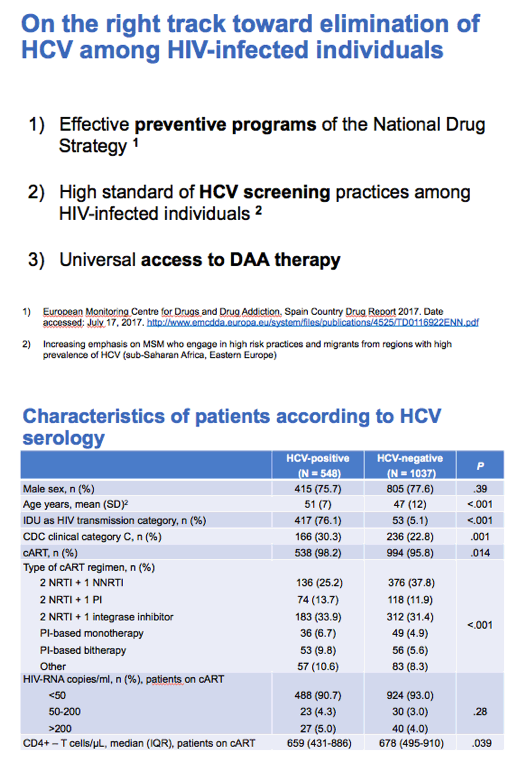
From 43 centers, 38,904 HIV patients yielded a study sample of 1588 people. Among 1585 people with HCV serology, 548 (34.6%) were HCV-antibody positive and 186 (11.7%) HCV RNA positive. Among the 186 people positive for HCV RNA, 121 (65%) never had anti-HCV therapy, 41 (22%) were receiving therapy at the time of the analysis, and 34 (18%) received treatment in the past (only 2 achieving sustained virologic response, SVR).
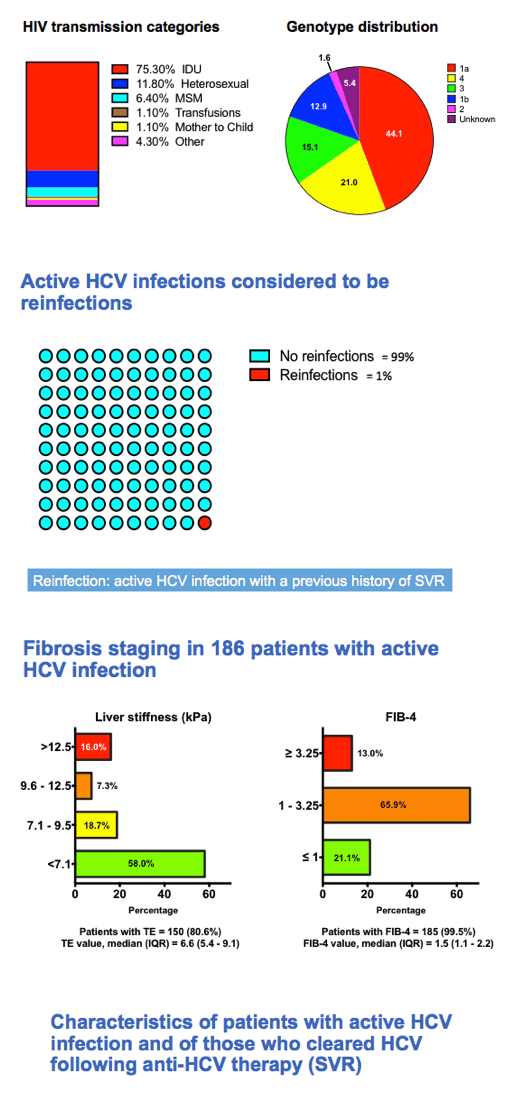
In this 186-person HCV RNA-positive group, drug injecting explained 75.3% of HIV transmissions, heterosexual sex 11.8%, gay sex 6.4%, and mother-to-child transmission and other routes the remaining transmissions. The researchers figured that only 1% of active HCV infections represented reinfection. Fibrosis estimated by transient elastography (TE) or FIB-4 was mild or moderate in a large majority of HCV RNA-positive people: By TE liver stiffness (kPa) was above 12.5 in 16% of 150 people assessed, 9.6 to 12.5 in 7.3%, 7.1 to 9.5 in 18.7%, and below 7.1 in 58%. By FIB-4 only 13% of 185 people assessed were at or above 3.25, 65.9% were between 1 and 3.25, and 21.1% were at or below 1.
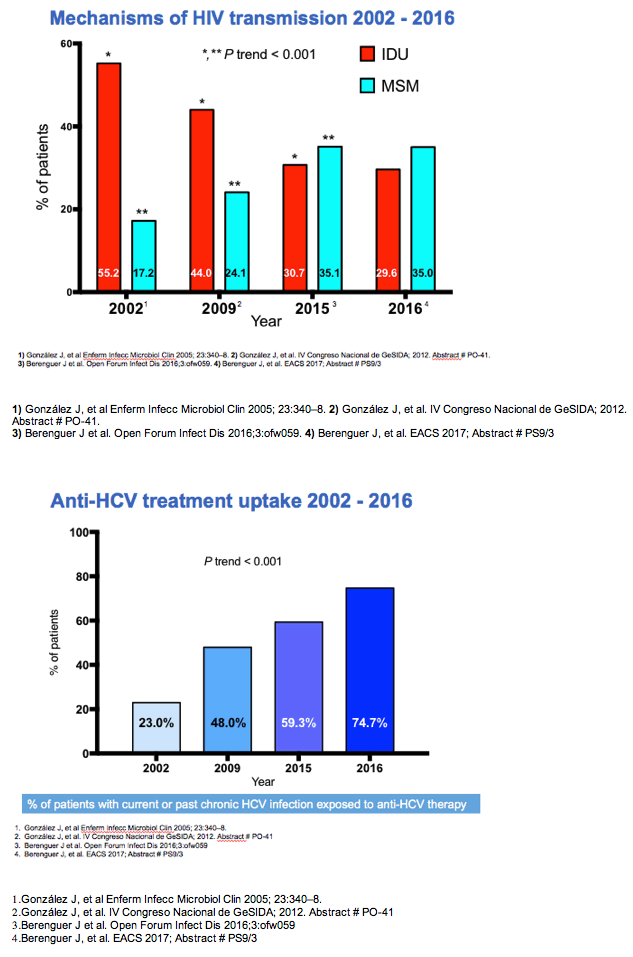
Among HCV/HIV-coinfected people, HCV RNA positivity fell from a prevalence of 54% in 2002, to 34% in 2009, to 22.1% in 2015, and to 11.7% in 2016 (a 47.1% drop from 2015, P < 0.001 for 2002-2016 trend). Injection drug use accounted for 55.2% of HIV transmissions in 2002, for 44% in 2009, for 30.7% in 2015, and for 29.6% in 2016. In those same four years, proportions of HIV transmission attributed to sex between men climbed from 17.2% to 24.1% to 35.1% and to 35.0%. Proportions of people with current or past chronic HCV infection who received anti-HCV therapy rose from 23% in 2002, to 48% in 2009, to 59.3% in 2015, and to 74.7% in 2016 (P < 0.001 for trend).
The researchers believe their findings indicate Spain is "on the right track toward elimination of HCV among HIV-infected individuals." In addition, the country put in place effective HCV prevention programs, promotes quality HCV screening programs for people with HIV, and offers universal access to DAA therapy. They credit wider DAA use for the rapidly waning HCV rate in people with HIV. "The elimination of HCV among HIV-infected individuals in Spain seems achievable in the short term," they proposed, "once DAA-based therapy is accessible to all coinfected patients."
Reference
1. Berenguer J, Gonzalez J, Vivanco MJ, et al. HIV/HCV coinfection in Spain: elimination is a stone´s throw away. 16th European AIDS Conference. October 25-27, 2017. Milan. Abstract PS9/3.



1) Gonzalez J, et al Enferm Infecc Microbiol Clin 2005; 23:340-8. 2) Gonzalez J, et al. IV Congreso Nacional de GeSIDA; 2012. Abstract # PO-41.
3) Berenguer J et al. Open Forum Infect Dis 2016;3:ofw059. 4) Berenguer J, et al. EACS 2017; Abstract # PS9/3
1) Gonzalez J, et al Enferm Infecc Microbiol Clin 2005; 23:340-8. 2) Gonzalez J, et al. IV Congreso Nacional de GeSIDA; 2012. Abstract # PO-41.
3) Berenguer J et al. Open Forum Infect Dis 2016;3:ofw059. 4) Berenguer J, et al. EACS 2017; Abstract # PS9/3
1.Gonzalez J, et al Enferm Infecc Microbiol Clin 2005; 23:340-8.
2.Gonzalez J, et al. IV Congreso Nacional de GeSIDA; 2012. Abstract # PO-41
3.Berenguer J et al. Open Forum Infect Dis 2016;3:ofw059
4.Berenguer J, et al. EACS 2017; Abstract # PS9/3
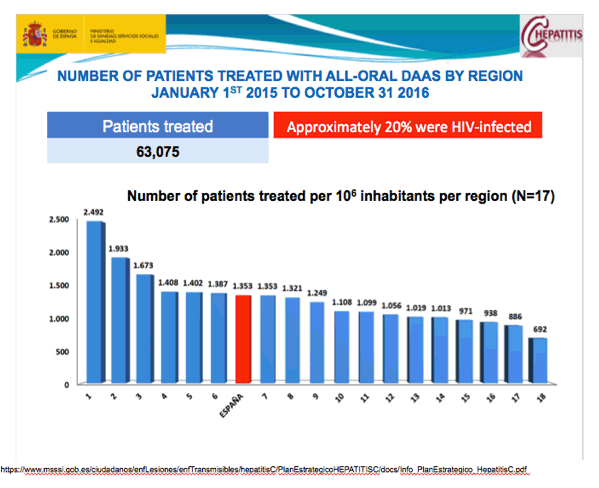
https://www.msssi.gob.es/ciudadanos/enfLesiones/enfTransmisibles/hepatitisC/PlanEstrategicoHEPATITISC/docs/Info_PlanEstrategico_HepatitisC.pdf
|
| |
|
 |
 |
|
|