 |
 |
 |
| |
No change in HCV-specific T cell functionality
after successful DAA treatment in chronic hepatitis C patients.
|
| |
| |
Reported by Jules Levin
EASL 2017 April 19-23 Amsterdam Netherlands
Femke Stelma1,2, Meike H. van der Ree1,2, Marjan J. Sinnige2, Anthony Brown3, Leo Swadling3, Sophie B. Willemse1, Marc van der Valk1, J. Marleen L. de Vree4, Paul Klenerman3, Eleanor Barnes3, Neeltje A. Kootstra2 and Hendrik W. Reesink1,2.
Department of Gastroenterology and Hepatology (1) and Experimental Immunology (2), Academic Medical Center, Amsterdam, The Netherlands 3. Nuffield department of Medicine and the Oxford NIHR BRC, University of Oxford, Oxford, UK 4. Department of Gastroenterology and Hepatology, University Medical Center Groningen, Groningen, The Netherlands

Program abstract
Background and Aims: In patients with chronic hepatitis C (CHC) infection, plasma IP-10 levels are increased, reflecting a state of prolonged immune activation. As a result of constant immune activation, virus-specific T cells are exhausted and functionally inactive. Here we analysed the changes in IP-10 levels as well as HCV-specific T cell function in CHC patients who were successfully treated with DAA's.
Methods: In this multicenter, investigator-initiated study, 29 patients with HCV genotype 1 (n = 11), 3 (n = 17) or 4 (n = 1) infection were treated with sofosbuvir and daclatasvir ± ribavirin for 12 or 24 weeks (HCV genotype 3 with cirrhosis for 24 weeks). 18 patients had previously participated in a phase 1 study where they had received a single subcutaneous injection with anti-miR-122 (RG-101). Plasma and peripheral blood mononuclear cells were collected at various time points during and after treatment. IP-10 levels were measured by ELISA, and ex vivo,HCV-specific T cell responses were quantified across the whole HCV genome using genotype specific peptides in IFN-γ ELISpot assays.
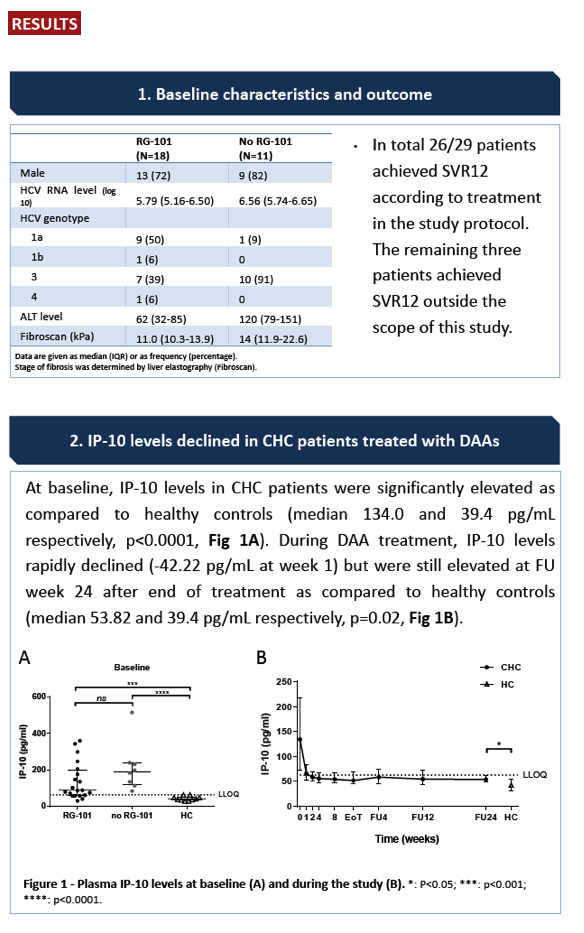
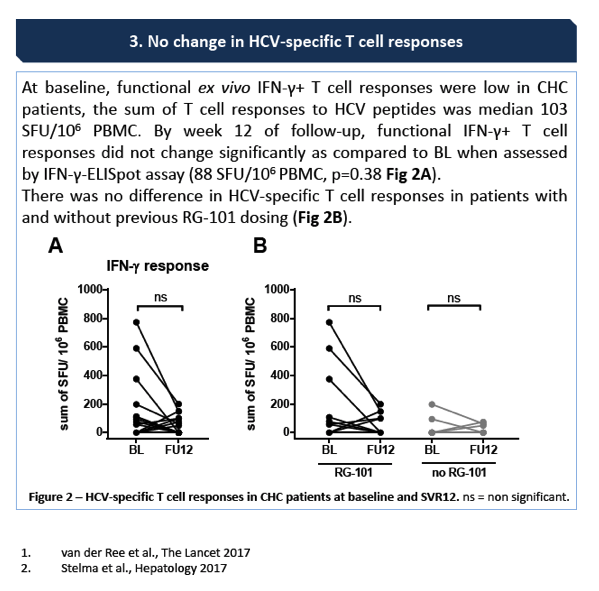
Results: All patients had an SVR12 after treatment. At baseline, IP-10 levels in CHC patients were significantly elevated as compared to healthy controls (median 134.0 and 39.4 pg/mL respectively, p <0.0001). During DAA treatment, IP-10 levels rapidly declined (-42.22 pg/mL at week 1) but were still elevated at FU week 24 after end of treatment as compared to healthy controls (median 53.82 and 39.4 pg/mL respectively, p = 0.02). At baseline, functional T cell IFN-γ responses were low in CHC patients, the sum of T cell responses was median 103 SFU/ 10^6 PBMC. By week 12 of follow-up, functional T cell IFN-γ responses did not change significantly as compared to BL when assessed by ELISpot assay (88 SFU / 10^6 PBMC, p = 0.38). The median change between BL and FU week 12 was -75 SFU / 10^6 PBMC (range -562 to +190 SFU).
Conclusions: After successful DAA treatment, broad immune activation reduces in CHC patients. The magnitude and functionality of ex vivoHCV-specific T cell responses does not increase following successful DAA treatment. Our data suggests that there is no restoration of HCV adaptive immunity after sustained virological response in CHC patients.

|
| |
|
 |
 |
|
|