 |
 |
 |
| |
The Safety and Tolerability of SOF/VEL/VOX for 8 or 12 Weeks in >1000 Patients Treated in the
POLARIS-1, POLARIS-2, POLARIS-3, and POLARIS-4 Studies: an Integrated Analysis
|
| |
| |
Reported by Jules Levin
EASL 2017 April 19-23 Amsterdam Netherlands
Michael Manns,1 Edward J. Gane,2 Bernard E. Willems,3 Stuart K. Roberts,4 Steven Flamm,5 Marc Bourliere,6 Tarik Asselah,7 Laurent Alric,8 Robert H. Hyland,9 Luisa M. Stamm,9 K.C. Huang,9 Diana M. Brainard,9
Mandana Khalili,10 Graham R. Foster,11 Stuart C. Gordon,12 K. Rajender Reddy,13 Stefan Zeuzem,14 Ira M. Jacobson,15 Curtis L. Cooper,16 Alex J. Thompson,17 Kris Kowdley,18 Eric Lawitz19
1Medizinischen Hochschule Hannover, Germany; 2Auckland Clinical Studies Ltd, Auckland, New Zealand; 3Centre Hospitalier de l'Universite de Montreal, Quebec, Canada; 4The Alfred, Melbourne, Victoria, Australia; 5Northwestern University, Chicago, Illinois, USA; 6Hopital Saint Joseph, Marseille, France; 7Hopital Beaujon, Universite Paris Diderot-Paris 7, Clichy, France; 8Hopital Purpan, Universite Toulouse III-Paul Sabatier, Toulouse, France; 9Gilead Sciences, Inc., Foster City, California, USA; 10University of California San Francisco;
11The Royal London Hospital, London, UK; 12Henry Ford Health System, Detroit, Michigan, USA; 13University of Pennsylvania, Philadelphia, USA; 14Universitätsklinikum Frankfurt, Goethe University, Frankfurt, Germany; 15Mount Sinai Beth Israel, New York, New York, USA; 16Ottawa Hospital Research Institute, Ottawa, Ontario, Canada; 17St Vincent's Hospital Melbourne, Fitzroy, Victoria, Australia; 18Swedish Medical Center, Seattle, Washington, USA; 19Texas Liver Institute, The University of Texas Health Science Center at San Antonio, USA

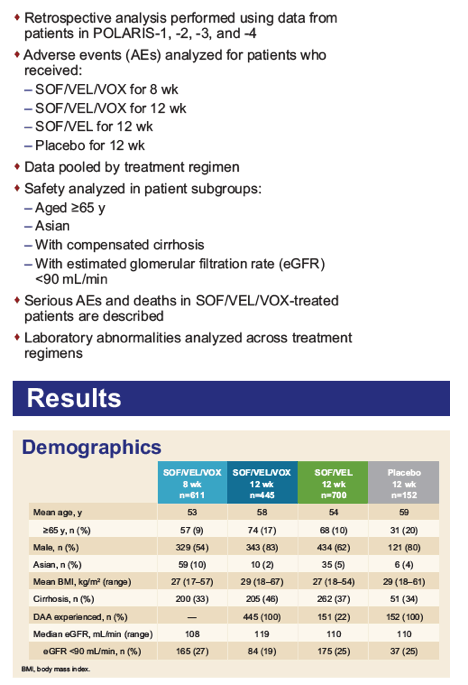
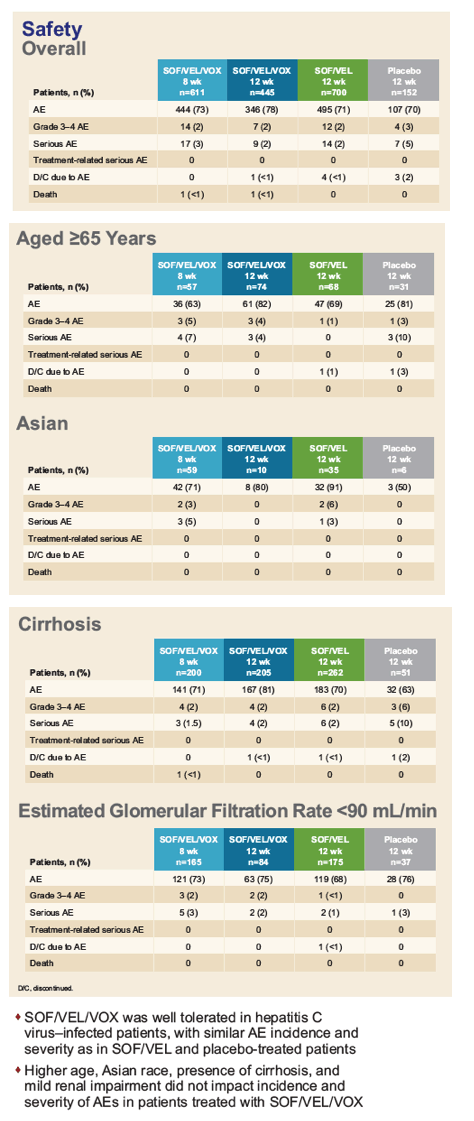
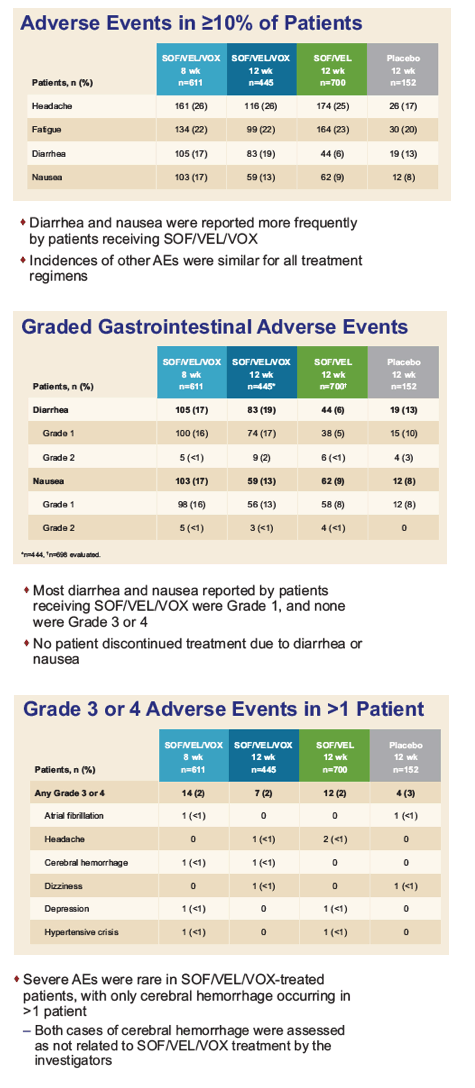
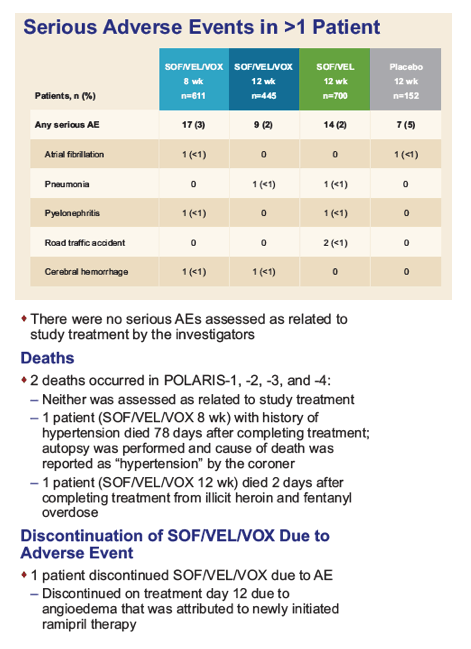
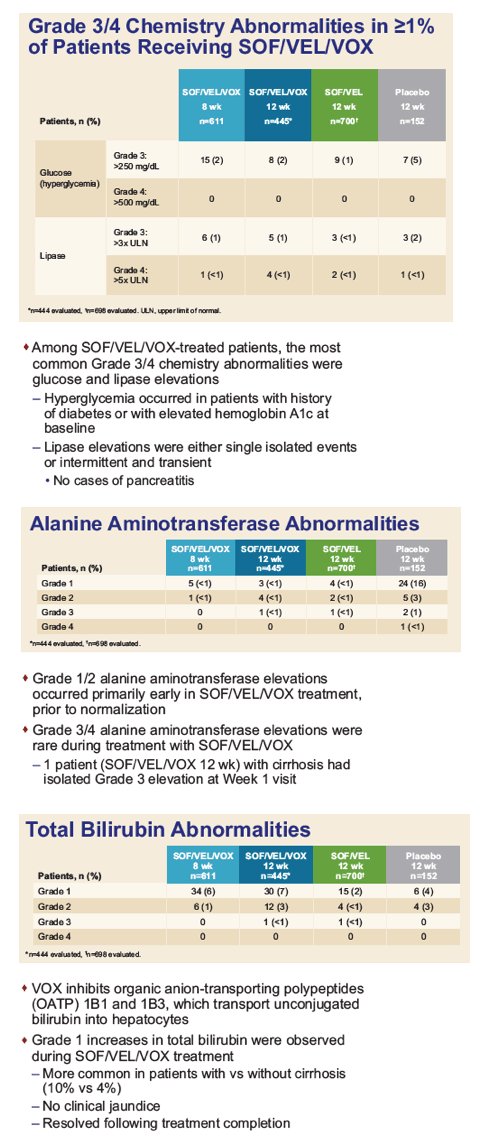

|
| |
|
 |
 |
|
|