 |
 |
 |
| |
Virologic Success Rate 90% at Week 24 With First-Line DTG/3TC in ACTG A5353: ACTG A5353: A pilot study of dolutegravir (DTG) + lamivudine (3TC) for initial treatment of HIV-1-infected participants with HIV-1 RNA <500,000 copies/ml
|
| |
| |
9th IAS Conference on HIV Science (IAS 2017), July 23-26, 2017, Paris
Mark Mascolini
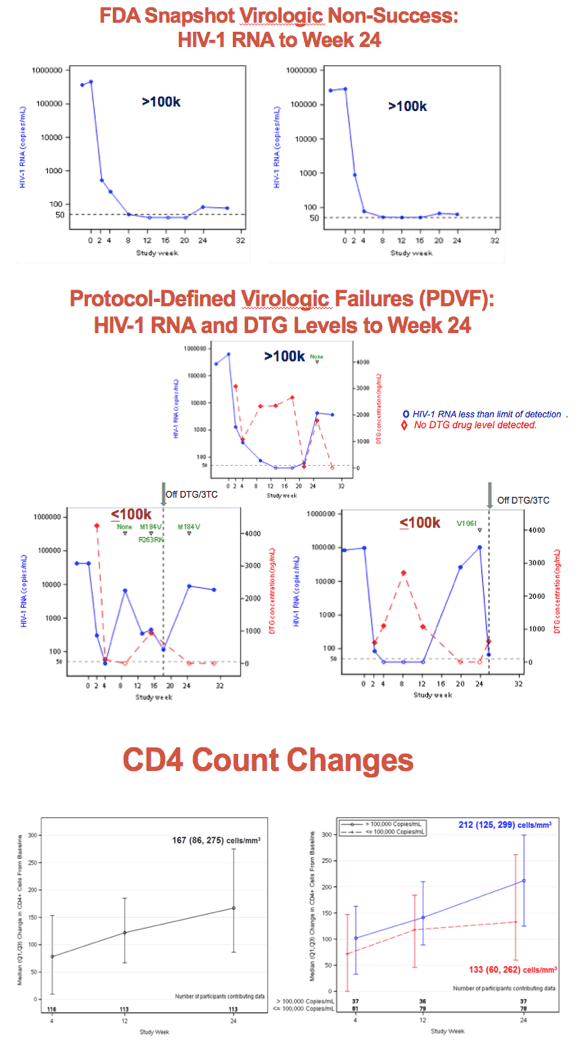
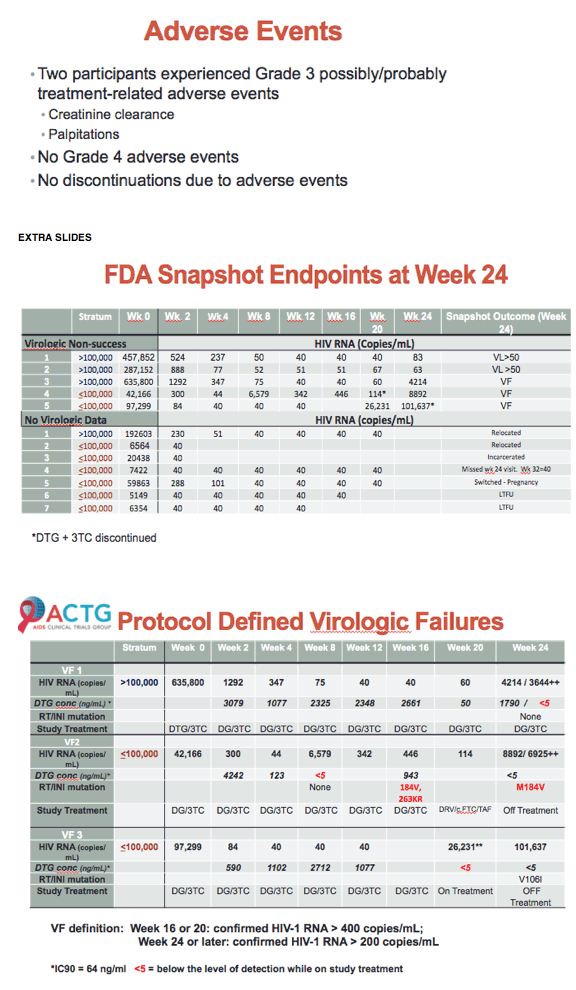
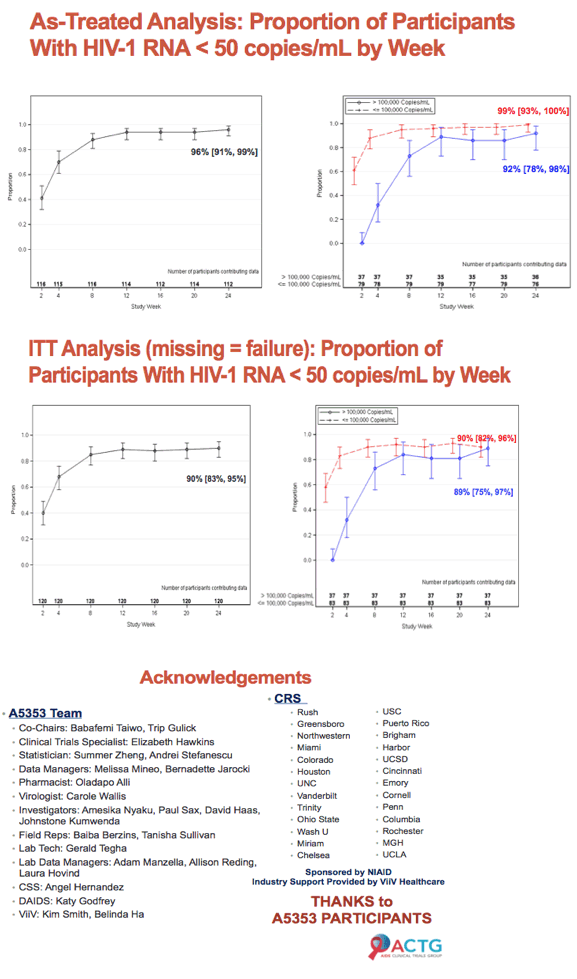
After 24 weeks of first-line dolutegravir/lamivudine (DTG/3TC), 108 of 120 ACTG A5353 participants (90%) had a viral load below 50 copies [1]. There were only 3 protocol-defined virologic failures, all in people with poor adherence. Pretreatment viral loads measured up to 500,000 copies.
ACTG investigators believe DTG/3TC is an attractive first-line candidate because both antiretrovirals are potent and well tolerated, because DTG has a high barrier to resistance, and because it should be possible to combine the drugs in one pill. In a pilot study of 50/300 mg of DTG/3TC once daily, PADDLE, only 1 of 20 participants had protocol-defined virologic failure at week 48 [2]. But that trial excluded people with a viral load above 100,000 copies.
ACTG A5353 enrolled antiretroviral-naive people with a pretreatment viral load between 1000 and 500,000 copies and with no mutations in reverse transcriptase, integrase, or protease. Participants had to be negative for hepatitis B surface antigen; they could have HCV but were not expected to begin HCV therapy during the study period. The primary outcome was virologic success after 24 weeks of therapy, defined as an on-treatment load below 50 copies with the FDA snapshot algorithm. Protocol-defined virologic failure meant (1) a confirmed load above 400 copies at week 16 or 20 or (2) a confirmed load above 200 copies at week 24 or later.
Among 120 people who began DTG/3TC at 50/300 mg once daily, 37 (31%) had a viral load above 100,000 copies. Median age stood at 30 years in the under-100,000 and over-100,000 groups, 86% and 89% were men, and 29% and 27% were white. Median pretreatment viral load stood at 4.23 log in the under-100,000 group and at 5.23 log in the over-100,000 group. Respective median CD4 counts were 413 and 350.
At 24 weeks 75 of 83 people (90%) in the under-100,000 group and 33 of 37 (89%) in the over-100,000 group met the study definition of virologic success (viral load below 50 copies) for an overall success rate of 90%. Three people in the over-100,000 group and none in the under-100,000 group had a viral load above 50 copies. Two people in the under 100,000 group and none in the other group dropped out of the study for reasons other than a viral load above 50 copies. Two people in the under-100,000 group and one in the over-100,000 group had protocol-defined virologic failure. All had evidence of suboptimal antiretroviral adherence. Genotyping detected an evolving integrase mutation (R263RK) and a reverse transcriptase mutation (M184V) in one person with virologic failure.
Median CD4 count rose by 133 cells at 24 weeks in the under-100,000 group and by 212 cells in the over-100,000 group. Two people had a grade 3 adverse event possibly or probably related to study drug (palpitations and low creatinine clearance). There were no grade 4 adverse events and no one stopped study drugs for adverse events.
Two large randomized trials of DTG/3TC versus DTG/TDF/FTC, GEMINI 1 and GEMINI 2 [3,4], are under way and should provide more insight into the resistance barrier with this regimen.
References
1. Taiwo BO, Zheng L, Nyaku AN, et al. ACTG A5353: a pilot study of dolutegravir (DTG) + lamivudine (3TC) for initial treatment of HIV-1-infected participants with HIV-1 RNA < 500,000 copies/mL. 9th IAS Conference on HIV Science (IAS 2017), July 23-26, 2017, Paris. Abstract MOAB0107LB.
2. Cahn P, Rolon MJ, Figueroa MI, Gun A, Patterson P, Sued O. Dolutegravir-lamivudine as initial therapy in HIV-infected, ARV naive patients: 48 week results of the PADDLE trial. 21st International AIDS Conference (AIDS 2016). July 18-22, 2016. Durban, South Africa. Abstract FRAB0104LB. http://www.natap.org/2016/IAC/IAC_62.htm
3. ClinicalTrials.gov. An efficacy, safety, and tolerability study comparing dolutegravir plus lamivudine with dolutegravir plus tenofovir/emtricitabine in treatment naive HIV infected subjects (Gemini 1). https://clinicaltrials.gov/ct2/show/NCT02831673
4. ClinicalTrials.gov. An efficacy, safety, and tolerability study comparing dolutegravir (DTG) plus lamivudine (3TC) with dolutegravir plus tenofovir/emtricitabine in treatment naive HIV infected subjects (Gemini 2). https://clinicaltrials.gov/ct2/show/NCT02831764




|
| |
|
 |
 |
|
|