 |
 |
 |
| |
Three-Part Strategy Boosts Extragenital STI Screening 23% in HIV Clinic
|
| |
| |
Download the PDF here
IDWeek2017/IDSA, October 4-8, 2017, San Diego
Mark Mascolini
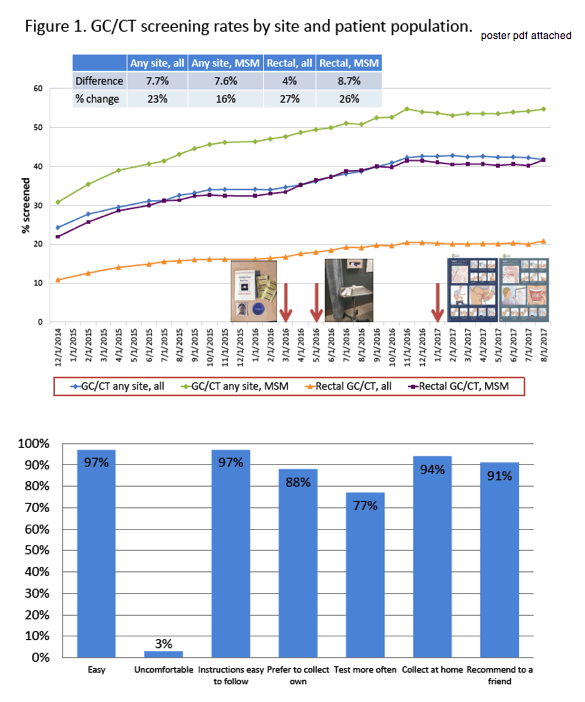
A three-phase strategy raised sexually transmitted infection (STI) screening rates 23% in people with HIV over the course of a 31-month study at the University of North Carolina (UNC) clinic [1]. The rectal screening rate rose 27% over the study period.
At many centers screening rates for extragenital gonococcal and chlamydial STIs remain low in people with HIV. Urogenital screening is more common. But research shows that urogenital testing alone misses 70% to 80% of extragenital infections in men who have sex with men (MSM) and 15% to 30% of such infections in women [2,3]. UNC investigators noted several barriers to STI screening--limited provider time, discomfort with a sexual exam and history taking, and patient reluctance to be tested.
To address these obstacles, the UNC Infectious Diseases clinic launched a three-phase strategy aiming to increase extragenital gonorrhea/chlamydia screening by 10%. (1) In March 2016, nurses began counseling patients on STI risk. (2) In May 2016, STI screening supplies were regularly displayed on clinic Mayo stands. (3) In January 2017, patients began collecting their own screening samples and completed a survey on acceptability of extragenital self-screening. The researchers tracked screening rates from December 1, 2014 through August 1, 2007.
Throughout the study period, overall gonorrhea/chlamydia screening at any site rose 23%, screening at any site for MSM rose 16%, overall rectal screening rose 27%, and rectal screening among MSM rose 26%.
Forty people completed the self-screening acceptability survey. Almost everyone, 97%, agreed self-screening was easy, and 97% agreed instructions were easy to follow. While 94% of respondents said they would collect samples at home, 88% preferred to collect their own sample rather than have a health worker do it; 91% of respondents said they would recommend self-screening to a friend, and 77% believed self-screening would permit more frequent screening. Only 3% felt uncomfortable collecting their own samples.
Steady improvements in screening rates showed that self-screening sustained gains in rates that followed the first two phases of the strategy. The researchers concluded that "express clinic-based self-collection or home-based self-collection may be acceptable interventions to further increase [gonorrhea/chlamydia] screening rates."
References
1. Menza T, Anita Holt A, Hankins T, et al. Improving extra-genital GC/CT screening among HIV-positive patients at the University of North Carolina infectious diseases clinic. IDWeek2017/IDSA. October 4-8, 2017. San Diego. Abstract 2261.
2. Clinic-based testing for rectal and pharyngeal Neisseria gonorrhoeae and Chlamydia trachomatis infections by community-based organizations--five cities, United States, 2007. MMWR Morb Mortal Wkly Rep. 2009;58:716-719.
3. Trebach JD, Chaulk CP, Page KR, et al. Neisseria gonorrhoeae and Chlamydia trachomatis among women reporting extragenital exposures. Sex Transm Dis. 2015;42:233-239.

|
| |
|
 |
 |
|
|