 |
 |
 |
| |
Only 5% DAA Failure Rate in Large French Cohort With HCV/HIV
|
| |
| |
9th IAS Conference on HIV Science (IAS 2017), July 23-26, 2017, Paris
Mark Mascolini
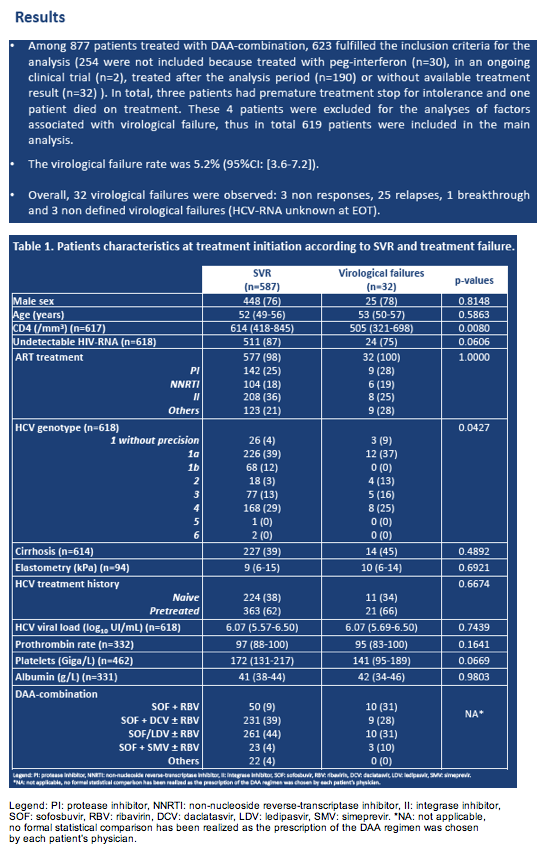
Only 32 of 619 HCV/HIV-coinfected people in the French HEPAVIH cohort (5.2%) had virologic failure with direct-acting antivirals (DAAs) for HCV [1]. Pretreatment with non-DAA regimens and 12 versus 24 weeks of therapy predicted virologic failure in a marginal structural model.
HEPAVIH investigators noted that the main cause of DAA failure in people infected only with HCV is severity of cirrhosis, indicated by ascites, low albumin, or high total bilirubin. Because factors linked to DAA failure in HCV/HIV-coinfected people are not well established, the researchers conducted this study. They focused on coinfected cohort members taking a DAA regimen with or without ribavirin and without peg-interferon. No one was enrolled in clinical trials, and all had sustained virologic response (SVR) data available.
Among 623 people who met enrollment criteria, the researchers excluded 4 who prematurely stopped treatment because of intolerance or who died during treatment. Among the remaining 619 cohort members, 587 had an SVR and 32 (5.2%, 95% confidence interval [CI] 3.6 to 7.2) did not. Of these 32 virologic failures, the HEPAVIH investigators attributed 3 to nonresponse, 25 to relapse, 1 to breakthrough, and 3 to unknown causes.
People with virologic failure had a lower median CD4 count (505 versus 614, P = 0.008) and tended to include a lower proportion with an undetectable HIV load (75% versus 87%, P = 0.0606) and a lower platelet count (141 versus 172, P = 0.0669). Presence of cirrhosis or type of DAA regimen did not affect chances of virologic failure.
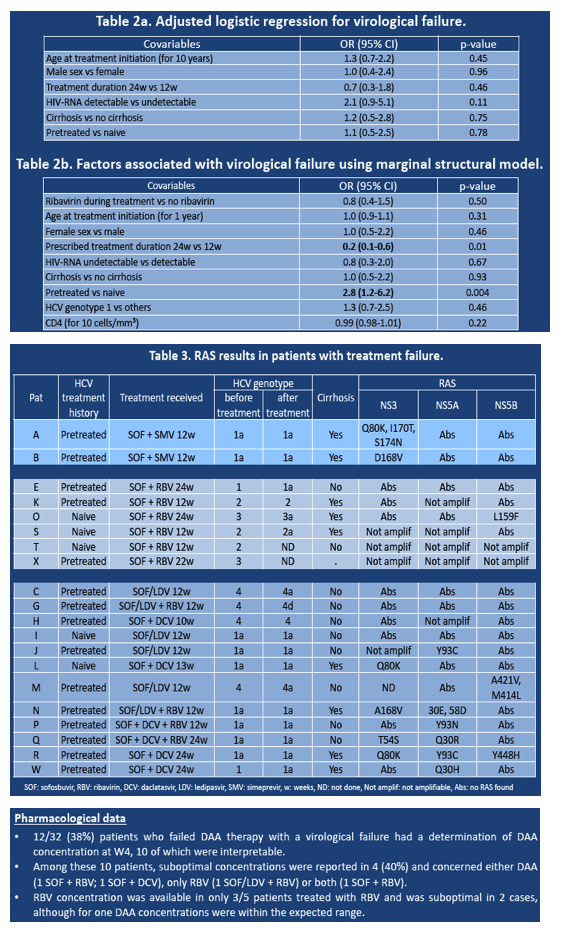
A marginal structural model adjusted for ribavirin use, age, sex, cirrhosis, HCV genotype, and CD4 count identified two independent predictors of virologic failure: Pretreatment with a non-DAA regimen almost tripled the chance of failure (odds ratio [OR] 2.8, 95% CI 1.2 to 6.2, P = 0.004), and DAA treatment for 24 weeks versus 12 weeks lowered the chance of failure 80% (OR 0.2, 95% CI 0.1 to 0.6, P = 0.01). Age, gender, HCV genotype, HIV load, and CD4 count did not predict failure in this analysis.
The investigators suggested that faulty DAA adherence may also have contributed to virologic failure, indicated by (1) suboptimal DAA concentrations in 4 of 10 people with virologic failure and pharmacologic data that could be interpreted, and (2) a higher rate of detectable HIV RNA among people with DAA failure in a separate logistic regression analysis (OR 2.1, 95% CI 0.9 to 5.1, P = 0.11).
Reference
1. Salmon D, Gilbert C, Chas J, et al. Association of uncontrolled HIV-RNA with failure to direct acting antiviral (DAA) combinations in HIV/HCV co-infected patients on antiretroviral therapy (ART): ANRS CO13 HEPAVIH cohort. 9th IAS Conference on HIV Science (IAS 2017), July 23-26, 2017, Paris. Abstract TUPEB0385.
-------------------
Failure to direct acting antiviral (DAA) combinations in HIV/HCV coinfected patients is rare and higher in pretreated patients - ANRS CO13 HEPAVIH cohort
D. Salmon1,2, C. Gilbert3, J Chas4, L. Piroth5, K. Lacombe6, C Katlama7, P. Trimoulet8, C. Solas9, G. Peytavin10, H Aumaitre11, L Alric12, F Boue13, P Morlat14, I Poizot-Martin15, E Billaud16, E Rosenthal17, A Naqvi18, P Miailhes19, L. Esterle3, P. Carrieri20,21, P. Sogni22,23, F. Dabis24, L. Wittkop3,25,26 for the ANRS CO13 Hepavih study group
1Department of Internal Medicine and Infectious Diseases, Hotel Dieu, Cochin Hospital, AP-HP, Paris, France. 2Paris Descartes University, Sorbonne Paris Cite, Paris, France. 3INSERM, ISPED, Centre INSERM U1219- Epidemiologie-Biostatistique, Bordeaux, France. 4Assistance Publique des Hopitaux de Paris, Hopital Tenon, Service Maladies infectieuses et tropicales, Paris, France. 5Department of Infectiology, CHU Dijon, Dijon, France. 6Department of Infectious Diseases, Saint-Antoine Hospital, AP-HP, Paris, France. 7Assistance Publique des Hopitaux de Paris, Hopital Pitie-Salpetriere, Service Maladies infectieuses et tropicales, Paris, France. 8Department of Virology, CHU Pellegrin, Bordeaux, France. 9APHM, Hopital La Timone, Laboratoire de Pharmacocinetique et Toxicologie, Marseille, France. 10APHP, Service de pharmaco-virologie, Paris, France. 11Centre Hospitalier de Perpignan, Service Maladies infectieuses et tropicales, Perpignan, France 12Centre Hospitalier Universitaire de Toulouse, Hopital Purpan, Service Medecine interne-Pole Digestif, Toulouse, France. 13Assistance Publique des Hopitaux de Paris, Groupe Hospitalier Paris Sud, Hopital Antoine-Beclere, Service Medecine interne et immunologie, Clamart, France. 14Centre Hospitalier Universitaire de Bordeaux, Service de medecine interne, Hopital Saint-Andre, Bordeaux, France. 15Aix Marseille Univ, APHM Sainte-Marguerite, Service d'Immuno-hematologie clinique, Marseille, France. 16Centre Hospitalier Universitaire de Nantes, Service Maladies infectieuses et tropicales, Nantes, France. 17Department of Internal Medicine, CHU de Nice, Archet Hospital, University of Nice Sophia Antipolis,Nice, France. 18Department of Infectious Diseases, CHU de Nice, Archet Hospital, Nice, France. 19Service des Maladies Infectieuses et Tropicales, CHU Lyon, Hopital de la Croix Rousse, Lyon, France. 20INSERM, U912 (SE4S)-University of Aix Marseille, IRD, France. 21UMR-S912-ORS PACA, Marseille, France. 22Hepatology Unit, Cochin Hospital, AP-HP, Paris, France. 23Cochin Institute, INSERMU1016, Paris, France. 24ANRS, Paris, France. 25CHU de Bordeaux, Pole de Sante Publique, Service d'Information Medicale, Bordeaux, France. 26Universite de Bordeaux, ISPED, Centre INSERM U1219-Epidemiologie-Biostatistique, Bordeaux, France.

Failure to direct acting antiviral (DAA) combinations in HIV/HCV coinfected patients is rare and higher in pretreated patients - ANRS CO13 HEPAVIH cohort
D. Salmon1,2, C. Gilbert3, J Chas4, L. Piroth5, K. Lacombe6, C Katlama7, P. Trimoulet8, C. Solas9, G. Peytavin10, H Aumaitre11, L Alric12, F Boue13, P Morlat14, I Poizot-Martin15, E Billaud16, E Rosenthal17, A Naqvi18, P Miailhes19, L. Esterle3, P. Carrieri20,21, P. Sogni22,23, F. Dabis24, L. Wittkop3,25,26 for the ANRS CO13 Hepavih study group
1Department of Internal Medicine and Infectious Diseases, Hotel Dieu, Cochin Hospital, AP-HP, Paris, France. 2Paris Descartes University, Sorbonne Paris Cite, Paris, France. 3INSERM, ISPED, Centre INSERM U1219- Epidemiologie-Biostatistique, Bordeaux, France. 4Assistance Publique des Hopitaux de Paris, Hopital Tenon, Service Maladies infectieuses et tropicales, Paris, France. 5Department of Infectiology, CHU Dijon, Dijon, France. 6Department of Infectious Diseases, Saint-Antoine Hospital, AP-HP, Paris, France. 7Assistance Publique des Hopitaux de Paris, Hopital Pitie-Salpetriere, Service Maladies infectieuses et tropicales, Paris, France. 8Department of Virology, CHU Pellegrin, Bordeaux, France. 9APHM, Hopital La Timone, Laboratoire de Pharmacocinetique et Toxicologie, Marseille, France. 10APHP, Service de pharmaco-virologie, Paris, France. 11Centre Hospitalier de Perpignan, Service Maladies infectieuses et tropicales, Perpignan, France 12Centre Hospitalier Universitaire de Toulouse, Hopital Purpan, Service Medecine interne-Pole Digestif, Toulouse, France. 13Assistance Publique des Hopitaux de Paris, Groupe Hospitalier Paris Sud, Hopital Antoine-Beclere, Service Medecine interne et immunologie, Clamart, France. 14Centre Hospitalier Universitaire de Bordeaux, Service de medecine interne, Hopital Saint-Andre, Bordeaux, France. 15Aix Marseille Univ, APHM Sainte-Marguerite, Service d'Immuno-hematologie clinique, Marseille, France. 16Centre Hospitalier Universitaire de Nantes, Service Maladies infectieuses et tropicales, Nantes, France. 17Department of Internal Medicine, CHU de Nice, Archet Hospital, University of Nice Sophia Antipolis,Nice, France. 18Department of Infectious Diseases, CHU de Nice, Archet Hospital, Nice, France. 19Service des Maladies Infectieuses et Tropicales, CHU Lyon, Hopital de la Croix Rousse, Lyon, France. 20INSERM, U912 (SE4S)-University of Aix Marseille, IRD, France. 21UMR-S912-ORS PACA, Marseille, France. 22Hepatology Unit, Cochin Hospital, AP-HP, Paris, France. 23Cochin Institute, INSERMU1016, Paris, France.

|
| |
|
 |
 |
|
|