 |
 |
 |
| |
IFN-free cure of HCV infection alters the soluble inflammatory milieu in patients with liver cirrhosis which could affect HCC surveillance by CD8+ T cells
|
| |
| |
Reported by Jules Levin
EASL 2017 April 19-23 Amsterdam Netherlands
Solomon Owusu Sekyere, Christine S. Falk, Amare Aregay, Bernhard Schlevogt, Katja Deterding, Michael Peter Manns, Arndt Vogel, Thomas Wirth, Markus Cornberg, Heiner Wedemeyer
Hannover Medical School
Germany

Program Abstract
Interferon-free cure of hepatitis C virus infection does alter the soluble inflammatory milieu which could affect hepatocellular carcinoma surveillance by CD8+ T cells in patients with liver cirrhosis
Solomon Owusu Sekyere* 1, Christine Falk2, 3, Amare Aregay1, Bernhard Schlevogt1, Katja Deterding1, Michael P. Manns1, 3, Arndt Vogel1, Thomas Wirth1, Markus Cornberg1, 3, Heiner Wedemeyer1, 3
1Department of Gastroenterology, Hepatology and Endocrinology, 2Institute of Transplantation Immunology (IFB-Tx), Hannover Medical School, Hannover, 3TTU-IICH, German Center for Infectious Diseases (DZIF), Hannover-Braunschweig, Germany
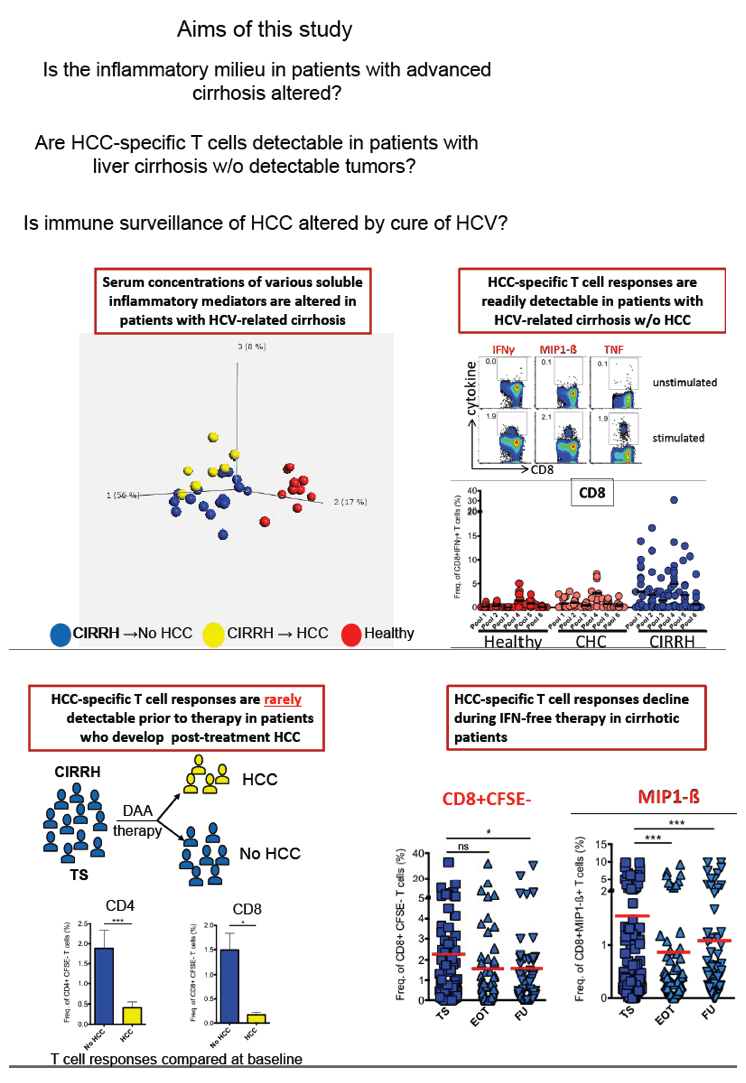
Background and Aims: Hepatitis C virus (HCV) clearance by current interferon (IFN)-free direct-acting antiviral (DAA) therapies tremendously improves clinical outcomes but falls short in eliminating the risk for developing hepatocellular carcinoma (HCC). Concerns about an even higher risk of HCC recurrence rates following IFN-free therapy have been raised and currently being debated. We recently showed that various soluble inflammatory mediators (SIM) are altered in patients with chronic hepatitis C but DAA-induced HCV clearance does not restore them. In this current study, we hypothesized that the changes in SIMs affect tumour-specific immune responses which could influence progression of HCC in patients with HCV-related cirrhosis.
Methods: We performed multi-analyte profiling of 50 SIMs in the plasma of two patient cohorts including cirrhotic patients without any HCC before or after HCV treatment (n=21) and those free of HCC at baseline but developed HCC during/after therapy (n=13). Samples were analysed at baseline, end of therapy and follow-up using the BioPlex bead array method. We further assessed T cell responses in vitro to a panel of Glypican-3-derived overlapping peptides at the defined time points upon short-term culture of PBMCs. Additionally, we analysed HCC-specific CD8+ T cell responses to HLA-A2 restricted epitopes of AFP, NY-ESO-1, MAGE-A3, MAGE-A10, SSX-2 and p53 as well as HCV-specific CD8+ T cell epitopes of NS31073, NS31406, Core132 and NS5B2594 in vitro.
Results: Plasma profiling at baseline showed differential SIM concentrations between patients with liver cirrhosis who developed HCC and those that did not. Of note, HCC-specific T cell responses were detectable in all patients suggesting an active tumour surveillance in liver cirrhosis. Treatment-induced HCV clearance significantly decreased the strength of HCC-specific CD8+ but not CD4+ T cell responses in cirrhotic patients. In contrast, HCV-specific CD8+ T cell responses failed to recover upon DAA therapy. Finally, our data showed that Glypican-3-specific T cell proliferative and functional responses in cirrhotic patients who developed HCC upon IFN-free therapy were weak at therapy start but experienced significant enhancement during follow-up.
Conclusions: We here provide experimental evidence that IFN-free cure of HCV infection does alter the soluble inflammatory milieu in patients with liver cirrhosis which could affect HCC surveillance by CD8+ T cells. A close HCC screening is recommended in cirrhotic HCV patients despite HCV clearance.

|
| |
|
 |
 |
|
|