 |
 |
 |
| |
High prevalence of chronic hepatitis C in injecting
drug users in Tanzania, East Africa: a neglected burden of disease
|
| |
| |
Reported by Jules Levin
EASL 2017 April 19-23 Amsterdam Netherlands
Zameer Mohamed* 1, Jessie Mbwambo2, John Rwegasha3, Yusuke Shimakawa4, Sanjay Bhagani5, Stephane Chevaliez6, Julie Makani7, Simon Taylor-Robinson1, Mark Thursz1, Maud Lemoine1
1Hepatology, Imperial College London, London, United Kingdom, 2Psychiatry, Muhimbili University of Health and Allied Sciences, 3Gastroenterology, Muhimbili National Hospital, Dar-es-Salaam, Tanzania, United Republic of, 4Epidemiology, Pasteur Institute, Paris, France, 5Infectious Diseases, Royal Free Hospital, London, United Kingdom, 6Virology, Hopital Henry Mondor, Universite Paris-Est , Paris, France, 7Haematology, Muhimbili University of Health and Allied Sciences, Dar-es-Salaam, United Republic of Tanzania

Program Abstract
Background and Aims: The World Health Organisation (WHO) has recently called for hepatitis C virus (HCV) elimination and has identified injecting drug users (IDUs) as a key population. In sub-Saharan Africa, especially in East Africa, the use of injectable drugs is an emerging issue and data on chronic HCV in IDUs is missing. This study aimed to assess the prevalence and severity of chronic hepatitis C among IDUs enrolled in an opioid substitution treatment (OST) programme in Dar-es-Salaam, Tanzania.
Methods: Between May and July 2015, consecutive patients with HCV seropositive results, enrolled in the local OST centre, were invited to participate in the study. All had an epidemiological questionnaire and were offered liver assessment including fasting liver stiffness measurement (LSM) using Fibroscan, abdominal ultrasound, liver function tests, HCV RNA detection (CAP/CTM HCV version 2.0, Roche Molecular Systems, Pleasanton, CA) and genotyping (NS5B gene phylogenetic analysis) if viral load detectable.
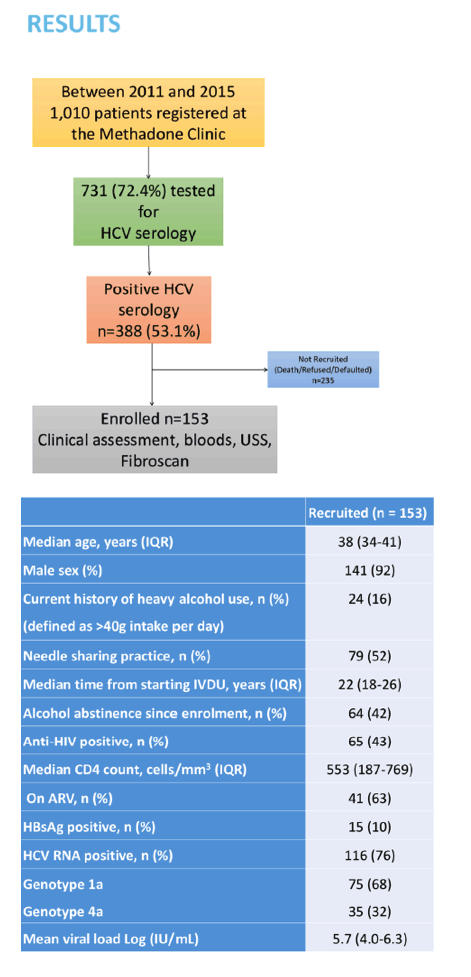
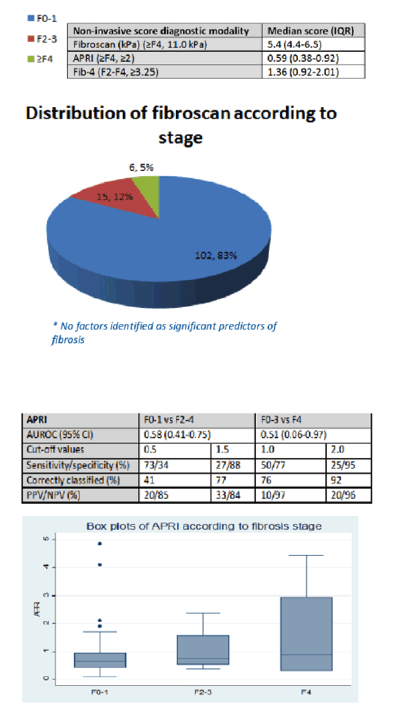
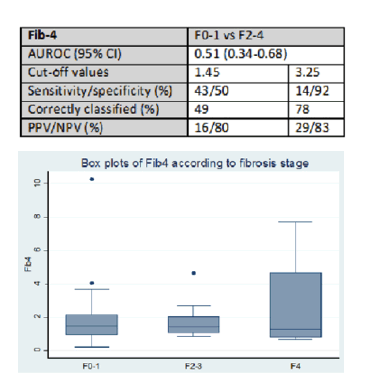
Results: When the study commenced 1,011 IDUs were registered in the local Methadone clinic. Out of them 731 (72%) were tested for HCV serology. Of these 388 (53%) had a positive result. However 235 (61%) were not recruited in the study because of death, loss of follow-up or refusal. Therefore 153 patients with a positive HCV serology were assessed: 141 (92%) were male, median age 38 (IQR 34-41). 41 patients (27%) had a significant history of excess alcohol intake (≥50g per day). The median time since the first injection of drug was 22 years (IQR 18-26). Of the 153 patients, 65 (44%) were HIV co-infected, 15 (10%) also had a positive HBs antigen, 116 (76%) had a detectable HCV RNA, median HCV RNA 5.7 (IQR 4-6.3) Log IU/mL. Only genotypes 1a (68%) and 4a (32%) were identified. The median LSM was 5.3 kPa (IQR 4.4-6.5) and 21 (17%) had clinically significant fibrosis (≥F2), 6 (5%) were considered cirrhotic (F4). None of these patients had access to HCV antiviral therapy.
Conclusions: Although the use of injectable drugs in East Africa is a poorly covered topic, there is clearly a significant burden of chronic hepatitis C, with many having HIV and HBV co-infections. Screening, access to care and treatment for HCV in IDUs in Africa should be urgently improved in order to comply with the HCV elimination goal.

|
| |
|
 |
 |
|
|