 |
 |
 |
| |
Zoledronic Acid Boosts Bone Density More Than Switch From TDF
|
| |
| |
9th IAS Conference on HIV Science (IAS 2017), July 23-26, 2017, Paris
Mark Mascolini
Intravenous zoledronic acid boosted bone mineral density (BMD) higher than switching from tenofovir disoproxil fumarate (TDF) in a randomized trial including mostly white adult men [1]. The investigators cautioned that only larger and longer studies can determine the relative impact of these strategies on fracture risk.
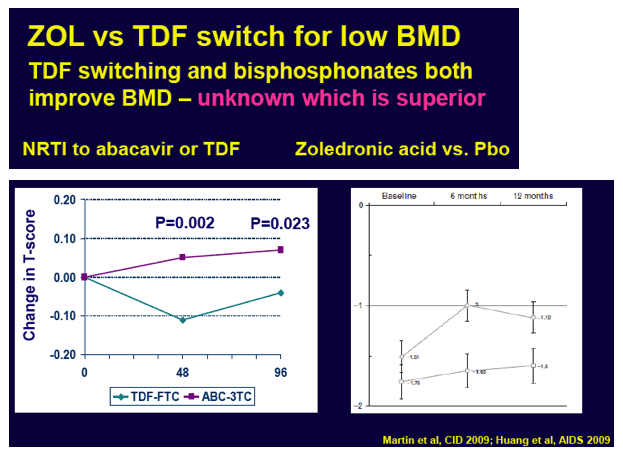
Low BMD in antiretroviral-treated people begins improving when patients switch from TDF to abacavir, an integrase inhibitor, or tenofovir alafenamide (TAF) or when they start bisphosphonate therapy with zoledronic acid or alendronate. Because the relative merits of those two strategies remained unknown, researchers in Australia and Spain conducted the ZEST trial. The primary outcome was average percent change in lumbar spine BMD by DXA scan. The investigators chose the spine instead of the hip because spine BMD responds more quickly to treatment than hip BMD.
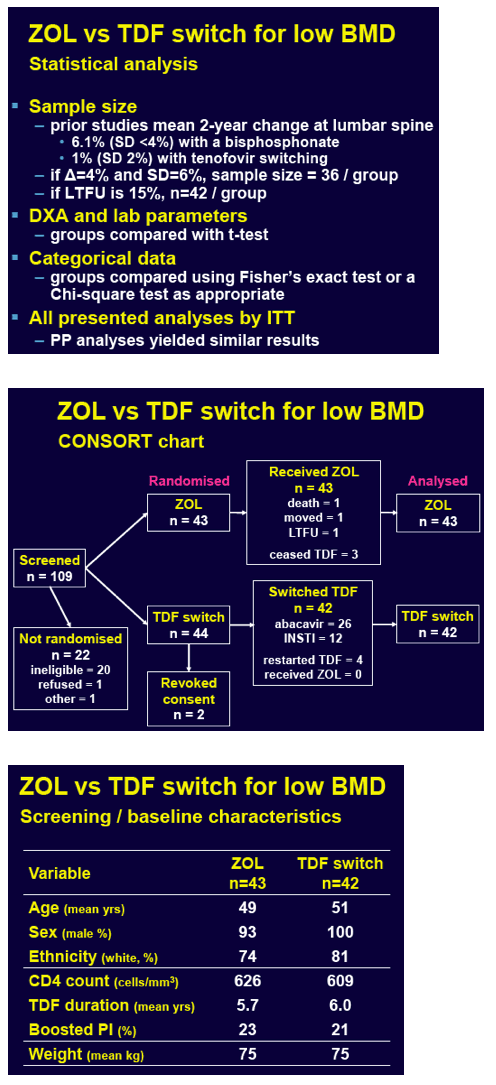
The ZEST team recruited adults with a viral load below 50 copies for at least 3 months while taking a TDF-containing regimen for at least 6 months. No one had a history of virologic failure, resistance, or a reason not to start a proposed antiretroviral replacement for TDF. The study also excluded people who had already taken a bisphosphonate, had secondary causes of osteoporosis, or needed therapy for low BMD. The researchers randomized participants to trade TDF for another antiretroviral or to take zoledronic acid (5 mg by intravenous infusion at months 0 and 12) while continuing TDF. All participants took 1500 mg of calcium daily and had vitamin D replacement to promote BMD gains and prevent zoledronic acid-induced low calcium.
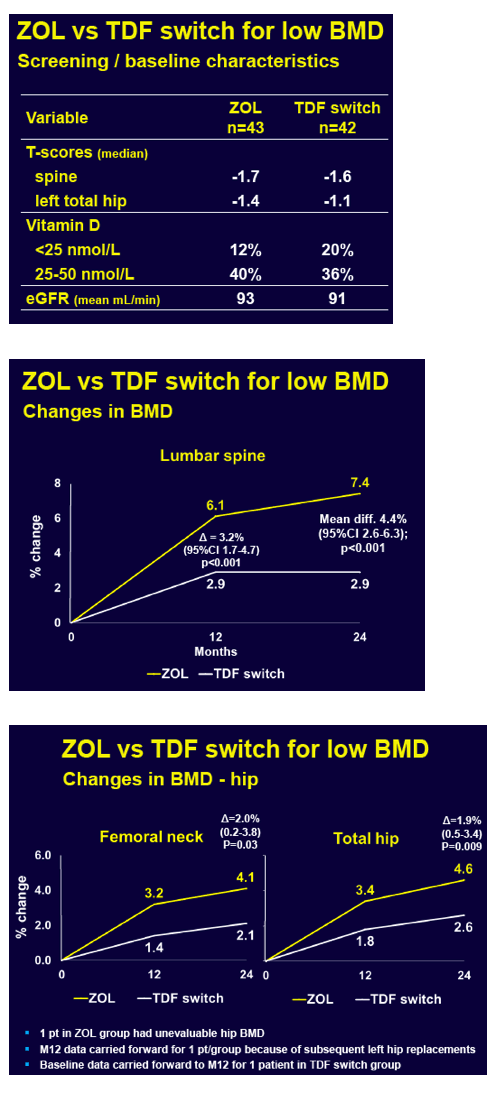
ZEST investigators randomized 43 people to zoledronic acid and 44 to switch TDF. Two people in the TDF-switch group withdrew consent and are not considered in the analysis. Age averaged 49 in the zoledronic acid group and 51 in the TDF-switch group. Respective proportions of men were 93% and 100%, while proportions of whites were 74% and 81%. The zoledronic acid group and the TDF-switch group had taken TDF for an average 5.7 and 6.0 years.
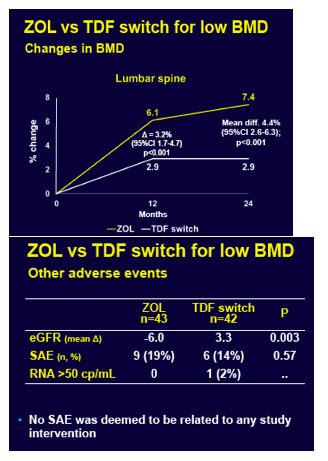
Through 12 months lumbar spine BMD rose 6.1% in the zoledronic acid group and 2.9% in the TDF-switch group, a significant difference (3.2% difference, 95% confidence interval [CI] 1.7 to 4.7, P < 0.001). At 24 months lumbar spine BMD climbed 7.4% with zoledronic acid and 2.9% with the TDF switch (4.4% difference, 95% CI 2.6 to 6.3, P < 0.001). At 24 months the zoledronic acid group also gained significantly more BMD at the femoral neck (4.1% versus 2.1%, P = 0.03) and total hip (4.6% versus 2.6%, P = 0.009).
There was one fracture (2%) in the zoledronic acid group and 7 fractures (17%) in 4 people in the TDF-switch group (P = 0.03), but the trial was not powered to assess relative strategy impact on fractures. Estimated glomerular filtration rate dropped with zoledronic acid (-6.0) and rose with a TDF switch (+3.3), a significant difference (P = 0.003). There were 9 serious adverse events (19%) with zoledronic acid and 6 (14%) with a TDF switch, but that difference lacked statistical significance (P = 0.57) and none of these problems were attributed to a study intervention.
The researchers stressed that "much larger and longer studies [are] required to determine impact on fracture outcomes," adding that the clinical significance of these findings will probably depend on underlying fracture risk.
Reference
1. Hoy J, Richardson R, Ebeling PR, et al. Zoledronic acid is superior to TDF-switching for increasing bone mineral density in HIV-infected adults with osteopenia: a randomised trial. 9th IAS Conference on HIV Science (IAS 2017), July 23-26, 2017, Paris. Abstract WEAB0106LB.




|
| |
|
 |
 |
|
|