| |
The HCV care continuum does not end with cure:
A call to arms for the prevention of reinfection - Editorial
|
| |
| |
Download the PDF here
Download the PDF here
...... HCV reinfection incidence and spontaneous clearance rates in HIV-positive men who have sex with men in Western Europe - 2 reports
from Jules: other study reported reinfection occurred 3 times. from Jules: a major barrier to access for treatment by medicaid & commercial insurers is their concern & cost for reinfection.
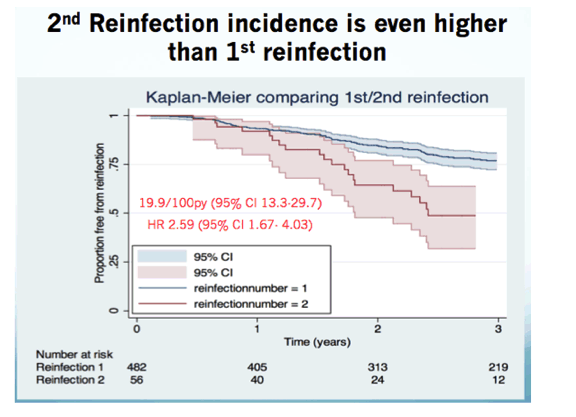
"In total, 143 HCV reinfections occurred at a rate of 7.3/100 py (95% confidence interval (CI) 6.2-8.6). The median duration to reinfection was 2.0 years (IQR 1.1-3.3 years)......This is the largest cohort of HIV infected MSM patients, who were followed-up longitudinally for HCV reinfections after initial HCV cure. We found a high reinfection incidence of 7.3/100 py, with an estimate that almost one third of patients were reinfected after 5 years. .....This indicates a maintained risk behavior in a potentially specific high risk group, who require urgent targeting for prevention measures related to risk behaviors. How often these individuals require testing for HCV infection de novo is unclear but the present suggestion that this occurs annually does not appear sufficient, and we suggest HCV RNA testing every three to six months after an incident HCV infection and every three months in patients that had been reinfected.....Studies in PWID, which are all much smaller than the number of included individuals in this study, have shown HCV reinfection incidence rates between 0.8 and 4.7 per 100 py [30]. In a meta-analysis performed by Aspinall et al. the pooled reinfection incidence in those reporting intravenous drug use after HCV cure was 6.44 (95% CI, 2.49-16.69) per 100 py [31]. Hill et al. reported a five-year risk for HCV reinfection of 10.6% in PWID while it was over 15% in MSM [21]. These findings underline the need for risk-adapted interventions during follow-up post HCV cure. HIV-positive MSM with a history of HCV infection and especially those presenting with an HCV reinfection require close monitoring and behavioral interventions to reduce the risk of reinfection.....Sixty-four men either spontaneously cleared or were successfully treated for their subsequent HCV reinfection and had follow-up data available representing 143 py of follow-up (median follow-up 1.8 years, IQR 0.9-2.8). Of these 64 men, 27 presented with a second reinfection at a median of 1.7 years (IQR 1.2-2.4) after cure of the prior infection. The second reinfection incidence rate was significantly higher than the first reinfection incidence at 18.8/100 py (95% CI 12.9-27.5; HR for second reinfection 2.51, 95% CI 1.7-3.8, p <0.001)."
Reported originally at EASL 2016: Hepatitis C virus reinfection incidence and outcomes among HIV-positive MSM in Western Europe - (04/15/16)
AASLD: ELEVATED HCV REINFECTION INCIDENCE AFTER SUCCESSFUL TREATMENT AMONG HIV-INFECTED MEN WHO HAVE SEX WITH MEN IN SAN DIEGO- (11/21/16)
AASLD: Impact of Drug Use and Opioid Substitution Therapy on Hepatitis C Reinfection: The BC Hepatitis Testers Cohort - (11/21/16)
--------------------------
Editorial - The HCV care continuum does not end with cure: A call to arms for the prevention of reinfection
from Jules: a major barrier to access for treatment by medicaid & commercial insurers is their concern & cost for reinfection.
Jnl of Hepatology Feb 2017
Of these 64 men, 27 presented with a second reinfection at a median of 1.7 years (IQR 1.2-2.4) after cure of the prior infection. The second reinfection incidence rate was significantly higher than the first reinfection incidence at 18.8/100 py (95% CI 12.9-27.5; HR for second reinfection 2.51, 95% CI 1.7-3.8, p <0.001).
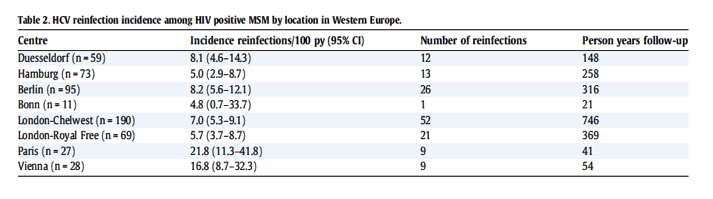
Table 2 depicts the reinfection incidence per center, with the highest being in Paris (21.8/100py, 95% CI 11.3-41.8), followed by Vienna (16.8/100 py, 95% CI 8.7-32.3), Berlin (8.2/100 py, 95% CI 5.6-12.1), Duesseldorf (8.1/100 py, 95% CI 4.6-14.3), and London/Chelsea Westminster (7.0/100 py, 95% CI 5.3-9.1). The lowest in incidence rate was seen in Hamburg (5.0/100 py, 95% CI 2.9-8.7). The incidence rate decreased only slightly over time (Fig. 2).
Oluwaseun Falade-Nwulia, Mark Sulkowski
Johns Hopkins University School of Medicine, Baltimore, MD, United States
"HCV treatment needs to be combined with harm reduction strategies for PWID, at-risk MSM and other high risk groups to reduce the risk of reinfection....Are there subgroups of HIV-infected MSM that are at highest risk of HCV infection and reinfection? The work carried out by Ingiliz et al. and others would suggest so.....To date, relatively high rates of reinfection with hepatitis C following cure have been observed in two patient groups: People who inject drugs (PWID) and HIV-infected men who have sex with men (MSM)....In this issue of the Journal of Hepatology, Ingiliz et al. present additional data highlighting the high rates of HCV reinfection among HIV-infected MSM from eight centers in Europe [9]. These men had either previously cleared HCV spontaneously or after successful treatment. They found a reinfection prevalence of 7.3.....However, what was most striking was the increase in reinfection incidence rates with each subsequent reinfection (HCV reinfection incidence 7.3/100 PY for the first reinfection and 18.8/100PY for the second reinfection). As described by the authors, this is suggestive of ongoing high risk behaviors in certain groups.....These high-risk behaviors such as unprotected, traumatic sex with concomitant illicit drug use and the more recent proliferation of chemsex (the use of drugs to increase sexual disinhibition and arousal) likely drive incident HCV infection and reinfection.... Without behavior change, gains in population level HCV control from HCV treatment may be significantly compromised....reinfections need to be prevented and when they occur, quickly treated to prevent onward transmission. And although it has eluded us thus far, work toward the development of an effective vaccine to prevent HCV infection must be intensified if the ambitious WHO HCV elimination targets are to be achieved by 2030"
After the approval of safe and highly effective oral direct acting agents (DAA) for the treatment of hepatitis C virus (HCV) in 2014, commentaries and discussions about the elimination of hepatitis C on a population level have rapidly proliferated in scientific literature and popular press. Indeed, the World Health Organization (WHO) has announced proposed global targets of an 80% reduction in new cases of HCV infection from the 2010 level and several countries, including Australia, Egypt, Georgia and Iceland have embarked on ambitious plans to control HCV infection in their populations [1].
The launch of such programs has led to increased focus on the hepatitis C care continuum with particular emphasis on the identification of HCV-infected persons and linkage of such persons to HCV care and treatment to achieve HCV cure. Modeling data from multiple groups provide the basis for optimism and suggest that the crucial step in reducing the prevalence of HCV infection over a period of time is an increase in HCV treatment uptake in key populations [[2], [3]]. In particular, persons living with HIV infection represent a population for whom HCV control has been prioritized due to the high prevalence of HCV infection and high burden of HCV-related morbidity and mortality. Further, ongoing global efforts to improve the HIV care continuum, linking HIV-infected persons to long-term antiretroviral therapy provide an ideal foundation on which to launch concerted efforts to eliminate HCV coinfection. As these programs take shape, it is important to recognize that the HIV and HCV care continuum are not identical. In the absence of HIV cure, patients must continue antiretroviral therapy indefinitely, whereas HCV treatment is finite and curative. However, in the absence of an effective HCV vaccine, persons cured of chronic HCV are at-risk for reinfection if exposed. Thus, the HCV care continuum does not end with cure; and the prevention of reinfection must be addressed in persons at-risk (Fig. 1). To date, relatively high rates of reinfection with hepatitis C following cure have been observed in two patient groups: People who inject drugs (PWID) and HIV-infected men who have sex with men (MSM) [[4], [5], [6], [7]].
The global incidence of HCV among HIV-infected MSM is estimated to be 0.53/100 person-years (PYs) [8]. Estimates of the incidence of reinfection are much higher at 9.6-15.2/100 PYs suggesting that there is a finite pool of HIV-infected MSM who require focused concerted effort for HCV control [[4], [5], [6]]. In this issue of the Journal of Hepatology, Ingiliz et al. present additional data highlighting the high rates of HCV reinfection among HIV-infected MSM from eight centers in Europe [9]. These men had either previously cleared HCV spontaneously or after successful treatment. They found a reinfection prevalence of 7.3 (95% CI 6.2-8.6)/100 PYs with a trend for a lower incidence of reinfection among men who had spontaneously cleared their incident infection than among individuals who were treated (Hazard ratio 0.62, 95% CI 0.38-1.02, p = 0.06). The strengths of this study include the large number of individuals studied (606 MSM) and the analysis of HCV reinfection among these individuals from eight centers in multiple countries (Austria, France, Germany and the UK). The authors were thus able to describe regional differences in HCV reinfection risk. However, what was most striking was the increase in reinfection incidence rates with each subsequent reinfection (HCV reinfection incidence 7.3/100 PY for the first reinfection and 18.8/100PY for the second reinfection). As described by the authors, this is suggestive of ongoing high risk behaviors in certain groups.
These high-risk behaviors such as unprotected, traumatic sex with concomitant illicit drug use and the more recent proliferation of chemsex (the use of drugs to increase sexual disinhibition and arousal) likely drive incident HCV infection and reinfection. Without behavior change, gains in population level HCV control from HCV treatment may be significantly compromised [10]. HCV treatment interventions will need to be combined with harm reduction and behavioral interventions to reduce rates of HCV reinfection to preserve reductions achieved in prevalent HCV infections at the population level. While every patient treated for HCV should receive counseling regarding the risk of and strategies to prevent HCV reinfection, it will be critical to identify factors associated with particularly high risk of reinfection in specific groups. This knowledge should guide design of effective combined behavioral and biomedical harm reduction and treatment interventions that specifically target these groups. For at-risk HIV-infected MSM, these interventions should focus on addressing the risks of disease transmission surrounding chemsex, unprotected anal intercourse and reinforce the need for condom use and avoidance of sex while using drugs. Similarly, at-risk PWID will require counseling on safer injection practices including the risk of HCV transmission with sharing of any drug use paraphernalia including cookers, cotton or water, access to clean needles and syringes, needle/syringe exchange programs and treatment for addiction including opioid agonist therapy.
Are there subgroups of HIV-infected MSM that are at highest risk of HCV infection and reinfection? The work carried out by Ingiliz et al. and others would suggest so [[9], [11], [12]]. A better understanding of predictors of HCV reinfection in high risk populations is urgently needed and will require improved surveillance for HCV reinfection in clinical practice and funding of research studies aimed at understanding the modes and mechanisms of HCV transmissions among these populations. In clinical practice, individuals with ongoing risk after HCV cure should receive at least annual HCV RNA testing with more frequent testing in some cases to assess for reinfection. Acute HCV infection also needs to be quickly diagnosed and treated as individuals at the highest risk of infection and reinfection are also those most likely to transmit HCV to others. Acute hepatitis C infection is most often asymptomatic; thus, clinicians need to be vigilant. For persons without prior HCV infection, hepatitis C antibody testing at regular intervals may be sufficient for HCV screening in an individual with ongoing risk factors for HCV infection. However, HCV re-screening may also be indicated in the context of self-reported high risk exposures and/or newly elevated alanine aminotransferase (ALT) levels. The sooner acute HCV infection is diagnosed, the sooner it can be addressed to reduce the risk of onward transmission to others.
As we continue to scale-up HCV treatment, careful thought and research must focus on the development of innovative treatment models for populations at-risk for repeated infections. One such model is that utilizing the concept of treating individuals such as PWID and their injecting networks or HIV-infected MSM and their sexual networks at the same time, popularly referred to as the "treat your friends" strategy [13]. This is analogous to the contact tracing methods that have been used for many decades by public health officials to control sexually transmitted diseases such as syphilis. Among PWID populations, modeling data supports the efficiency of this approach to reduce the prevalence of HCV among PWID in 15 short years [2]. Achieving this reduction in HCV prevalence through HCV treatment will however require combined effective harm reduction interventions and prompt treatment of any new infections introduced into the network to prevent onward transmission. HIV-infected MSM who are in routine HIV care with regular contact with the health care system would appear to be a population for which this treatment model might be effective.
The availability of all oral DAAs has opened an inspiring door of opportunity for HCV control at the population level. While oral DAAs are necessary to achieve these goals, treatment alone is not going to be sufficient. Every step in the HCV care continuum must be optimized to ensure that HCV-infected individuals are aware of their infection and linked to HCV treatment. Additionally, HCV treatment needs to be combined with harm reduction strategies for PWID, at-risk MSM and other high risk groups to reduce the risk of reinfection. After HCV cure, particular attention needs to be paid to surveillance for reinfections. These reinfections need to be prevented and when they occur, quickly treated to prevent onward transmission. And although it has eluded us thus far, work toward the development of an effective vaccine to prevent HCV infection must be intensified if the ambitious WHO HCV elimination targets are to be achieved by 2030 [14].
---------------------
HCV reinfection incidence and spontaneous clearance rates in HIV-positive men who have sex with men in Western Europe
Background & Aims
Moderate cure rates of acute hepatitis C virus (HCV) infections with pegylated interferon and ribavirin have been described in the last decade in men who have sex with men (MSM), who are also coinfected with the human immunodeficiency virus (HIV). However, a subsequent high incidence of HCV reinfections has been reported regionally in men who both clear the infection spontaneously or who respond to treatment.
Methods
Retrospective analysis of reinfections in HIV infected MSM in eight centers from Austria, France, Germany, and the UK within the NEAT network between May 2002 and June 2014.
Results
Of 606 individuals who cleared HCV spontaneously or were successfully treated, 149 (24.6%) presented with a subsequent HCV reinfection. Thirty out of 70 (43%) who cleared again or were successfully treated, presented with a second reinfection, 5 with a third, and one with a fourth reinfection. The reinfection incidence was 7.3/100 person-years (95% CI 6.2-8.6). We found a trend for lower incidence among individuals who had spontaneously cleared their incident infection than among individuals who were treated (Hazard ratio 0.62, 95% CI 0.38-1.02, p = 0.06). Spontaneous clearance of reinfection was associated with ALT levels >1000 IU/ml and spontaneous clearance of a prior infection.
Conclusions
HCV reinfection is an issue of major concern in HIV-positive MSM. Prevention strategies are needed for high risk groups to reduce morbidity and treatment costs. HIV-positive MSM with a prior HCV infection should be tested every 3 to 6 months for reinfection. Those who had achieved a reinfection should be tested every 3 months.
Lay summary
We evaluated the occurrence of HCV reinfection in HIV-positive men who have sex with men. We found an alarming incidence of 7.3/100 person-years. Prevention measures need to address this specific subgroup of patients at high risk for HCV.
Discussion
This is the largest cohort of HIV infected MSM patients, who were followed-up longitudinally for HCV reinfections after initial HCV cure. We found a high reinfection incidence of 7.3/100 py, with an estimate that almost one third of patients were reinfected after 5 years. These numbers highlight the failure of current prevention strategies and the need for specific measures in the HIV infected MSM population in Europe. With a high treatment uptake in this population even in the interferon era and with higher response rates to treatment in the acute phase of infection [27], reinfections are most likely occurring due to maintained risk behaviors. As new, well tolerated, but costly HCV treatments have become the standard of care for HCV therapy, there is an urgent need to develop strategies to prevent reinfection at such a scale. It is essential to expand testing opportunities to identify at the earliest opportunity men in the early stages of infection to prevent onward transmission of infection through treatment and behavioral interventions. The men included in this study were all linked to care centers, where if retained in care they are closely monitored and frequently tested for HCV reinfection using HCV RNA. Rapid access to effective treatment in conjunction with interventions to reduce high risk behaviors are then required.
The mode of transmission is not entirely understood in this population, but seems to occur in the setting of HIV infection, potential traumatic sexual practices with increased risk of blood-blood contact, and increasing recreational drug use including intravenous administration, commonly referred to as "Chemsex" [28]. "Chemsex" is defined as the use of sexually disinhibiting recreational drugs to facilitate sexual sessions lasting often several days with multiple sexual partners and which put men at high risk of infection with HCV and other sexually transmitted infections [29]. Our patients had a median age of 41 years at their first reinfection and well-controlled HIV infection, indicating that they were aware and compliant to health interventions, but potentially also driving their willingness for unprotected and/or chemically enhanced sex. The dramatic incidence rate observed here confirms on a European level what has previously been reported [[19], [20], [22]] on a regional level.
Studies in PWID, which are all much smaller than the number of included individuals in this study, have shown HCV reinfection incidence rates between 0.8 and 4.7 per 100 py [30]. In a meta-analysis performed by Aspinall et al. the pooled reinfection incidence in those reporting intravenous drug use after HCV cure was 6.44 (95% CI, 2.49-16.69) per 100 py [31]. Hill et al. reported a five-year risk for HCV reinfection of 10.6% in PWID while it was over 15% in MSM [21]. These findings underline the need for risk-adapted interventions during follow-up post HCV cure. HIV-positive MSM with a history of HCV infection and especially those presenting with an HCV reinfection require close monitoring and behavioral interventions to reduce the risk of reinfection.
We observed large regional differences in reinfection incidence within our dataset, with the highest being in Paris (21.8/100 py) and the lowest being in Hamburg (5.04/100 py). These differences may reflect the lack of precision in the incidence estimates from the smaller centers due to lower numbers of men included in the dataset and the shortest follow-up time, or it may represent specific risk behavior patterns in men seen in the different centers, rather than absolute differences in incidences in the regions.
The reinfection incidence rates increased from the first reinfection (7.3/100 py, 95% CI 6.2-8.6) to the second reinfection (18.8/100 py, 95% CI 12.9-27.5, p <0.001). This indicates a maintained risk behavior in a potentially specific high risk group, who require urgent targeting for prevention measures related to risk behaviors. How often these individuals require testing for HCV infection de novo is unclear but the present suggestion that this occurs annually does not appear sufficient, and we suggest HCV RNA testing every three to six months after an incident HCV infection and every three months in patients that had been reinfected.
We found a spontaneous HCV clearance rate of 15.6% at the first reinfection episode consistent with findings from others in HIV-HCV coinfected populations [26]. However, at second reinfection the spontaneous clearance rate showed a non-significant increase to 28.6%.
Individuals who had spontaneously cleared their initial infection were more likely to spontaneously clear (p = 0.004) their reinfection, and there was also a trend for lower reinfection incidence for people who had spontaneously cleared their initial infection compared to those that were treated (HR 0.62, 95% CI 0.38-1.02, p = 0.06). These findings would suggest the development of a degree of HCV-specific immunity with repeated exposure, which has been previously demonstrated in humans and chimpanzees [[24], [25]]. The absence of this observation in the setting of PWID may be explained by the type or frequency of HCV exposure, or the testing interval [32].
Several mechanisms of the host's immune response have been associated with spontaneous HCV clearance, including interferon-mediated natural killer cell response [33], a broad multi-specific CD4 T cell response [34], neutralizing antibodies [35], and the presence of specific HLA epitopes [36].
The ability to clear an acute HCV infection spontaneously is reduced in HIV infected patients probably due to a compromised immune response. Factors such as female sex [37], coinfection with hepatitis B virus, a favorable IL28B genotype [38], and a higher CD4 cell count have been associated with clearance in clinical cohorts, as well as higher ALT levels, higher bilirubin levels, a faster decline in HCV viral load [26], and a less diverse viral quasispecies [39]. Likewise, in this study, ALT levels above 1000 IU/ml were associated with spontaneous clearance as it has been described by other groups [26]. These observations potentially reflect a stronger and more directed immune response against HCV in those who clear spontaneously. It remains however unclear why we observed spontaneous clearance in those with their first reinfection that did not clear the succeeding infection.
This study has a number of limitations. First of all, the retrospective nature of the study limits the conclusions that may be drawn from the analysis. In addition, detailed patient information was only available for men that acquired reinfection, limiting the analysis of risk. We may have underestimated the number of reinfections as those that cleared their infection within testing intervals may have been missed. On the other hand, a false positive HCV RNA assay may have led to overestimation of cases. The HCV RNA PCR testing interval was not standardized across centers and depended largely on the treating physician or national guidelines. But we believe that reasonable HCV RNA PCR testing would have been performed as all our patients were HIV-seropositive MSM with a high grade of linkage-to-care and regular medical visits at experienced sexually transmitted disease centers/HIV outpatient clinics, usually every three months. Some datasets were incomplete leading to exclusion from detailed analysis. In addition, the majority of centers did not routinely test for IL28B genotype substantially limiting the analysis of this factor for risk of reinfection and spontaneous clearance.
Our study lacks detailed behavioral information, notably on injection drug use in this population, which hampers conclusions on prevention efforts.
Phylogenetic analysis was not performed in our cohort, and nearly half were reinfected with the same HCV subtype. Others have described a prolonged fluctuating viraemia that may be confounded with a new HCV infection [18]. However, our patients were nearly exclusively diagnosed as acute HCV infection and the median ALT levels at reinfection were 302 and 268 IU/ml at the first and second reinfection, which makes chronic infection less likely. As we included treated patients and did not perform next-generation sequencing, we can not rule out the re-emergence of a resistant minority variant as has been previously postulated [40]. However, the high treatment response rates and high spontaneous clearance rates in our patients' succeeding episodes make this hypothesis unlikely.
Conclusions
HCV reinfection is a critical health concern among HIV infected MSM and frequently occurs after successful treatment or spontaneous clearance of acute HCV infection. Prevention strategies - both treatment and behavioral - are needed to target high risk groups to reduce morbidity and treatment costs. Patients as well as clinicians have to be aware of the specific risk behavior in this setting and counseling should be accompanied by behavioral interventions to avoid reinfections.
HIV-positive MSM with a prior HCV infection should be regularly tested for reinfection. The increase in spontaneous clearance rates observed in our cohort indicates a possible increase of HCV-specific responses with repeated infection.
Introduction
Liver disease represents a major cause of morbidity and mortality among patients infected with the human immunodeficiency virus (HIV) in the developed world [1]. In the setting of effective combined antiretroviral therapy (cART) and the successful preservation of a patient's immune function, chronic infection with hepatitis C virus (HCV) is currently the main cause for liver related mortality due to liver failure and hepatocellular carcinoma [[2], [3]].
In recent years, HCV seroconversions within European HIV cohorts have been reported among people who inject drugs (PWID) and men who have sex with men (MSM) [[4], [5]]. In the case of the MSM community, several outbreaks of acute HCV infection have been described in Western metropolitan areas over the last decade associated with high risk sexual practices, genital ulcer disease and recreational drug use including parenteral administration [[6], [7], [8]]. Treatment uptake with interferon-based therapy has generally been high in the HIV-positive MSM population and high sustained virological response (SVR) rates have been reported as many, if not most, are treated in the acute infection phase [9]. Yet despite these outcomes the epidemic has continued unabated [[5], [10]].
Several new direct-acting antivirals (DAAs) have been approved for interferon-free treatment of chronic HCV in Europe. Most of these agents are characterized by a favorable interaction profile with antiretroviral medication and SVR rates above 90% in clinical trials in the HIV/HCV coinfected population [[11], [12], [13], [14]]. Mathematical modeling predicts if the required scale-up in treatment uptake with these new compounds is achieved the result would be substantial reductions in HCV prevalence in HIV infected MSM within a decade [[15], [16]]. Further benefits have been predicted if treatment is combined with an intervention to reduce behavioral risk, which makes the eradication of HCV an achievable goal in the HIV-HCV coinfected population in Western Europe.
In the presence of maintained risk behavior, HCV reinfections have been described in PWID and MSM who either cleared their initial infection spontaneously or were successfully treated with interferon-based therapies [[17], [18], [19], [20]]. In a recent meta-analysis of 61 studies, the five-year risk of HCV reinfection in HIV infected MSM was as high as 15% and higher than in studies on PWID [21]. Two studies to date have described reinfection incidence among HIV-HCV coinfected MSM with reported rates of 8-15 per 100 person-years (py) [[19], [22]]. More recently, HCV reinfections have also been reported in phase III trials of DAA HCV compounds [[12], [14], [23]] nearly all of which have occurred among HIV infected MSM.
Data from London reported that individuals who spontaneously clear their acute infection may be at lower risk of future HCV reinfection when compared to those who are treated and achieve SVR. This indicates that a degree of protective immunity may develop for some patients [19]. An effective immune response against HCV through multiple infections has been shown in animal models [24]; however, studies among PWID have failed to consistently demonstrate a protective effect [[18], [25]].
An accurate description of the HCV epidemic including a concise observation of reinfections in specific populations will be crucial to achieve the goal of HCV eradication and to reduce costs of repeated DAA treatment. This study quantifies the rate of HCV reinfections among HIV infected MSM from seven urban European areas and investigates potential variables associated with repeat spontaneous viral clearance.
Methods
The dataset for this analysis was merged from eight centers in four countries within the NEAT (European AIDS Treatment Network) consortium: The Chelsea and Westminster Hospital and the Royal Free Hospitals, London, the St. Antoine Hospital, Paris, the Center for Infectiology, Berlin, the Center for Infectious Medicine Hamburg, the Center for HIV and Hepatogastroenterology, Duesseldorf, the University Hospital Bonn, and the Medical University of Vienna.
In all centers, the available data have been homogenized due to previous collaborations such as the NEAT Probe-C cohort.
All HIV-positive MSM from these centers with a history of a cured first HCV infection were identified with subsequent HCV PCR results followed through time to detect reinfection.
HCV cure was defined as follows:
⋅Patients with SVR defined by a negative HCV PCR at least 12 weeks after the end of an interferon-based treatment and at least one subsequent HCV PCR measurement.
⋅Patients with a spontaneously cleared HCV infection, defined by at least two negative HCV PCR measurements at least 24 weeks apart following HCV infection.
The following data were collected for all patients: age, date of diagnosis of acute HCV infection, HCV genotype, date of HCV cure, whether cure was a result of treatment or spontaneous clearance, and date of last follow-up visit.
HCV RNA measurements were not standardized and depended on local operating procedures which ranged between once per year and every three months and in the case of newly developed ALT elevation.
Reinfection was defined as a detectable HCV RNA at any time point after cure, or within the above mentioned time frames, if a HCV genotype/subtype switch occurred. In men who had a reinfection episode, the following data was obtained at the first HCV infection and at each new infection episode: age, duration of HIV infection, date of first HCV infection, HCV genotype, HCV viral load at diagnosis, maximum alanine aminotransferase (ALT), CD4 cell count, HIV viral load, HIV treatment status. Date of HCV cure, whether cure was as a result of treatment or spontaneous clearance and date of last follow-up visit were further collected.
In patients who achieved multiple SVRs or spontaneous clearances, the time point of subsequent reinfections and the described variables were documented.
Reinfection incidence was calculated using Kaplan-Meier survival time methods. The start of follow-up was defined as the date of end of HCV treatment for individuals who were successfully treated and as the first negative HCV RNA PCR for individuals who spontaneously cleared their infection. For individuals that underwent reinfection, the date of failure was taken as the date of newly positive HCV RNA PCR. Individuals who did not undergo reinfection were censored at the date of last negative HCV PCR available. Comparison of reinfection incidences were calculated using log-rank test and Cox proportional hazards model. Variables associated with spontaneous clearance of reinfection were evaluated using logistic regression. Continuous variables were grouped (Age: 30, ALT 1000 IU/ml at initial infection and reinfection, HCV VL of 1st infection 500,000 copies/ml, HIV VL undetectable at 1st infection, CD4 at first infection by brackets of 100 cells/ml, CD4 at reinfection by brackets of 100 cells/ml). All variables that had an association with a p value <0.1 were further explored for association using multiple logistic regression. Only age and initial infection outcomes were available for regression with reinfection as the outcome. McNemar's test was used to compare the spontaneous clearance proportions for the 1st and 2nd reinfections. All analyses were performed using Stata version 12.0.
Results
Six hundred and six HIV-positive MSM with a documented cure of HCV infection between May 2002 and February 2014 were identified. SVR following treatment with pegylated interferon ± ribavirin was achieved in 494 (81.5%) men, while 111 (18.3%) exhibited spontaneous viral clearance, and in one patient the type of cure was not documented.
During follow-up until June 2014, 149 of these men (24.6%) presented with a de novo acute HCV reinfection episode. In 95% (135/142) of cases, an ALT elevation above 41 IU/ml was observed.
The median CD4 cell count at reinfection was 533/mm3 (IQR 412-760) and 82% of reinfected patients had an HIV viral load less than 50 copies/ml at time of reinfection. The median duration of diagnosed HIV infection at HCV reinfection was 9 years (IQR 6-14). Seventy patients had a documented cure or spontaneous clearance of their first reinfection, of which 30 (43%) presented with a second reinfection, a median of 1.8 years (IQR 1.2-2.4) after clearance of their prior infection. Five patients had a third reinfection, and in one patient a fourth reinfection occurred. Table 1 shows details of primary and reinfection genotypes, ages, SVR proportions and spontaneous clearance proportions.
Reinfection incidence
In calculating reinfection incidence, 54 patients from three centers were excluded due to incomplete datasets (no date for end of follow-up, no date for start of follow-up, incorrectly entered end of follow-up date). 552 patients were therefore included in the analysis representing 1952 py of follow-up, with a median follow-up time of 3.0 years (IQR 1.6-4.9, min 0.02/max 11.4 years).
The overall median follow-up time was 3.0 years (interquartile range (IQR) 1.6-4.9 years).
In total, 143 HCV reinfections occurred at a rate of 7.3/100 py (95% confidence interval (CI) 6.2-8.6). The median duration to reinfection was 2.0 years (IQR 1.1-3.3 years). There was a trend for higher reinfection incidence among individuals who achieved SVR following treatment for their incident infection (7.8/100 py) compared to reinfection incidence among individuals who had spontaneously cleared their incident infection (4.9/100 py; crude unadjusted hazard ratio (HR) for reinfection 0.62, 95% CI 0.32-0.95, p = 0.06). Fig. 1 is a Kaplan-Meier curve showing survival from reinfection for all patients with 95% CI.
Sixty-four men either spontaneously cleared or were successfully treated for their subsequent HCV reinfection and had follow-up data available representing 143 py of follow-up (median follow-up 1.8 years, IQR 0.9-2.8). Of these 64 men, 27 presented with a second reinfection at a median of 1.7 years (IQR 1.2-2.4) after cure of the prior infection. The second reinfection incidence rate was significantly higher than the first reinfection incidence at 18.8/100 py (95% CI 12.9-27.5; HR for second reinfection 2.51, 95% CI 1.7-3.8, p <0.001).
Table 2 depicts the reinfection incidence per center, with the highest being in Paris (21.8/100py, 95% CI 11.3-41.8), followed by Vienna (16.8/100 py, 95% CI 8.7-32.3), Berlin (8.2/100 py, 95% CI 5.6-12.1), Duesseldorf (8.1/100 py, 95% CI 4.6-14.3), and London/Chelsea Westminster (7.0/100 py, 95% CI 5.3-9.1). The lowest in incidence rate was seen in Hamburg (5.0/100 py, 95% CI 2.9-8.7). The incidence rate decreased only slightly over time (Fig. 2).
Spontaneous clearance rates
Twenty-one of 135 patients (15.6%) spontaneously cleared their first reinfection and 7 of 22 patients (28.6%) spontaneously cleared their second reinfection (p = 0.43 for increase in spontaneous clearance proportion).
Men who spontaneously cleared their incident infection were less likely to present with a subsequent reinfection episode than men who had achieved SVR to their incident infection (OR 0.52, 95% CI 0.29-0.91, p = 0.02). In multivariable analysis, spontaneous clearance of the first HCV infection (OR = 7.47, 95% CI 1.9-29.2, p = 0.004) and a maximum ALT level above 1000 IU/ml (OR = 13.9, 95% CI 4.3-45.4, p <0.001) were associated with spontaneous clearance of the reinfection [26].
|
|
| |
| |
|
|
|