 |
 |
 |
| |
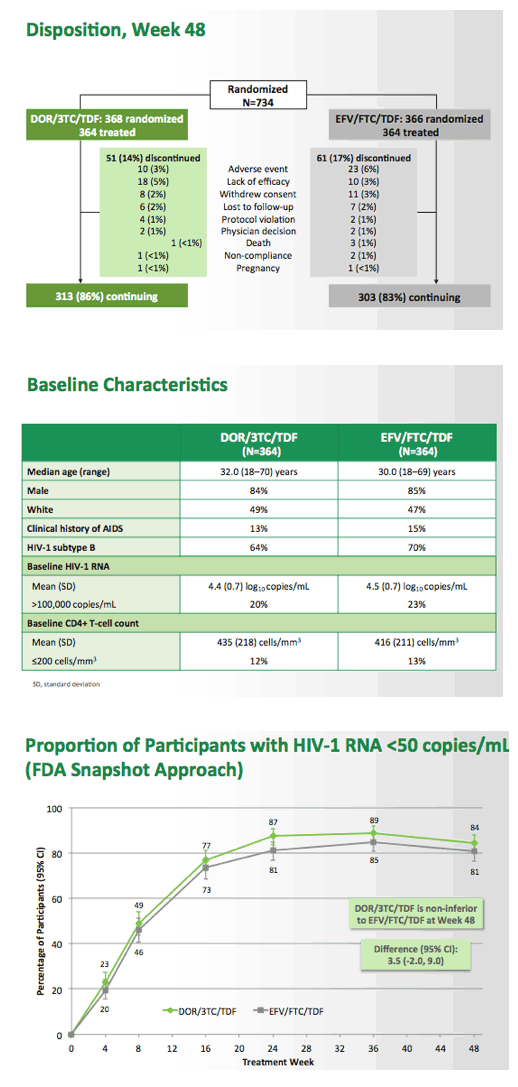
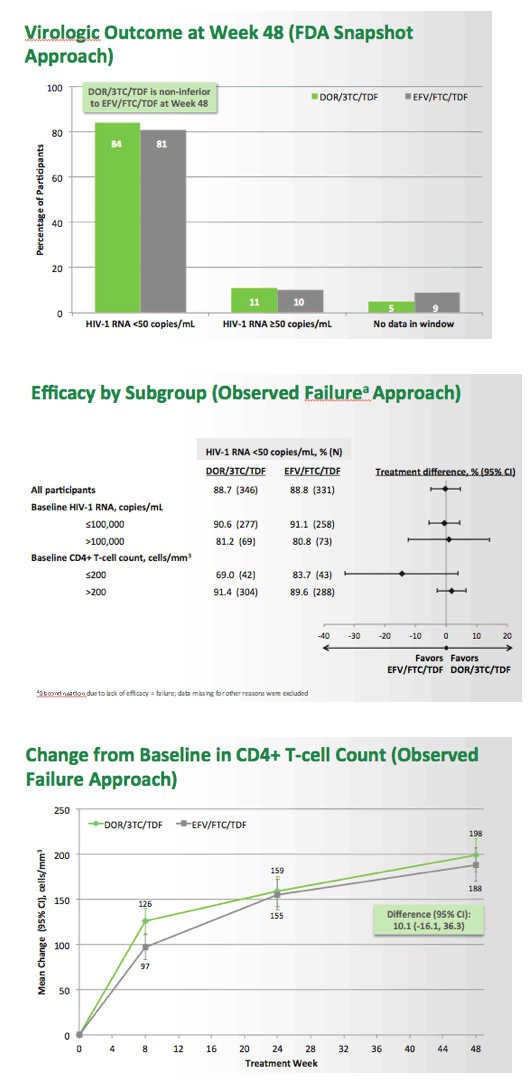
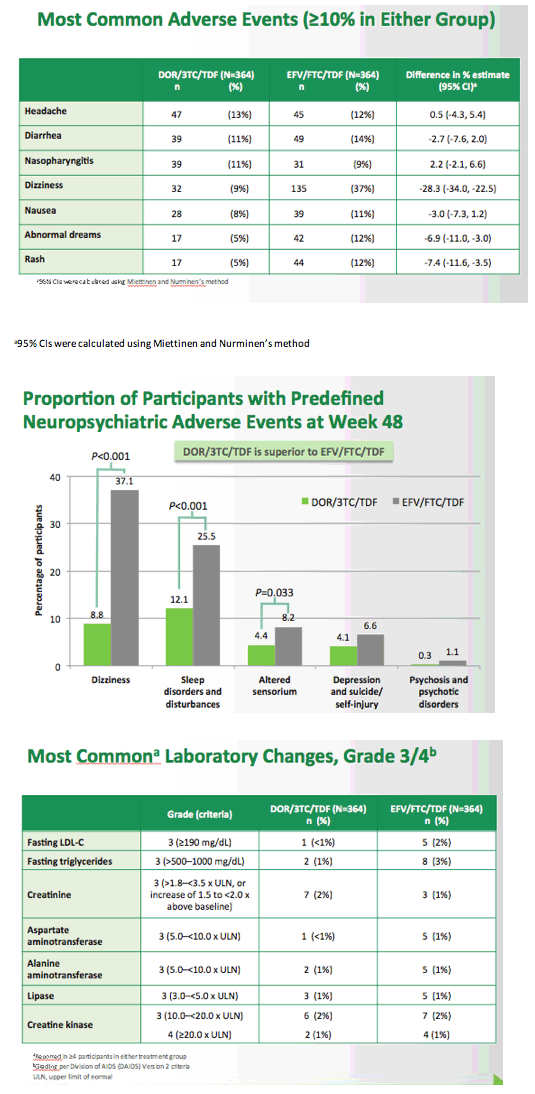
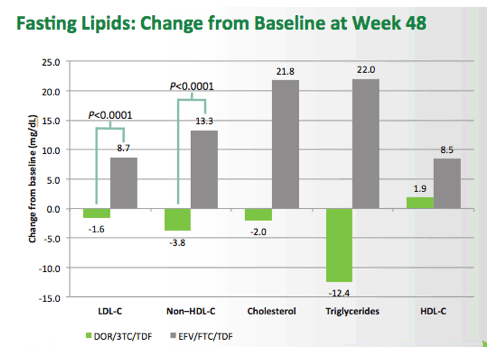
Fixed-Dose Combination of Doravirine/Lamivudine/TDF is Non-Inferior to Efavirenz/Emtricitabine/TDF in Treatment-Naïve Adults With HIV-1 Infection: Week 48 Results of the Phase 3 DRIVE-AHEAD Study
|
| |
| |
Doravirine in Non-Inferior To Darunavir+Ritonavir in Phase 3 Treatment-Naive Trial at Week 48 - (02/16/17)
Doravirine 100 mg QD vs Efavirenz +TDF/FTC in ART-Naïve HIV+ Patients: Week 48 Results - (02/26/16)
Reported by Jules Levin
9th IAS Conference on HIV Science (IAS 2017), July 23-26, 2017, Paris
Kathleen E Squires1, Jean-Michel Molina2, Paul E Sax3, Wing-Wai Wong4,
Chloe Orkin5, Otto Sussmann6, Richard Kaplan7, Lisa Lupinacci8,
Anthony Rodgers8, Xia Xu8, Gina Lin8, Sushma Kumar8, Peter Sklar8,
Bach-Yen Nguyen8, George J Hanna8, Carey Hwang8, Elizabeth Martin8 on behalf of the DRIVE-AHEAD study team
1Thomas Jefferson University, Philadelphia, PA, USA; 2University of Paris Diderot, Hopital Saint-Louis, Paris, France; 3Brigham and Women's Hospital, Harvard Medical School, Boston, MA, USA; 4Taipei Veterans General Hospital, Taipei, Taiwan; 5Royal London Hospital, London, UK; 6Asistencia Cientifica de Alta Complejidad SAS, Bogota, Colombia; 7Desmond Tutu HIV Foundation, Cape Town, South Africa; 8Merck & Co., Inc., Kenilworth, NJ, USA



|
| |
|
 |
 |
|
|