 |
 |
 |
| |
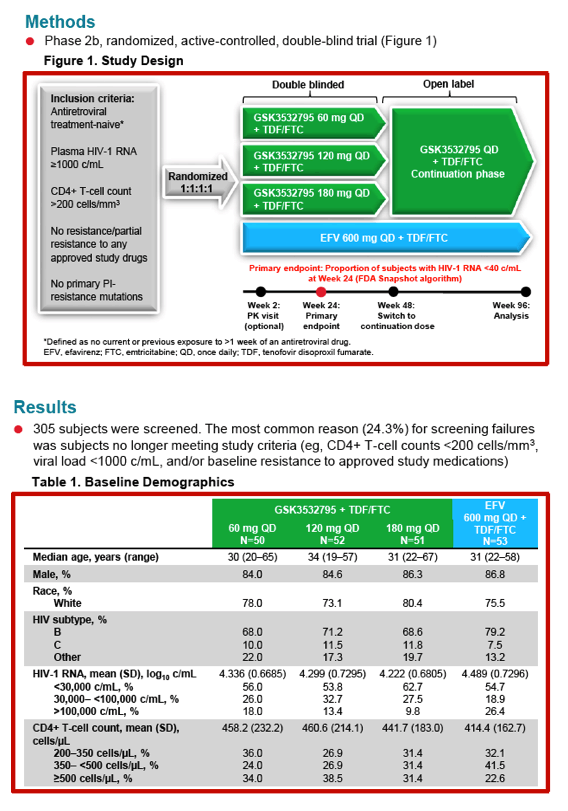
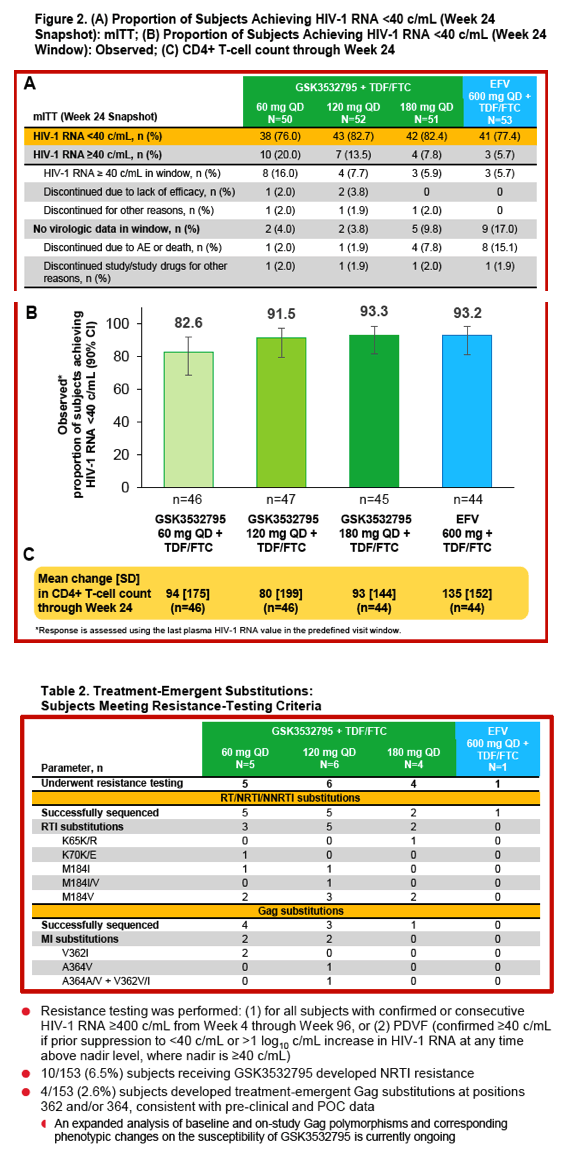
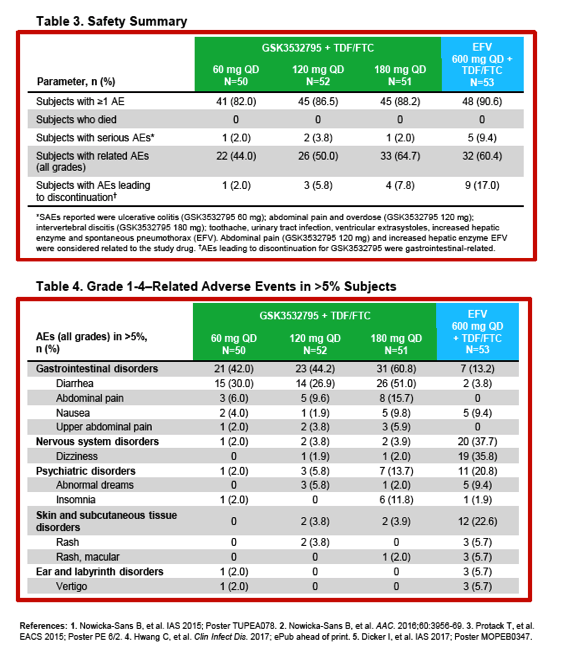
Safety, Efficacy, and Dose-Response of GSK3532795/BMS-955176 Plus Tenofovir/Emtricitabine in Treatment-Naïve HIV-1-Infected Adults: Week 24 Primary Analysis
|
| |
| |
Reported by Jules Levin
9th IAS Conference on HIV Science; July 23-26, 2017; Paris, France
J. Morales-Ramirez,1J. Bogner,2J.-M. Molina,3J. Lombaard,4I. Dicker,5D. Stock,6S. Min,7C. Llamoso,5S.R. Joshi,5M. Lataillade5
1Clinical Research Puerto Rico Inc., San Juan, Puerto Rico; 2Med IV, Hospital of the University of Munich, Munich, Germany; 3Hôpital St Louis, Paris, France; 4Josha Research, Bloemfontein, South Africa; ViiV Healthcare, Wallingford, CT, USA; 6Bristol-Myers Squibb, Wallingford, CT, USA; 7ViiV Healthcare, Research Triangle Park, NC, USA
Pharm Wk: Early Safety, Tolerability and Pharmacokinetic Profile of GSK2838232, a Novel 2nd Generation HIV Maturation Inhibitor, as Assessed in Healthy Subjects - (06/20/17)
HIV Combinectin GSK3732394: A Long-Acting Inhibitor With Multiple Modes of Action - (10/30/16)
HIV-1 Attachment Inhibitor Prodrug BMS-663068 [GSK3684934 (formerly BMS-663068)] in Antiretroviral-Experienced Subjects: Week 96 Safety Analysis - (11/02/16)
HIV-1 Attachment Inhibitor Prodrug BMS-663068 [GSK3684934 (formerly BMS-663068)] in Antiretroviral-Experienced Subjects: Week 96 Subgroup Analysis - (10/30/16)
The Effect of Food on the Pharmacokinetics of the HIV-1 Attachment Inhibitor Temsavir, the Active Moiety of the Prodrug Fostemsavir - (06/26/17)
The Effect of Increased Gastric pH on the Bioavailability of Extended Release Tablet of Fostemsavir in Healthy Subjects / Combinectin-GSK3732394 / 2nd Gen Maturation Inhibitor GSK2838232 - (06/26/17)

|
| |
|
 |
 |
|
|