 |
 |
 |
| |
No HIV Infections With Generic TDF/FTC PrEP in Large London Group
|
| |
| |
9th IAS Conference on HIV Science (IAS 2017), July 23-26, 2017, Paris
Mark Mascolini
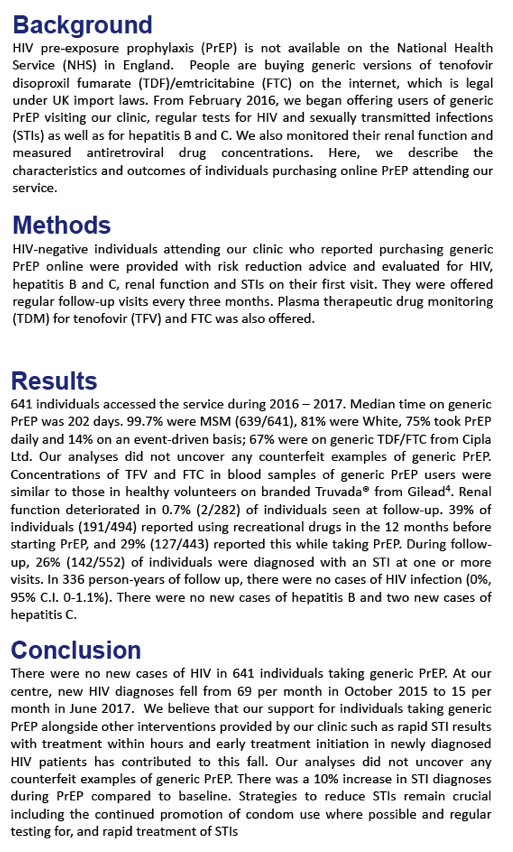
None of 641 people buying generic tenofovir DF/emtricitabine (TDF/FTC) on the Internet and using it for pre-exposure prophylaxis (PrEP) became infected with HIV during 202 days of follow-up [1]. At the London clinic these PrEP users attended, new HIV diagnoses fell from 69 to 15 per month around the time people started using generic PrEP.
Because the UK National Health Service does not pay for PrEP, some people who want to use it buy generic TDF/FTC on the Internet. Since February 2016 clinicians at a London's Chelsea and Westminster Hospital HIV clinic began offering generic PrEP users regular testing for HIV and other sexually transmitted infections (STIs), including HBV and HCV. Clinicians also monitored renal function in this group and measured their tenofovir and FTC concentrations.
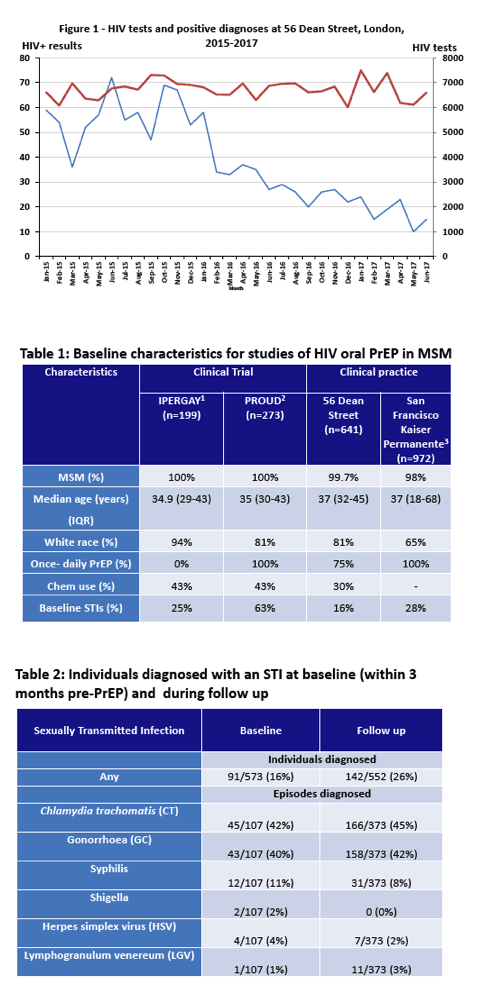
So far 641 generic PrEP users have taken advantage of these clinic services. Almost all, 99.7%, were men who have sex with men, 81% were white, 75% took PrEP daily, and 14% took PrEP as needed. Median age of the group stood at 37 years, and median time using generic PrEP measured 202 days. Participants bought generic TDF/FTC from Cipla, Emcure, or Mylan. Clinicians spotted no fake TDF/FTC during follow-up. Blood levels of the drugs proved similar to those of healthy volunteers taking branded TDF/FTC (Truvada).
Renal function declined in 2 of 282 people assessed (0.7%). Before starting PrEP, 39% of the group used recreational drugs, and 29% reported using recreational drugs after starting PrEP.
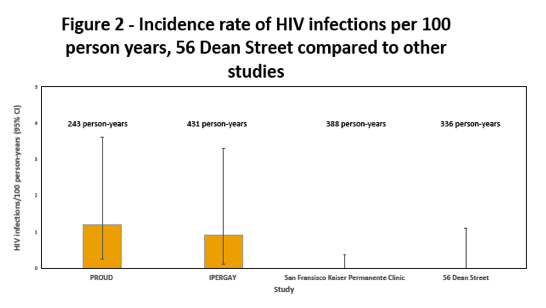
Before starting PrEP, 91 of 573 people (16%) had an STI. While taking PrEP, 142 of 552 people tested (26%) picked up a new STI. No one acquired HBV infection and 2 picked up HCV. Through 336 person-years of follow-up, no one became infected with HIV (0%, 95% confidence interval 0 to 1.1%).
From October 2015 through June 2017, new HIV diagnoses dropped from 69 to 15 per month at this London clinic. The researchers attribute that decline to (1) support for people taking generic PrEP, (2) rapid STI results and treatment, and (3) prompt antiretroviral therapy for people diagnosed with HIV.
Reference
1. Aloysius I, Zdravkov J, Savage A, et al. InterPrEP (II): Internet based pre-exposure prophylaxis with generic tenofovir DF/emtricitabine in London--an analysis of outcomes in 641 patients. 9th IAS Conference on HIV Science (IAS 2017), July 23-26, 2017, Paris. Abstract WEPEC0962.
------------------

InterPrEP (II): Internet based pre-exposure prophylaxis with generic tenofovir DF/emtrictabine in London - an analysis of outcomes in 641 patients
56 Dean Street
Chelsea and Westminster Hospital NHS Foundation Trust
Isaac Aloysius1, Jey Zdravkov1, Anna Savage2 Gary Whitlock1, Xinzhu Wang3,
Isaac Day-Weber3, Rachel Smith5, Andrew Hill4, Myra McClure3, Marta Boffito 1,3,, Nneka Nwokolo1
1. 56 Dean Street, Chelsea and Westminster Hospital, London, UK. 2. School of Medicine, Cardiff University, UK. 3. Imperial College London, UK
4. Department of Translational Medicine, University of Liverpool, UK. 5. MetaVirology Ltd, London, UK.
References: 1. Molina et al. On-Demand Pre-exposure Prophylaxis in Men at High Risk for HIV-1 Infection. N Engl J Med 2015; 373:2237-2246. 2. McCormack et al. Pre-exposure prophylaxis to prevent the acquisition of HIV-1 infection (PROUD): effectiveness results from the pilot phase of a pragmatic open-label randomised trial. The Lancet; 387: 53 – 60. 3. Volk et al. No New HIV Infections With Increasing Use of HIV Pre-exposure Prophylaxis in a Clinical Practice Setting. Clin Infect Dis 2015; 61:1601-1603. 4. Wang X et al. InterPrEP: internet-based pre-exposure prophylaxis with generic tenofovir disoproxil fumarate/emtrictabine in London – analysis of pharmacokinetics, safety and outcomes. HIV Med 2017doi:10.1111/hiv.12528

|
| |
|
 |
 |
|
|