 |
 |
 |
| |
Effect of cobicistat on tenofovir disoproxil fumarate
(TDF) durability: is it time to rethink at TAF trials?
|
| |
| |
Does TDF Need Dose Reduction When Given With Boosting Agent?
18th International Workshop on Clinical Pharmacology of Antiviral Therapy, June 14-17, 2017, Chicago
Mark Mascolini
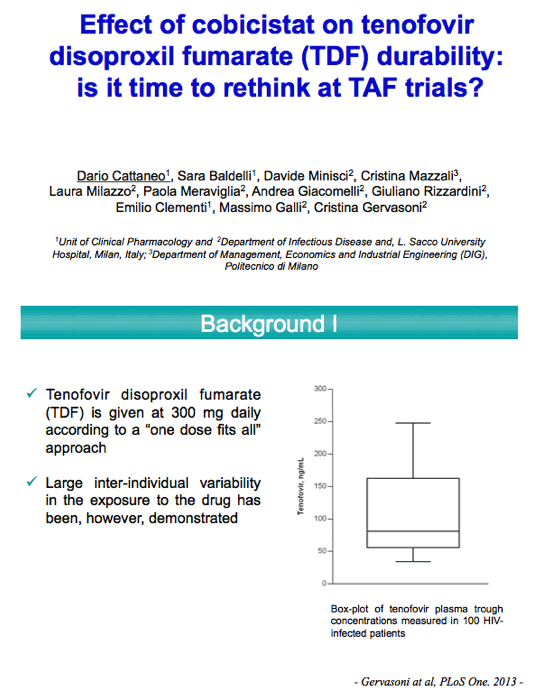
Giving cobicistat with tenofovir disoproxil fumarate (TDF) raised tenofovir concentrations more than ritonavir boosting and limited TDF durability in an analysis of more than 500 patients in Italy [1]. Researchers from Milan's Luigi Sacco University Hospital suggested that TDF may need a dose reduction when given with a boosting agent. They also believe their findings may raise question about results of trials comparing TDF with tenofovir alafenamide (TAF).
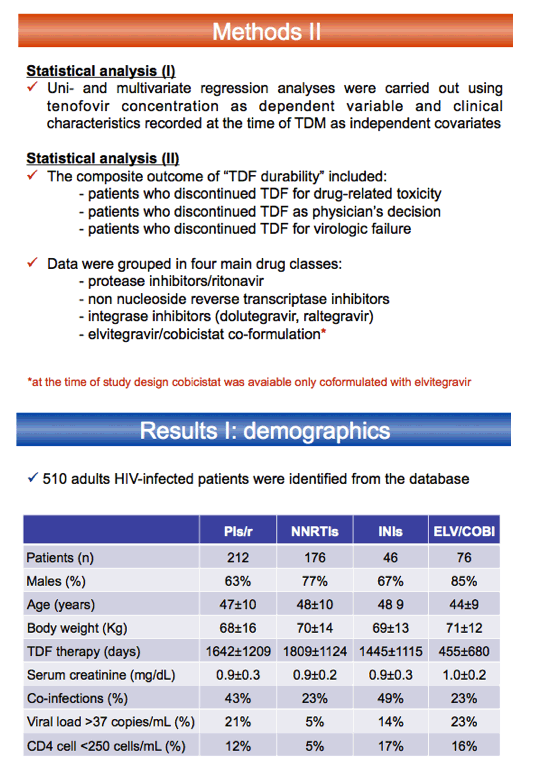
The Luigi Sacco team noted that the TAF dose drops from 25 to 10 mg daily when given in a regimen including cobicistat or ritonavir. But no such reduction has been proposed for TDF in boosted regimens. The researchers compared tenofovir trough concentrations and discontinuations when patients took TDF in boosted or nonboosted regimens. The study included adults taking a TDF regimen for at least 3 months who had at least one tenofovir trough level measured. It excluded people with liver impairment (Child-Pugh class B or C) or with creatinine clearance below 80 mL/min before starting TDF.
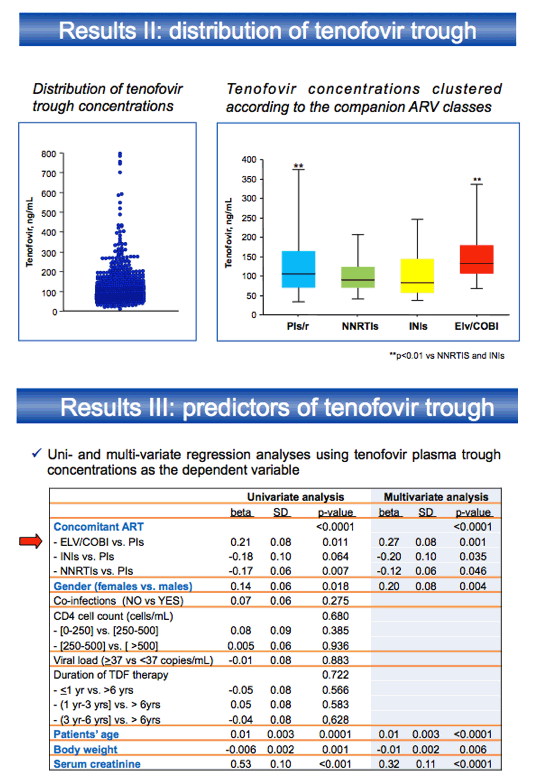
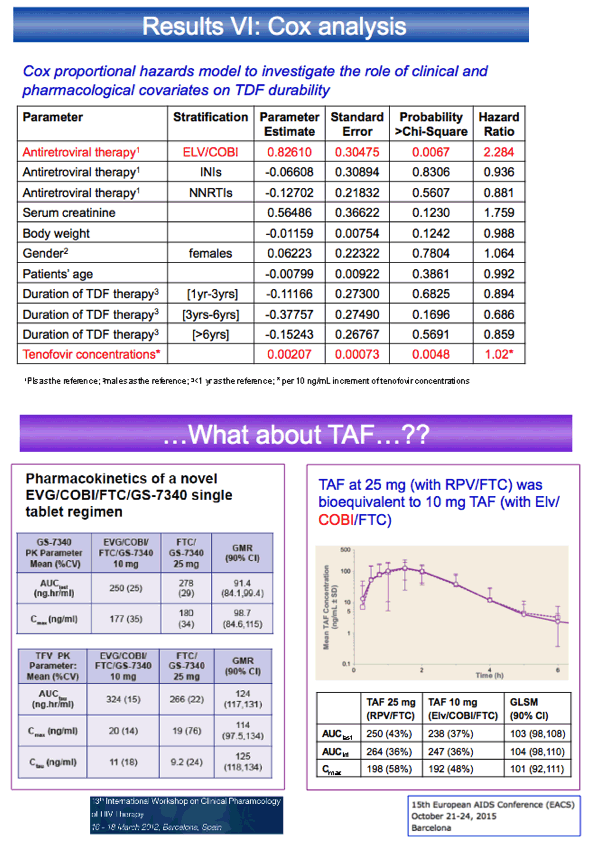
This retrospective single-center analysis included 212 people taking TDF with ritonavir-boosted protease inhibitors (PIs), 176 with nonnucleosides, 46 with dolutegravir or raltegravir, and 76 with elvitegravir/cobicistat. Tenofovir troughs proved significantly higher with elvitegravir/cobicistat or with boosted PIs than with nonnucleosides or integrase inhibitors (P < 0.01). Multivariate regression analysis with tenofovir trough concentration as the dependent variable indicated significantly higher tenofovir troughs with elvitegravir/cobicistat than with ritonavir-boosted PIs (beta 0.27, P = 0.001). In the same analysis, women had higher tenofovir troughs than men (beta 0.20, P = 0.004). Older age and lower weight were also associated with higher tenofovir troughs.
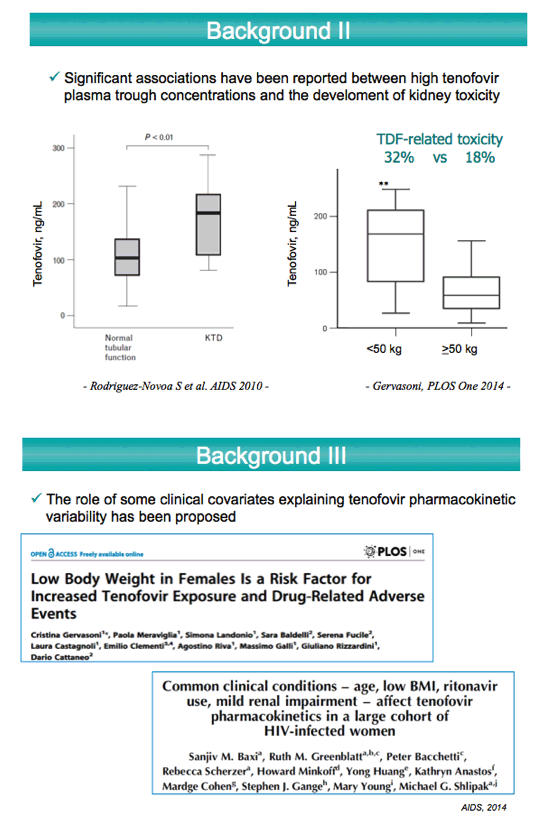
A separate exploratory analysis of tenofovir troughs with individual antiretroviral components found the highest tenofovir exposure with ritonavir- boosted lopinavir and cobicistat-boosted elvitegravir, followed by boosted atazanavir. Boosted amprenavir or darunavir had the lowest tenofovir troughs.
Through an average 1149 days of follow-up, the probability of stopping TDF in the first year was much higher with elvitegravir/cobicistat (43.6%) than with boosted PIs (15.6%), nonnucleosides (13.1%), or integrase inhibitors (10.6%) (P = 0.0002). Cox regression analysis linked two baseline factors to stopping TDF--taking elvitegravir/cobicistat (hazard ratio 2.284) and higher tenofovir trough (hazard ratio 1.02 per 10 ng/mL higher).
The researchers concluded that, despite shorter follow-up of people taking elvitegravir/cobicistat, they stopped TDF 3 times more often than people taking other TDF regimens in the first year of therapy. They believe this finding adds to evidence supporting a TDF dose reduction with boosting agents. The authors suggested that "lack of proper dose adjustment for TDF when given with cobicistat (or ritonavir) might have biased the safety results between TAF and TDF during registration trials."
Reference
1. Cattaneo D, Baldelli S, Minisci D, et al. Effects of cobicistat on tenofovir durability: is it time to rethink TAF trials? 18th International Workshop on Clinical Pharmacology of Antiviral Therapy, June 14-17, 2017, Chicago. Abstract O_02.
-----------------------------------
Effect of cobicistat on tenofovir disoproxil fumarate (TDF) durability: is it time to rethink at TAF trials?
Reported by Jules Levin
18th International Workshop on Clinical Pharmacology of Antiviral Therapy, June 14-17, 2017, Chicago
Dario Cattaneo1, Sara Baldelli1, Davide Minisci2, Cristina Mazzali3,
Laura Milazzo2, Paola Meraviglia2, Andrea Giacomelli2, Giuliano Rizzardini2, Emilio Clementi1, Massimo Galli2, Cristina Gervasoni2
1Unit of Clinical Pharmacology and 2Department of Infectious Disease and, L. Sacco University Hospital, Milan, Italy; 3Department of Management, Economics and Industrial Engineering (DIG), Politecnico di Milano





|
| |
|
 |
 |
|
|