 |
 |
 |
| |
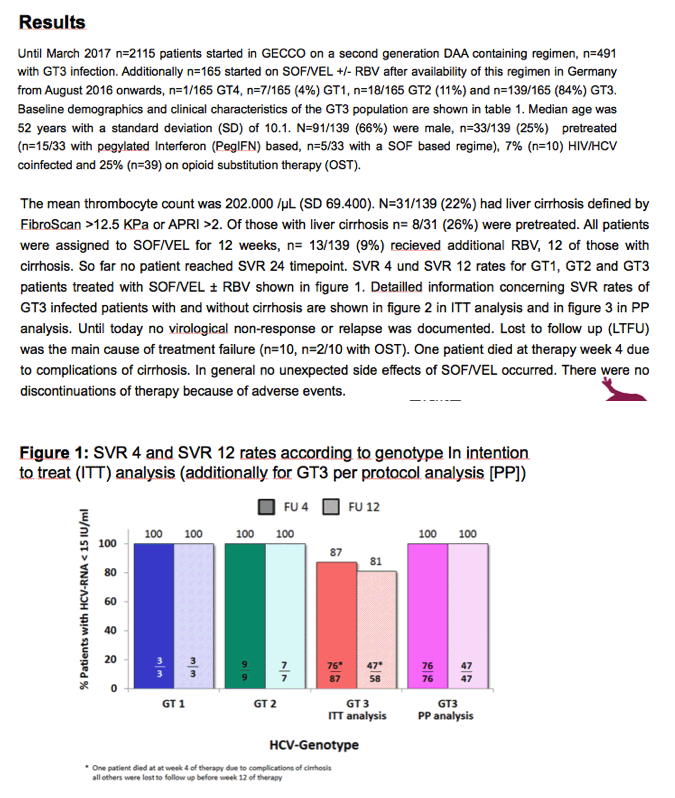
Does Sofosbuvir (SOF)/Velpatasvir (VEL) with or without Ribavirin (RBV) change the landscape of therapy in chronic hepatitis C genotype 3 (GT 3) infection? - Results from the GErman hepatitis C COhort (GECCO)
|
| |
| |
Reported by Jules Levin
EASL 2017 April 19-23 Amsterdam Netherlands
Stefan Christensen1, Patrick Ingiliz2, Knud Schewe3, Juergen Rockstroh4, Dietrich Hueppe5, Axel Baumgarten2, Thomas Lutz6, Guenther Schmutz7, Karl Georg Simon8, Florian Berger7, Heiner Busch1, Julian Schulze zur Wiesch9, Stefan Mauss7
1 CIM Infectious diseases, Muenster, 2 Center for Infectiology Berlin (CIB), Berlin, 3Infektionsmedizinisches Centrum Hamburg (ICH), Hamburg, 4 Medical Department 1, University Hospital Bonn, Bonn, 5Practice for Gastroenterology Herne, Herne, 6Infektiologikum, Frankfurt, 7Center for HIV and Hepatogastroenterology, Duesseldorf, 8 Practice for Gastroenterology Leverkusen, Leverkusen, Germany, 9 Ambulanzzentrum Virushepatologie Universitätsklinikum Hamburg-Eppendorf, Hamburg, Germany.
EASL:No Impact of RASs on the High Efficacy of SOF/VEL/VOX for 12 Weeks in DAA-Experienced Patients: an Integrated Resistance Analysis of the POLARIS-1 and POLARIS-4 Studies - (04/26/17)
AASLD:A Randomized, Phase 3 Trial of Sofosbuvir/Velpatasvir/Voxilaprevir for 8 Weeks and Sofosbuvir/Velpatasvir for 12 Weeks for Patients with Genotype 3 HCV Infection and Cirrhosis: The POLARIS-3 Study - (11/15/16)
AASLD:A Randomized Phase 3 Trial of Sofosbuvir/Velpatasvir/Voxilaprevir for 8 Weeks Compared to Sofosbuvir/Velpatasvir for 12 Weeks in DAA-Naïve Genotype 1-6 HCV Infected Patients: The POLARIS-2 Study - (11/15/16)
AASLD:Sofosbuvir/Velpatasvir/Voxilaprevir for 12 Weeks as a Salvage Regimen in NS5A Inhibitor-Experienced Patients With Genotype 1-6 Infection: The Phase 3 POLARIS-1 Study - (11/15/16)
AASLD:Sofosbuvir/Velpatasvir Fixed-Dose Combination for 12 Weeks Compared to Sofosbuvir with Ribavirin for 24 Weeks in Genotype 3 HCV-Infected Patients: The Randomized Controlled Phase 3 ASTRAL-3 Study - (11/30/15)
AASLD:Sofosbuvir and Velpatasvir for HCV Genotype 2 and 3 Infection - (11/30/15) published


|
| |
|
 |
 |
|
|