 |
 |
 |
| |
Dolutegravir Cmax Higher in Elderly, Tied to Shorter Sleep Duration
|
| |
| |
18th International Workshop on Clinical Pharmacology of Antiviral Therapy, June 14-17, 2017, Chicago
Mark Mascolini
Dolutegravir maximum concentration (Cmax) proved significantly higher in HIV-positive people 60 or older than in people younger than 51 taking the integrase inhibitor [1], Overall dolutegravir exposure (area under the concentration-time curve, AUC) did not differ between the two age groups, but this 56-person British study linked higher Cmax and higher AUC to shorter sleep duration in the 60-plus group.
The researchers noted that prior study yielded several findings on dolutegravir-related central nervous system (CNS) side effects: (1) They are less frequent with dolutegravir than efavirenz. (2) They affected up to 5% of people enrolled in trials, and dropout rates were low. (3) They led to dropout rates up to 5% in observational cohorts. Because trials usually enroll few elderly patients, the UK team conducted this comparative study to learn whether dolutegravir exposure at 50 mg once daily correlates with sleep problems in older versus younger individuals.
The analysis involved people 60 years old or older switching from a suppressive antiretroviral regimen to coformulated dolutegravir/abacavir/lamivudine (Triumeq). The study protocol stipulated that 70% of the older population had to be at least 65 years old and everyone at least 60 years old.
At the switch to dolutegravir on day 1 and again on day 28, participants had intensive pharmacokinetic sampling over 24 hours while fasted. The researchers compared results with findings in people younger than 50 enrolled in the SPRING-1 trial of dolutegravir [2], who also had intensive sampling while fasted. The 60-plus group completed three sleep questionnaires: the Pittsburgh Sleep Quality Index (PSQI), the Functional Outcomes of Sleep Questionnaire (FOSQ), and the Insomnia Severity Index (ISI) when they switched to Triumeq and 28 days later.
Among the 40 people in the older group, 1 was a woman and median age stood at 65.5 (range 60 to 78). Median age in the 16 younger people measured 37 (range 22 to 50), and 1 was a woman. Seventeen people in the older group (43%) were switching from an efavirenz regimen. No grade 3 or 4 side effects arose after the switch and everyone maintained virologic control 4 weeks after the switch.
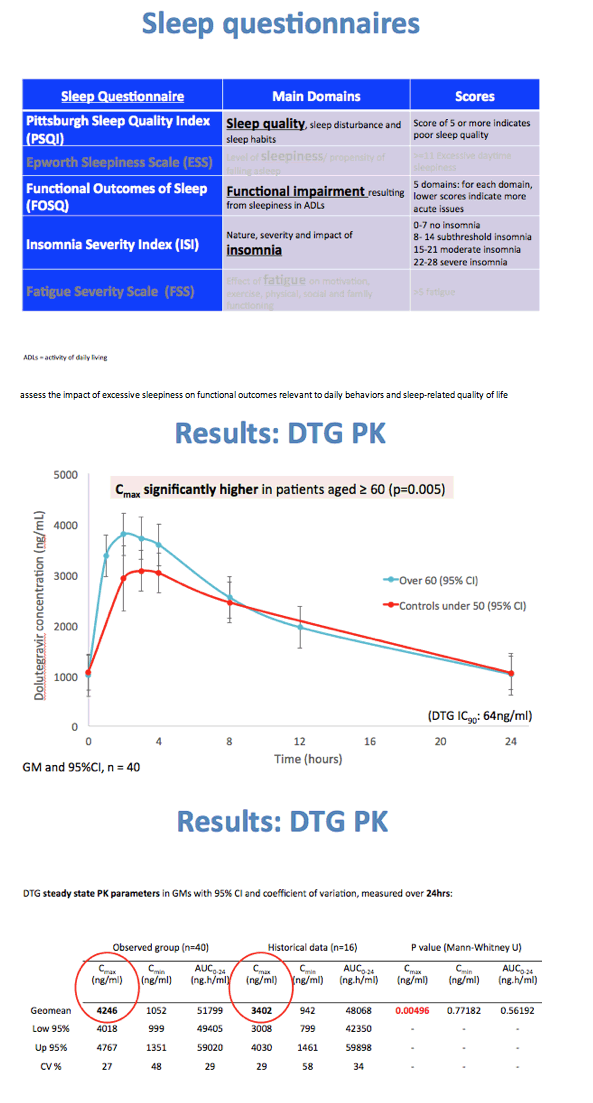
Dolutegravir AUC and trough concentration (Ctrough) did not differ significantly between the older group and younger controls (geometric mean [GM] 51,799 versus 48,068 ng*h/mL, P = 0.56, for AUC; GM 1052 versus 942 ng/mL, P = 0.77, for Ctrough). But dolutegravir Cmax was significantly higher in older people than in younger controls (GM 4246 versus 3402 ng/mL) (P = 0.00496).
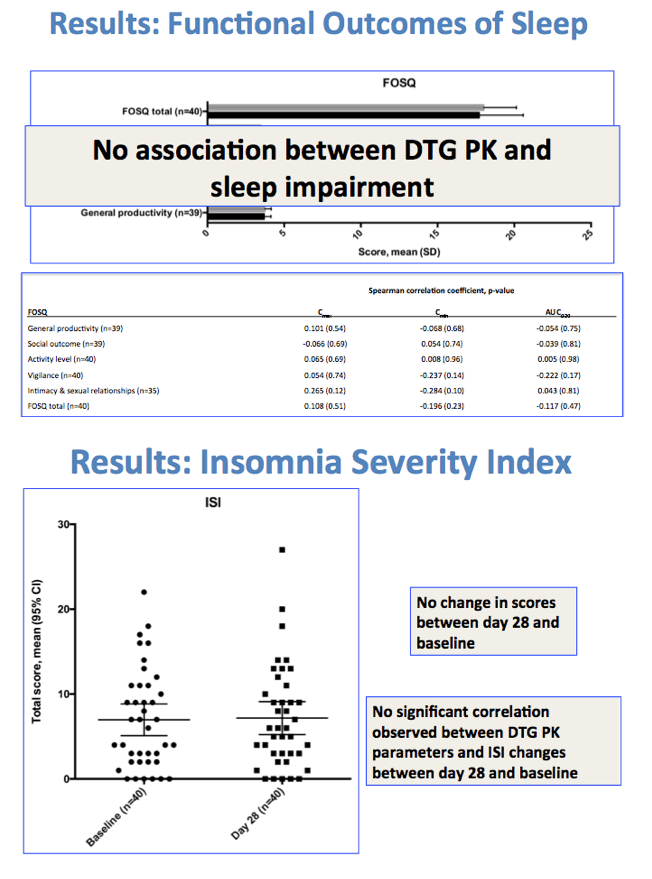
Twenty-eight days after the switch to Triumeq, global scores and individual domains on the PSQI did not differ significantly from preswitch measures in people 60 or older. But higher dolutegravir Cmax and AUC were associated with shorter sleep duration on the PSQI (P = 0.05 and P = 0.03).
On the FOSQ global and individual sleep scores in these older people did not change significantly between day 1 and day 28. And dolutegravir pharmacokinetics did not correlate with sleep impairment on these tests. Likewise, the ISI did not change through 28 days of dolutegravir, and dolutegravir exposure did not predict insomnia severity.
The researchers suggested that higher Cmax in the older group "may represent differences in drug absorption" in older people. They added that the link between higher dolutegravir exposure and shorter sleep duration needs further study.
References
1. Elliot E, Wang X, Simmons B, et al. Relationship between dolutegravir plasma exposure, quality of sleep and its functional outcome in patients living with HIV over the age of 60 years. 18th International Workshop on Clinical Pharmacology of Antiviral Therapy, June 14-17, 2017, Chicago. Abstract O_08.
2. Stellbrink HJ, Reynes J, Lazzarin A, et al. Dolutegravir in antiretroviral-naive adults with HIV-1: 96-week results from a randomized dose-ranging study. AIDS. 2013;27:1771-1778.

Emilie Elliot1,2, Xinzhu Wang3, Bryony Simmons4, Alison Jenkin1, Nicole Pagani1, Susanna Wong4, Jamie Vera5, Robert Miller6, Myra McClure3, Marta Boffito1,3
1St Stephen's Centre, Chelsea and Westminster Hospital, London, UK, 2University of Liverpool, Liverpool UK, 3Department of Medicine, Imperial College, London, UK, 4Faculty of Medicine, Imperial College, London, UK, 5Brighton and Sussex Medical School, Brighton, UK, 6Institute of Global Health, University College London, UK


|
| |
|
 |
 |
|
|