 |
 |
 |
| |
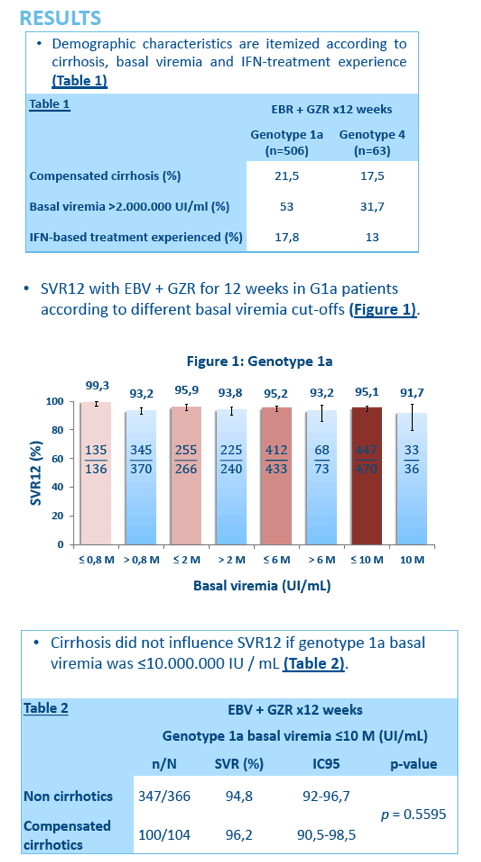
HIGH SUSTAINED VIROLOGICAL RESPONSE RATES IN GENOTYPES 1a AND 4 HEPATITIS C PATIENTS TREATED WITH ELBASVIR AND GRAZOPREVIR AND BASAL VIREMIA >800.000 UI/ML
|
| |
| |
Reported by Jules Levin
EASL 2017 April 19-23 Amsterdam Netherlands
A. OLVEIRA 1, M. MONTES 6, A. LOPEZ NAVAS 5, J. GARCIA-SAMANIEGO 2, L. MARTIN CARBONERO 6, J. RAMON LARRUBIA 3, P. CASTILLO 1, A. MADEJON 4, M. ROMERO 1, A. GARCIA 1, J. C. ERDOZAIN 1, J. GONZALEZ GARCIA 6.
1 Aparato Digestivo, Hospital La Paz, Madrid, Spain; 2 Aparato Digestivo, Hospital La Paz, Madrid, CIBE Rehd, Spain; 3 Aparato Digestivo, Hospital de Guadalajara, Spain; 4 Hospital La Paz, Madrid, CIBERehd, Spain; 5 Departamento Medicamentos Uso Humano, AEMPS; 6 Medicina Interna, Hospital La Paz, Madrid, Spain.
EASL: Safety and Efficacy of the Fixed-dose Combination Regimen of Uprifosbuvir (MK-3682) / Grazoprevir / Ruzasvir in Cirrhotic or Non-cirrhotic Patients with Chronic HCV GT1 Infection Who Previously Failed a Direct-acting Antiviral Regimen (C-SURGE) - (04/24/17)
EASL: Elbasvir/Grazoprevir Plus Sofosbuvir ± Ribavirin in Treatment-Naive And Treatment-Experienced People With Hepatitis C Virus Genotype 3 Infection and Compensated Cirrhosis: SVR24 Results of the C-ISLE Study - (04/21/17)
EASL: Real-World Observational Study in the U.S. Veterans Affairs System Evaluating Use of Merck's ZEPATIER® (Elbasvir and Grazoprevir) Shows High Sustained Virologic Response Rates in Patients with Chronic Hepatitis C - (04/24/17)
EASL: Elbasvir/Grazoprevir effectiveness in patients with Chronic Hepatitis C and Chronic Kidney Disease: Real-world experience from the TRIO Network - (04/26/17)
EASL: Real-world use of elbasvir/grazoprevir and outcomes in patients with Chronic Hepatitis C: Retrospective data analyses from the TRIO Network. - (04/26/17)


REFERENCES
1.Zepatier: EPAR-Public assessment report EMA/419807/2016.
2.Sulkowski M, Hezode C, Gerstoft J, Vierling JM, Mallolas J, Pol S, et al. Efficacy and safety of 8 weeks versus 12 weeks of treatment with grazoprevir (MK-5172) and elbasvir (MK-8742) with or without ribavirin in patients with hepatitis C virus genotype 1 mono-infection and HIV/hepatitis C virus co-infection (C WORTHY): a randomized, open-label phase 2 trial. Lancet. 2015 Mar 21; 385(9973):1087-97.
3.Lawitz E, Gane E, Pearlman B, Tam E, Ghesquiere W, Guyader D, et al. Efficacy and safety of 12 weeks versus 18 weeks of treatment with grazoprevir (MK-5172) and elbasvir (MK-8742) with or without ribavirin for hepatitis C virus genotype 1 infection in previously untreated patients with cirrhosis and patients with previous null response with or without cirrhosis (C-WORTHY): a randomized, open-label phase 2 trial. Lancet. 2015 Mar 21;385(9973):1075-86.
4. Roth D, Nelson DR, Bruchfeld A, Liapakis A, Silva M, Monsour H Jr, et al. Grazoprevir plus elbasvir in treatment-naive andtreatment-experienced patients with hepatitis C virus genotype1 infection and stage 4-5 chronic kidney disease (the C-SURFER study): a combination phase 3 study. Lancet. 2015 Oct 17;386(10003):1537-45.
5. Zeuzem S, Ghalib R, Reddy KR, Pockros PJ, Ben Ari Z, Zhao Y, et al. Grazoprevir-Elbasvir Combination Therapy for Treatment-Naive Cirrhotic and Noncirrhotic Patients With Chronic Hepatitis C Virus Genotype 1, 4, or 6 Infection: A Randomized Trial. Ann Intern Med.2 015 Jul 7;163(1):1-13.
6. Rockstroh JK, Nelson M, Katlama C, Lalezari J, Mallolas J, Bloch M,et al. Efficacy and safety of grazoprevir (MK-5172)and elbasvir (MK-8742) in patients with hepatitis C virus and HIV co-infection (C-EDGE CO-INFECTION): a non-randomised, open-label trial. Lancet HIV. 2015 Aug;2(8):e319-27.
7.Kwo P, Gane EJ, Peng CY, Pearlman B, Vierling JM, Serfaty L, et al. Effectiveness of Elbasvir and Grazoprevir Combination, With or Without Ribavirin, for Treatment-Experienced Patients With Chronic Hepatitis C Infection. Gastroenterology. 2017 Jan;152(1):164-175.e4.
|
| |
|
 |
 |
|
|