 |
 |
 |
| |
Treatment as Prevention for Hepatitis C in Iceland (TraP HepC).
Real-world experience from a nationwide elimination program
using direct acting antiviral agents
|
| |
| |
Reported by Jules Levin
EASL 2017 April 19-23 Amsterdam Netherlands
Sigurdur Olafsson* 1, 2, Thorarinn Tyrfingsson3, Valgerdur Runarsdottir3,
Ottar M. Bergmann1, Einar S. Bjornsson1, 2, Birgir Johannsson4, Bryndis
Sigurdardottir4, Ragnheidur H. Fridriksdottir1, Arthur Love2, 5,
Thorvardur J. Love2, 6, Gudrun Sigmundsdottir7, Maria Heimisdottir2, 8,
Magnus Gottfredsson2, 4,6 and the TraP HepC working group
1Gastroenterology and Hepatology, Landspitali University Hospital, 2Faculty of
Medicine, School of Health Sciences, University of Iceland, 3Vogur addiction
treatment center, 4Infectious Diseases, 5Virology, 6Department of Science,
Landspitali University Hospital, 7State Epidemiologist, Directorate of Health,
8Department of Finance , Landspitali University Hospital, Reykjavik, Iceland

Program abstract
Background and Aims: Hepatitis C virus (HCV) infection is associated with significant morbidity and mortality world-wide. Iceland, with a population of 330,000 has a HCV seroprevalence of 0.3% (estimated total of 800-1000 patients). The most common genotypes are 1 (45%) and 3a (50%). At the current rate of treatment uptake the effect on prevalence and long term burden of the disease will be limited. Direct-acting antiviral agents (DAAs) have made treatment on a larger scale feasible which may reduce spread and near-eliminate HCV in communities.
Methods: A nationwide treatment effort was launched in Iceland in January 2016, where all patients infected with HCV are offered treatment with DAAs according to national guidelines with SOF/LDV+/-RBV through October 2016 and SOF/VEL+/-RBV thereafter. People with recent injection drug use (IDU), prisoners and patients with advanced liver disease are prioritized for treatment. People who inject drugs receive additional support to facilitate compliance. Various strategies are employed to enhance surveillance and strengthen harm reduction. We aim to initiate treatment for up to 20% of the total HCV-infected population every 4 months so that every patient in Iceland will be treated within a 36 month period (end-2018).The goal of the program is a reduction in domestic transmission of HCV.
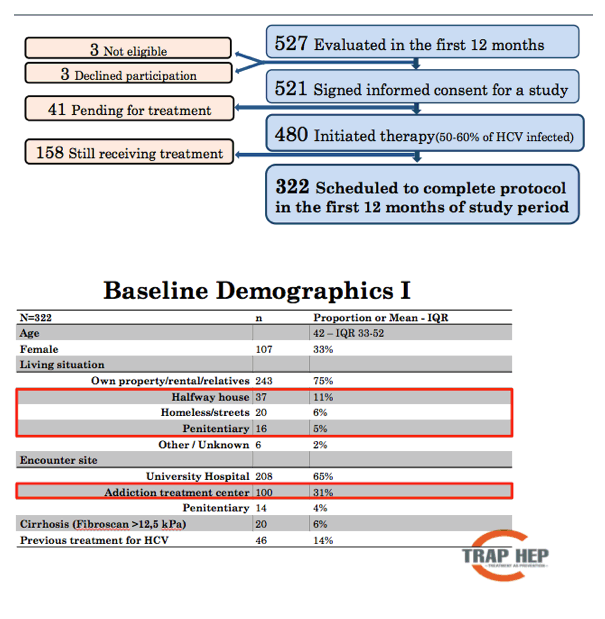
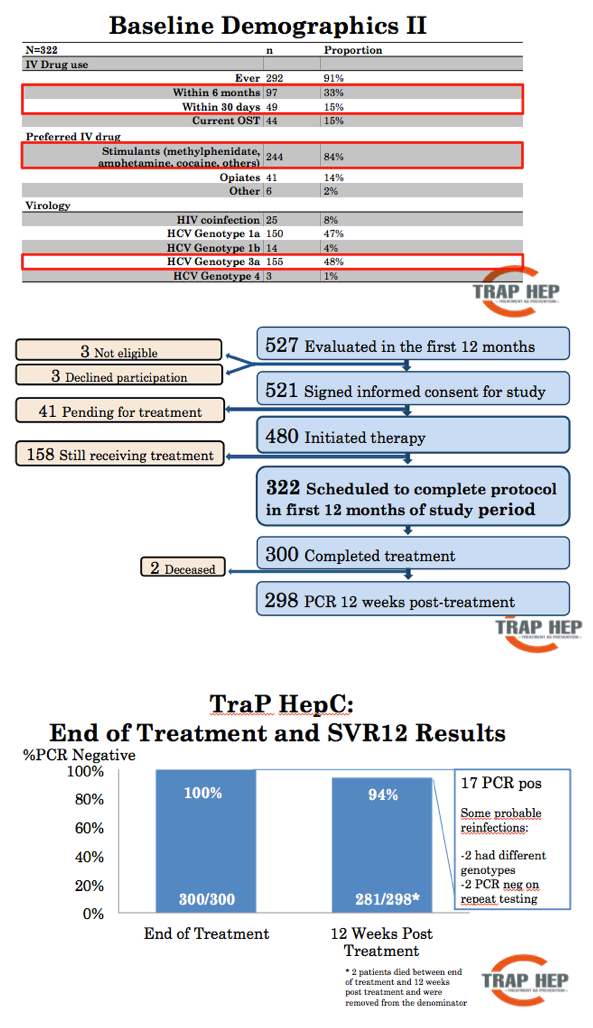
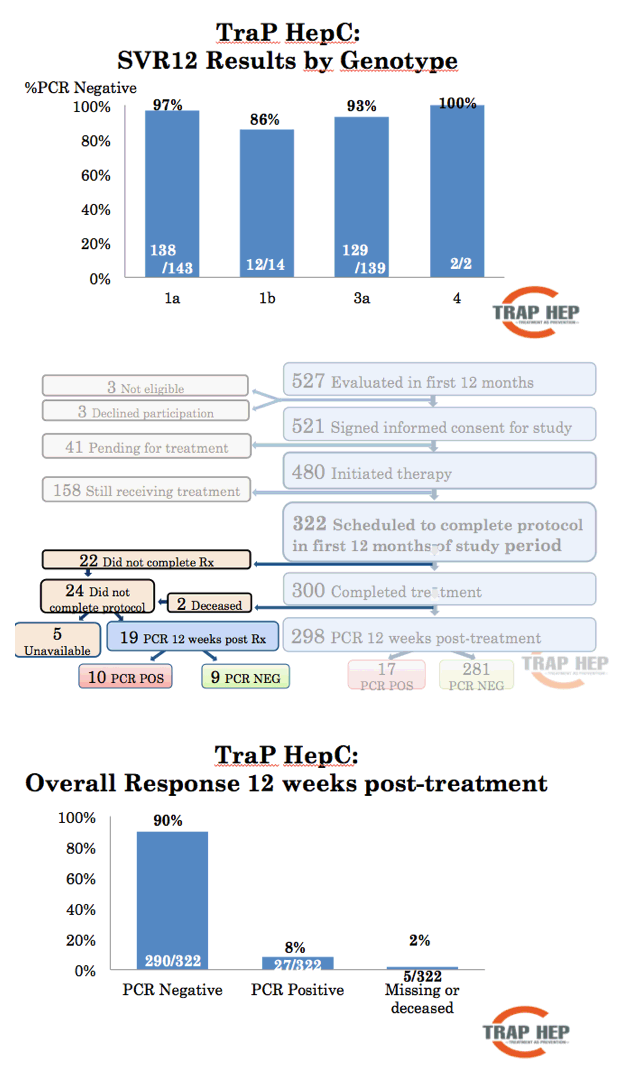
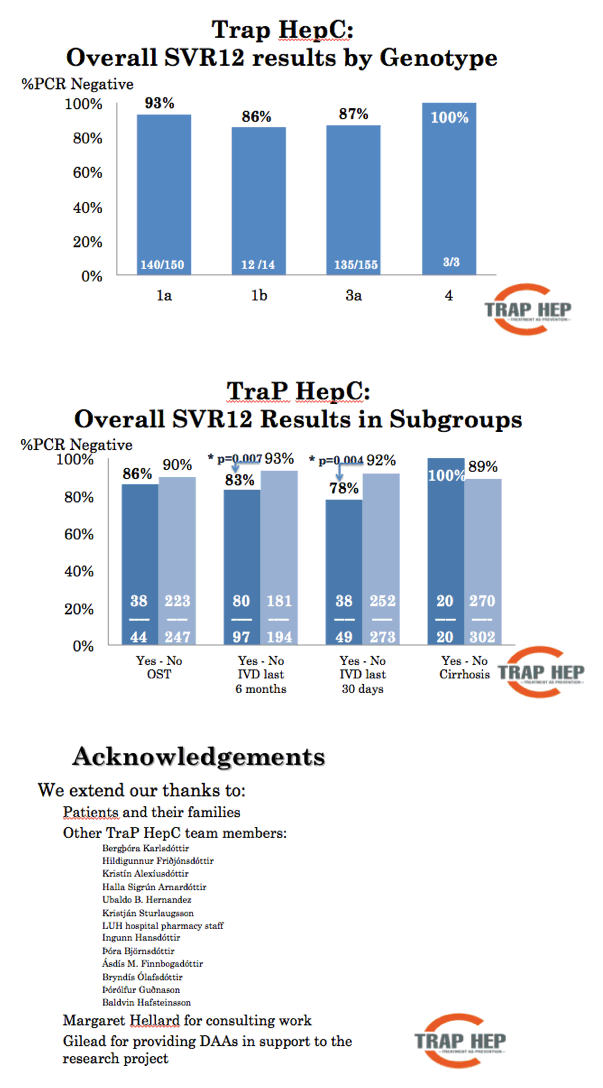
Results: On November 11th, nine months after launching the nationwide program 479 patients have been evaluated, or 48-60% of the estimated total patient population. The mean age was 41 years (range, 17-70 years), with 323 males and 161 females. The reported main route of infection was IDU (90%). At the time of evaluation, 38% of patients had a history of recent (within 6 months) IDU. Treatment with DAAs has been initiated in 406 patients (41-51% of total) and 292 (29-37%) have finished treatment. Results of HCV RNA at 12 weeks are available for 188 of whom 180 were negative, SVR12 96%. Nearly all (>90%) infected incarcerated individuals, HCV-HIV co-infected and known cirrhotic patients have been initiated on DAA treatment. So far, 22 (5%) patients have discontinued treatment and 2 (0.5%) are lost to follow-up.
Conclusions: TRAP HEP C in Iceland has been well received by patients and the community. Our experience indicates that by a well organized nationwide approach a relatively large proportion of infected patients in the community, including people actively injecting drugs, can be initiated on treatment in a short period of time.


|
| |
|
 |
 |
|
|