 |
 |
 |
| |
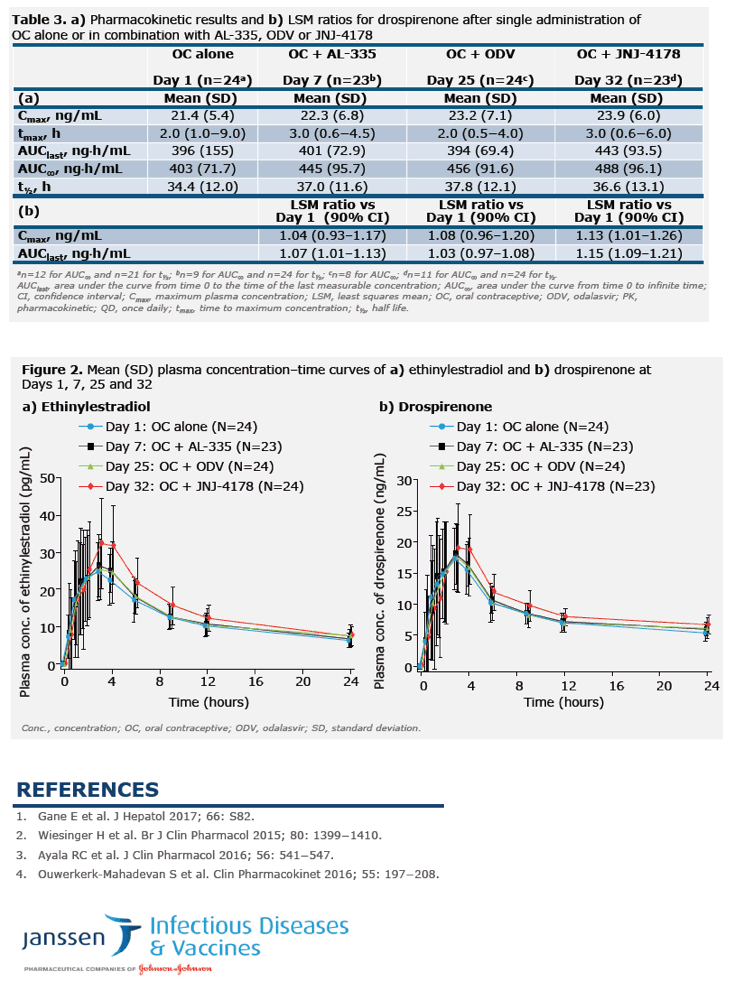
Lack of clinically significant pharmacokinetic interaction between the 3-DAA combination of AL-335, odalasvir and simeprevir for the treatment of chronic HCV infection and an oral contraceptive containing ethinylestradiol and drospirenone
|
| |
| |
Reported by Jules Levin
18th International Workshop on Clinical Pharmacology of Antiviral Therapy, 14-16 June 2017, Chicago, IL,USA
Ouwerkerk-Mahadevan S1, Gamil M1, van Hemelryck S1, Hillewaert V1, van Nimwegen M2, Kakuda T3, Biermer M1
1Janssen Research & Development, Janssen Pharmaceutica NV, Beerse, Belgium; 2Janssen Biologics BV, Leiden, Netherlands; 3Alios BioPharma, Inc., part of the Janssen Pharmaceutical Companies, South San Francisco, USA
Short Duration Treatment with AL-335 and Odalasvir, with or without Simeprevir, in Treatment Naïve Patients with Hepatitis C Infection with or without Cirrhosis - (04/24/17)
AL-335, A ONCE-DAILY PANGENOTYPIC NUCLEOTIDE HCV POLYMERASE INHIBITOR, DEMONSTRATES POTENT ANTIVIRAL ACTIVITY OVER 7 DAYS IN TREATMENT-NAïVE GENOTYPE 1-4 PATIENTS - (04/18/16)
PAN-GENOTYPIC EVALUATION OF AL-335, A CLINICAL STAGE URIDINE ANALOG INHIBITOR OF HEPATITIS C VIRUS POLYMERASE - (04/18/16)
Preclinical Characterization of AL-335, a Potent Uridine Based Nucleoside Polymerase Inhibitor for the Treatment of Chronic Hepatitis C - (05/05/15)


|
| |
|
 |
 |
|
|