| |
High Efficacy of ABT-493 and ABT-530 Treatment in Patients With HCV Genotype 1 or 3 Infection and Compensated Cirrhosis
|
| |
| |
Download the PDF here
Download the PDF here
Gastroenterology oct 2016 - Edward Gane,1 Fred Poordad,2 Stanley Wang,3 Armen Asatryan,3 Paul Y. Kwo,4 Jacob Lalezari,5 David L. Wyles,6 Tarek Hassanein,7 Humberto Aguilar,8 Benedict Maliakkal,9 Ran Liu,3 Chih-Wei Lin,3 Teresa I. Ng,3 Jens Kort,3 and Federico J. Mensa3
1University of Auckland, Auckland, New Zealand; 2Texas Liver Institute, University of Texas Health Science Center, San Antonio, Texas; 3AbbVie, Inc, North Chicago, Illinois; 4Indiana University School of Medicine, Indianapolis, Indiana; 5Quest Clinical Research, San Francisco, California; 6University of California San Diego, La Jolla, California; 7Southern California GI and Liver Centers and Southern California Research Center, Coronado, California; 8Louisiana Research Center, Shreveport, Louisiana; 9University of Rochester Medical Center, Rochester, New York
AASLD: SURVEYOR-II, PART 3: EFFICACY AND SAFETY OF GLECAPREVIR/PIBRENTASVIR (ABT-493/ABT-530) IN PATIENTS WITH HEPATITIS C VIRUS GENOTYPE 3 INFECTION WITH PRIOR TREATMENT EXPERIENCE AND/OR CIRRHOSIS - (11/14/16)
AASLD: A Randomized, Phase 3 Trial of Sofosbuvir/Velpatasvir/Voxilaprevir for 8 Weeks and Sofosbuvir/Velpatasvir for 12 Weeks for Patients with Genotype 3 HCV Infection and Cirrhosis: The POLARIS-3 Study - (11/15/16)
EASL: ENDURANCE-3: SAFETY AND EFFICACY OF GLECAPREVIR/PIBRENTASVIR COMPARED TO SOFOSBUVIR PLUS DACLATASVIR IN TREATMENT-NAïVE HCV GENOTYPE 3-INFECTED PATIENTS WITHOUT CIRRHOSIS - (04/21/17)
Background & Aims
The combination of ABT-493 (NS3/4A protease inhibitor) plus ABT-530 (NS5A inhibitor) has shown high rates of sustained virologic response at post-treatment week 12 (SVR12) in noncirrhotic patients infected with hepatitis C virus (HCV) genotypes (GTs) 1-6. We describe 2 open-label phase 2 studies investigating the efficacy and safety of ABT-493 plus ABT-530 with or without ribavirin (RBV) in GT1- or GT3-infected patients with compensated cirrhosis.
Methods
Patients with GT1 infection received 200 mg ABT-493 plus 120 mg ABT-530 for 12 weeks. Patients with GT3 infection were randomized 1:1 to receive 300 mg ABT-493 plus 120 mg ABT-530 with or without once-daily 800 mg RBV for 12 weeks; treatment-experienced patients who were not treated with RBV received 16 weeks of therapy. Efficacy was measured by SVR12, defined as an HCV-RNA level less than 25 IU/mL. Adverse events and laboratory parameters were evaluated throughout the study.
Results
Twenty-seven patients with GT1 infection and 55 patients with GT3 infection were enrolled. The majority were treatment-naive (84%) and male (65%). In patients with GT1 infection, SVR12 was achieved by 96% (26 of 27; 95% confidence interval [CI], 82-99) of patients, with 1 relapse. Among GT3-infected patients, SVR12 was achieved in 96% (27 of 28; 95% CI, 82-99) of patients in the RBV-free arm (1 relapse), and in 100% (27 of 27; 95% CI, 88-100) in the RBV-containing arm. The most common adverse events were headache, fatigue, and nausea. Laboratory abnormalities were rare; no patient discontinued treatment.
Conclusions
In cirrhotic HCV GT1- or GT3-infected patients, ABT-493 plus ABT-530 with or without RBV achieved SVR12 rates of 96%-100% and was well tolerated.
Patients with chronic hepatitis C virus (HCV) infection and cirrhosis are at greatest risk for progression to liver decompensation and death.1 The 2-year survival rates decrease from 90% to 54% once cirrhosis progresses to liver decompensation, as defined by ascites, jaundice, encephalopathy, or variceal bleeding.2 Achievement of a sustained virologic response (SVR) reduces the risk of hepatic decompensation events, end-stage liver disease, hepatocellular carcinoma, and liver-related mortality.3, 4, 5 Recognizing the need to prevent these complications, the American Association for the study of Liver Diseases/Infectious Disease Society of America, and the European Association for the Study of the Liver recommendations initially prioritized treatment of patients with cirrhosis, as did many health care systems around the world.6, 7, 8
Because the prevalence and burden of HCV-related cirrhosis is projected to increase significantly in the coming years,9, 10, 11 treatments that maximize SVR in the more difficult-to-cure patients with cirrhosis are needed. Significant improvements to safety and effectiveness in treatment of chronic HCV infection have occurred with first-generation interferon-free therapies, however, patients with genotype (GT) 3 infection remain a difficult-to-cure population and available treatments for GT1a-infected patients may require longer duration and/or ribavirin (RBV) co-administration.12 The current interferon-free standard of care for GT3-infected patients with cirrhosis, with reported SVR rates at post-treatment week 12 (SVR12) of 86%-88%, is daclatasvir plus sofosbuvir with or without RBV for 24 weeks.13, 14 Although more treatment options exist for GT1a-infected patient with cirrhosis, most are recommended for use with RBV, 24-week treatment duration, and/or baseline testing of viral resistance to direct-acting antivirals.7, 15, 16, 17, 18, 19 Interestingly, sofosbuvir plus peginterferon and RBV for 12 weeks remains a viable treatment option for patients with GT3 infection (93% SVR12) for those able to tolerate interferon.20 Therefore, a pangenotypic, interferon, and RBV-free therapy that offers shorter treatment duration with efficacy comparable with patients without cirrhosis is highly desired for patients with compensated cirrhosis.
ABT-493 is an HCV nonstructural (NS) protein 3/4A protease inhibitor, and ABT-530 is an HCV NS5A inhibitor. Both compounds have potent antiviral activity in vitro against all major HCV GTs, with a 50% effective concentration (EC50) for ABT-493 ranging from 0.9 to 2.8 nmol/L across replicons of 6 subgenotypes, and for ABT-530 ranging from 1 to 4 pmol/L across replicons of 8 subgenotypes. The antiviral activities of both compounds also are maintained against variants carrying common single amino acid substitutions that confer resistance to earlier-generation HCV direct-acting antiviral therapies, providing a high barrier to the development of resistance.21, 22, 23 For example, GT3 NS5A substitutions M28T, A30K, or Y93H each confers minimal increases in ABT-530 EC50 (0.4- to 2.3-fold), but confers 10- to 6700-fold increases in EC50 for other available NS5A inhibitors.24, 25, 26, 27 No NS3/4A protease inhibitors currently are indicated for use in GT3-infected patients. ABT-493 is highly active against wild-type HCV GT1a, GT1b, as well as GT3a, and maintains activity against variants carrying common NS3 amino acid substitutions in these genotypes at positions 155 or 168, which confer a high level of resistance to most earlier-generation protease inhibitors.28
In part 1 and part 2 of the SURVEYOR-I and SURVEYOR-II phase 2 studies, the all-oral, once-daily regimen of ABT-493 and ABT-530 showed high efficacy and good tolerability in patients without cirrhosis across all 6 major GTs without the need for RBV (unpublished data). In part 1, all 40 GT1-infected patients without cirrhosis treated for 12 weeks with 200 mg ABT-493 plus 120 mg ABT-530 achieved SVR12 irrespective of baseline substitutions that confer resistance to earlier-generation NS3/4A or NS5A inhibitors. In addition, among 30 GT3-infected patients without cirrhosis treated for 12 weeks with a higher dose of 300 mg ABT-493 plus 120 mg ABT-530, 28 (93%) achieved SVR12 with one virologic failure (post-treatment relapse). These encouraging results prompted initiations of part 2 of these studies to determine the efficacy and safety of the combination of ABT-493 and ABT-530 in patients with compensated cirrhosis and HCV GT1 or GT3 infection. The impact of adding 800 mg once-daily RBV in GT3-infected patients also was investigated.
Materials and Methods
Patients
Patients ages 18-70 years with chronic HCV GT1 or GT3 infection were screened at 41 sites in the United States, Canada, United Kingdom, New Zealand, Australia, and Puerto Rico. Patients were either treatment-naive or previously treated with peginterferon/RBV, excluding prior treatment with any direct-acting antiviral. Genotypes and subtypes were determined by the Versant HCV Genotype Inno-LiPA Assay, version 2.0 or higher (Siemens Healthcare Diagnostics, Tarrytown, NY). The presence of compensated cirrhosis (Child-Pugh score, ≤6) was required with cirrhosis determination based on one of the following criteria: histologic diagnosis of cirrhosis on liver biopsy conducted before or during screening by the local laboratory (eg, METAVIR score of >3, including 3/4 or 3-4; Ishak score of >4); or a screening FibroTest (Biopredictive; Paris, France) result that is 0.75 or greater and an alanine aminotransferase (ALT) to platelet ratio index greater than 2 (patients with conflicting FibroTest and ALT to platelet ratio index results underwent a qualifying FibroScan or liver biopsy); or a FibroScan score of 14.6 kPa or greater within 6 months of screening or during screening (patients with indeterminate FibroScan (Echosens; Paris, France) score [eg, 12.5 to <14.6] underwent a qualifying liver biopsy).
Patients were excluded for a calculated creatinine clearance less than 50 mL/min, albumin or hemoglobin level less than the lower limit of normal, ALT or aspartate aminotransferase level more than 5 times the upper limit of normal (ULN), platelet count less than 90 x 109/L, or with evidence of hepatitis B virus or human immunodeficiency virus at screening.
Key eligibility criteria are described in the Supplementary text
.
Study Design and Dose Selection
Patients with GT1 infection (SURVEYOR-I) received 200 mg ABT-493 plus 120 mg ABT-530 for 12 weeks, whereas GT3-infected patients underwent stratified 1:1 randomization to receive 300 mg ABT-493 plus 120 mg ABT-530 either with or without once-daily 800 mg RBV for 12 weeks (SURVEYOR-II) (Figure 1). The RBV dose selected was based on the prediction for reduced frequency of anemia compared with weight-based dosing while maintaining a high likelihood of SVR based on prior pharmacokinetic and pharmacodynamics findings with sofosbuvir plus RBV.29, 30 Both studies were open label without active or placebo comparator arms. Doses of ABT-493 and ABT-530 were selected based on the efficacy and safety findings of part 1 of the SURVEYOR studies in which 200 mg ABT-493 and 120 mg ABT-530 in GT1-infected noncirrhotic patients achieved 100% SVR12 compared with 97% in patients receiving 200 mg ABT-493 plus 40 mg ABT-530 (unpublished data). In addition, a higher dose of 300 mg ABT-493 plus 120 mg ABT-530 had fewer virologic failures than lower doses of either drug in GT3-infected noncirrhotic patients and was found to be well tolerated. Pharmacokinetic data also supported this dose selection for patients with compensated cirrhosis.23
Randomized GT3-infected patients were stratified by HCV treatment history (naive or peginterferon/RBV-experienced). Enrollment was halted for treatment-experienced cirrhotic patients with GT3 infection based on feedback from the US Food and Drug Administration to first show efficacy in treatment-naive cirrhotic and/or treatment-experienced noncirrhotic patients given limited data in these populations. A protocol amendment extended the duration of treatment to 16 weeks for the initial 4 treatment-experienced patients already randomized to the RBV-free arm when the regulatory agency's feedback was received; the initial 3 treatment-experienced patients already randomized to the RBV-containing arm received 12 weeks of treatment. Enrollment continued for treatment-naive GT3-infected patients, who received 12 weeks of treatment with (n = 24) or without (n = 24) RBV.
Patients were required to stop treatment owing to virologic failure if they met any of the following criteria: confirmed increase from nadir in HCV-RNA level, defined as 2 consecutive HCV-RNA measurements greater than 1 log10 IU/mL above nadir at any time point during treatment; failure to achieve unquantifiable HCV-RNA level by week 6; or confirmed quantifiable HCV-RNA level at any point after an unquantifiable level.
This study was conducted in accordance with the International Conference of Harmonization guidelines, applicable regulations, and guidelines governing clinical study conduct and ethical principles that have their origin in the Declaration of Helsinki. All patients provided written informed consent. The sponsor conducted the data analyses and the authors had full access to relevant data, reviewed, and approved the final manuscript.
Efficacy, Viral Resistance, and Safety Assessments
Plasma samples for HCV RNA measurements were collected at screening, days 1 and 3, treatment weeks 1, 2, 4, 6, 8, 10, and 12 (and week 16 if applicable, or early discontinuation), and post-treatment weeks 2, 4, 8, 12, and 24 (or early discontinuation). The primary efficacy end point was the percentage of patients who achieved SVR12, defined as an HCV-RNA level below the lower limit of quantitation using the COBAS TaqMan real-time reverse transcriptase-PCR assay with High Pure System v.2.0 (Roche, Molecular Systems Inc, Pleasanton, CA). The lower limit of detection for both GT1 and GT3 for this assay was 15 IU/mL and the lower limit of quantification was 25 IU/mL. Secondary end points included the percentage of patients with SVR at post-treatment week 4, with on-treatment virologic failure, and with post-treatment relapse.
Viral RNA isolated from plasma samples collected at baseline from all patients was used to identify NS3 or NS5A substitutions known to confer resistance to at least 1 direct-acting antiviral by population sequencing (detection threshold, ∼15%) or next-generation sequencing with a detection threshold of 15% (for 1 GT3b patient). For patients who experienced virologic failure and had a postbaseline HCV-RNA sample with 1000 IU/mL or higher, NS3 and/or NS5A substitutions in available postbaseline samples also were identified by population sequencing or next-generation sequencing and compared with baseline and reference sequences. Genotype 1a NS3 amino acid substitutions included in the analysis were V36A/G/I/L/M, F43L, T54A/S, V55A/I, Y56H, Q80K/R, V107I, S122G/R, I132V, R155(all), A156(all), V158I, D168(all), and I170F/T/V. Genotype 1b NS3 amino acid substitutions included in the analysis were V36A/G/I/L/M, T54A/C/G/S, V55A, Y56H/L, Q80K/R, V107I, S122A/D/G/I/N/T, R155(all), A156(all), V158I, D168(all), V170A/T, and M175L. For NS5A, the following amino acid substitutions were included in the analysis: GT1a: M28(all), Q30(all), L31(all), P32L, H58D/R, and Y93(all); GT1b: L28(all), P29S/del, R30G/H/P/Q, L31(all), P32L/del, P58A/S, Q62D, A92E/K, and Y93(all). GT3 amino acid substitutions at any of the following amino acid positions were included in the analyses: for NS3: 36, 56, 80, 155, 156, 166, and 168; and for NS5A: 24, 28, 29, 30, 31, 32, 58, 92, and 93.
Treatment-emergent adverse events (AEs) were collected beginning on day 1 until 30 days after the last dose of study drug, and were assessed for severity and relation to study drug and RBV. Serious AEs were recorded throughout the post-treatment period. At each study visit, patients underwent a routine physical examination, were monitored for vital signs, and had samples collected for clinical chemistry and hematology.
Statistical Analyses
All analyses were conducted on the intent-to-treat population, defined as all patients who received at least 1 dose of ABT-493 and ABT-530. Efficacy and safety populations were based on the intent-to-treat population. Confidence intervals (CIs) for binomial proportions were 2-sided, with an α level of .05 and calculated using the Wilson score method. Analyses were performed using SAS version 9.3 (SAS Institute, Inc, Cary, NC).
Results
Patient Demographics and Baseline Characteristics
Patients were enrolled between August 20, 2014, and July 29, 2015, and comprised 27 patients with GT1 infection and 55 patients with GT3 infection (Supplementary Figure 1). The majority of enrolled GT1- or GT3-infected patients were infected with subgenotypes 1a (20 of 27; 74%) or 3a (51 of 55; 93%), respectively (Table 1). Patients were predominantly male (65%) and treatment-naive (84%). In patients with GT1 infection, baseline NS3 substitutions conferring resistance to earlier-generation drugs were identified in 11 (41%) patients and NS5A substitutions were present in 5 (19%) patients; 3 (11%) patients had substitutions in both antiviral targets. NS5A substitutions in GT1-infected patients included M28V, Q30R, R30Q, and L31M; none possessed Y93H (Supplementary Table 1
Supplementary Data
). In patients with GT3 infection, baseline NS3 and NS5A substitutions were present in 13 (24%) and 12 (22%) patients, respectively; 4 (7%) GT3-infected patients had substitutions in both targets (Supplementary Table 2
Supplementary Data
). NS5A substitutions present at baseline in patients with GT3a infection included A30K/T, P58S, and Y93H. The 1 patient with a GT3b infection had no baseline substitutions in NS3, but K30M and V31M in NS5A.
Efficacy Outcomes
All 82 patients completed the planned duration of treatment. Among patients with GT1 infection who received 200 mg ABT-493 plus 120 mg ABT-530 for 12 weeks, SVR12 was achieved in 96% (26 of 27; 95% CI, 82-99) of patients (Figure 2). All 6 GT1-infected patients with prior peginterferon/RBV treatment experience and 20 of 21 (95%) GT1a-infected patients achieved SVR12. One treatment-naive female patient with GT1a infection relapsed at post-treatment week 4. At baseline, this patient had substitutions I170V in NS3 and L31M in NS5A, neither of which confer resistance to ABT-493 and ABT-530, respectively. At the time of virologic failure, NS3 I170V as well as NS5A L31M and Y93N were detected by population sequencing in this patient. The combination of NS5A substitutions L31M and Y93N confers a 195-fold increase in EC50 to ABT-530. Drug compliance was reportedly 100% in this patient.
In GT3-infected patients who received 300 mg ABT-493 plus 120 mg ABT-530, the SVR12 rate was 96% (27 of 28; 95% CI, 82-99). Among these 28 patients, 24 of 24 (100%) treatment-naive and 3 of 4 (75%) treatment-experienced patients achieved SVR12. One treatment-experienced male with GT3 infection relapsed at post-treatment week 2 after receiving treatment for 16 weeks. At baseline, this patient had NS3 substitution A166S, which confers no change in EC50 for ABT-493, and no substitutions in NS5A. At the time of virologic failure, this patient had no substitutions in NS3 and the M28G substitution in NS5A. A replicon containing NS5A M28G is not viable in vitro (replication efficiency, <5%), and therefore the fold change in EC50 this substitution confers to ABT-530 could not be determined. This patient was 95% or more compliant with study drugs and had comparable pharmacokinetic exposures at day 1 and week 4 as the remainder of the cohort.
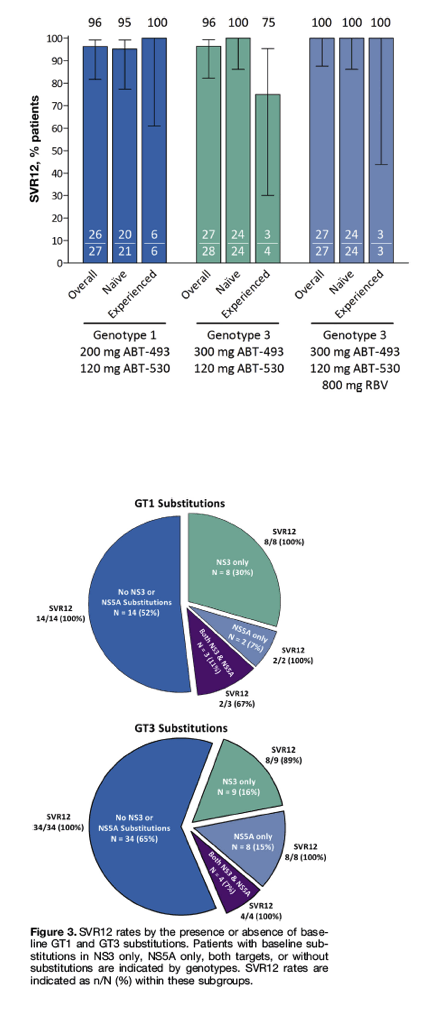
SVR12 was achieved by 100% (27 of 27; 95% CI, 88-100) of GT3-infected patients with cirrhosis who received 300 mg ABT-493 plus 120 mg ABT-530 with once-daily RBV for 12 weeks (Figure 2). SVR12 was achieved in 100% of GT1- or GT3-infected patients without baseline substitutions and efficacy remained high in the presence of baseline substitutions (Figure 3). All 11 GT3-infected patients with baseline NS5A substitutions, including 4 patients with additional NS3 substitutions, achieved SVR12.
Figure 2
SVR at post-treatment week 12. Intent-to-treat SVR12 rates are shown for GT1- or GT3-infected patients overall and by prior treatment status (naive or peginterferon/RBV-experienced). All patients received 12 weeks of treatment except for the 4 treatment-experienced patients with GT3 infection randomized to the RBV-free arm who received treatment for 16 weeks.
Safety Findings
Overall, 74% (61 of 82) of patients experienced AEs (Table 2), and the majority of AEs were mild or moderate in severity. No patient discontinued treatment prematurely owing to AEs. The most common AEs in patients receiving treatment without RBV were headache (15%), diarrhea (13%), and fatigue (11%). In GT3-infected patients receiving treatment plus once-daily RBV, AEs were more frequent, particularly headache (33%), fatigue (30%), nausea (22%), and insomnia (19%).
Serious AEs were reported in 5 patients, of which 1 event was considered to have a reasonable possibility of being related to ABT-493, ABT-530, and RBV. A 57-year-old GT3-infected man receiving ABT-493 plus ABT-530 and RBV experienced delusional disorder 3 days after the end of treatment after admitted amphetamine and alcohol use on the day of the event. The other serious AEs considered unrelated to ABT-493 and ABT-530 were dysplastic liver nodules (day 100), anemia (considered related to RBV only), pulmonary embolism (post-treatment day 6), and tibia fracture (post-treatment day 15). No clinical outcomes of hepatic decompensation were observed.
Laboratory Assessments
All patients with baseline ALT increases greater than the ULN (n = 74) had normalization of their levels during the course of treatment. No patient experienced a postnadir grade 2 (>3 x ULN) ALT increase (Table 2). One grade 3 laboratory abnormality was observed, a total bilirubin increase greater than 3 x ULN in a patient receiving treatment without RBV. At baseline, this patient had an increased total bilirubin level of 1.6 mg/dL and an ALT concentration of 82 U/L. Abnormal total bilirubin levels persisted during treatment, peaking on day 44 (5.5 mg/dL, composed of 3.1 mg/dL direct bilirubin and 2.4 mg/dL indirect bilirubin), returned to normal by post-treatment day 28, and remained normal throughout follow-up evaluation. This grade 3 increase was not coincident with a transaminase increase.
One patient in the RBV-containing arm had a single hemoglobin level measurement less than 10 g/dL and the RBV dosage was reduced; no patient had a decrease below 8 g/dL (Table 2). Five (19%) patients modified the RBV dose during treatment. One of these modifications was to correct administration of weight-based RBV (1000 mg/day) to the protocol-specified dosage of 800 mg/day. The remaining 3 RBV dose modifications were the result of AEs of depression, fatigue, insomnia, irritability, nausea, and headache in 1 patient, and a hemoglobin decrease greater than 2 g/dL in 2 patients as specified in the protocol for patients with a history of stable cardiac disease.
Discussion
The patient populations studied here include some of the more difficult to cure with direct-acting antiviral regimens and for whom there is still a need for improvement over currently available treatment options. The once-daily regimen of ABT-493 plus ABT-530 with or without RBV achieved high SVR12 rates and was well tolerated in HCV GT1- or GT3-infected patients with compensated cirrhosis. Twelve-week treatment was highly efficacious with or without RBV in treatment-naive GT3-infected patients (100%), and 1 treatment-experienced patient experienced relapse after 16-week treatment without RBV. Use of a lower dose of ABT-493 (200 mg) in GT1-infected patients resulted in high SVR12 rates with 1 post-treatment relapse. Overall, SVR12 rates were high irrespective of baseline substitutions in NS3 and/or NS5A known to confer resistance to earlier-generation compounds (31 of 33; 94%), or other baseline demographic or disease characteristics, consistent with data in noncirrhotic GT3-infected patients treated with this regimen (unpublished data).
Response rates in GT3-infected patients with cirrhosis for the combination of daclatasvir plus sofosbuvir with RBV for 12 weeks (71%-83%), or 24 weeks (81%-92%), or without RBV for 24 weeks (86%) are lower than those that have come to be expected with newer direct-acting antiviral therapies in other genotypes.13, 14, 30 Baseline NS5A resistance-associated substitutions confer significant reductions in susceptibility to daclatasvir, particularly Y93H, which confers a more than 2000-fold increase in EC50 for daclatasvir.31 Newer HCV regimens may provide improved genotypic coverage but SVR rates have room for improvement in the most difficult-to cure populations. The RBV-free regimen of sofosbuvir plus velpatasvir for 12 weeks achieved 91% SVR12 in GT3-infected patients with cirrhosis.32
Whether longer treatment duration or the addition of RBV would improve efficacy of sofosbuvir plus velpatasvir was not assessed. As with other regimens, NS5A baseline resistance-associated substitutions, including Y93H, reduced the SVR12 rates in patients receiving sofosbuvir plus velpatasvir (84%).32 In this study, baseline substitutions had minimal impact on SVR12 rates with ABT-493 plus ABT-530. Among those with baseline substitutions, there were 4 GT3-infected patients with NS5A Y93H at baseline, and all 4 achieved SVR12. One GT1a-infected patient had NS5A L31M at baseline and experienced relapse with the addition of a treatment-emergent Y93N substitution. The prevalence of HCV variants with both L31M and Y93N in NS5A treatment-naive GT1a-infected patients is expected to be very low owing to the infrequency of individual L31 (∼1%-5%) or Y93 (∼2%) baseline substitutions reported.33, 34, 35, 36
The safety profiles of ABT-493 and ABT-530 without RBV were favorable compared with the RBV-containing arm, and comparable across the treatment arms that did not include RBV (ie, low and high doses of ABT-493). The rates of some AEs were numerically higher with the higher ABT-493 dose, although the sample sizes were small and this was a cross-study comparison. To this point, a pooled analysis of 531 patients from the SURVEYOR studies did not identify a significant difference in safety profile of ABT-493 200-mg and 300-mg doses.37 Higher frequencies of the most common AEs were observed in the 800-mg once-daily RBV-containing arm than the RBV-free arms. There were no grade 2 ALT increases after normalization or treatment discontinuations resulting from AEs, and clinically significant laboratory abnormalities were rare. Overall, the higher frequency of AEs in the RBV-containing arm, in combination with the observation that RBV did not significantly improve SVR12 rates, suggests low utility of RBV for this regimen. Moreover, these results suggest that the higher dose of 300 mg ABT-493 in combination with 120 mg ABT-530 for patients with compensated cirrhosis would optimize the balance between safety and efficacy when treating chronic HCV infection across genotypes.
Generalizability of the results was limited by a few factors. Small numbers of treatment-experienced patients, particularly with GT3 infection, were enrolled as a result of feedback from the US Food and Drug Administration. One of 4 GT3-infected treatment-experienced patients receiving treatment without RBV for 16 weeks relapsed. Given the limited number of treatment-experienced GT3-infected patients, the treatment duration in this patient population requires further study. Few patients were enrolled with subgenotypes other than 1a and 3a, although GT1b historically has been easier to cure with currently approved direct-acting antiviral regimens, even in the presence of baseline NS3 and/or NS5A substitutions. Finally, laboratory values for study eligibility (eg, platelet, albumin, and liver enzyme levels) may not be representative of a wider population with advanced liver disease.
Although not included in this study, patients with severe or end-stage kidney disease are predicted to be able to be treated with ABT-493 and ABT-530 because both agents have negligible renal excretion.38 These drugs were well tolerated in HCV-uninfected patients with renal impairment and can be administered without dose adjustment.38
In conclusion, the results of these phase 2 studies suggest that the once-daily combination of ABT-493 and ABT-530 without RBV for 12 weeks is sufficient to achieve high rates of SVR12 in patients with GT1 or GT3 infection and compensated cirrhosis. The unique potency of these agents against all genotypes, even in the presence of common NS3 and/or NS5A baseline substitutions that confer resistance to most contemporary NS3/4A protease inhibitors and NS5A inhibitors, offers the potential for a pangenotypic therapy without RBV co-administration. Therefore, phase 3 studies of this regimen are investigating the combination of 300 mg ABT-493 plus 120 mg ABT-530 without RBV in patients with compensated cirrhosis across all HCV genotypes, including dedicated cohorts of treatment-experienced patients with GT3 infection, patients with renal impairment, or those who previously failed treatment with direct-acting antiviral-containing regimens.
|
|
| |
| |
|
|
|