 |
 |
 |
| |
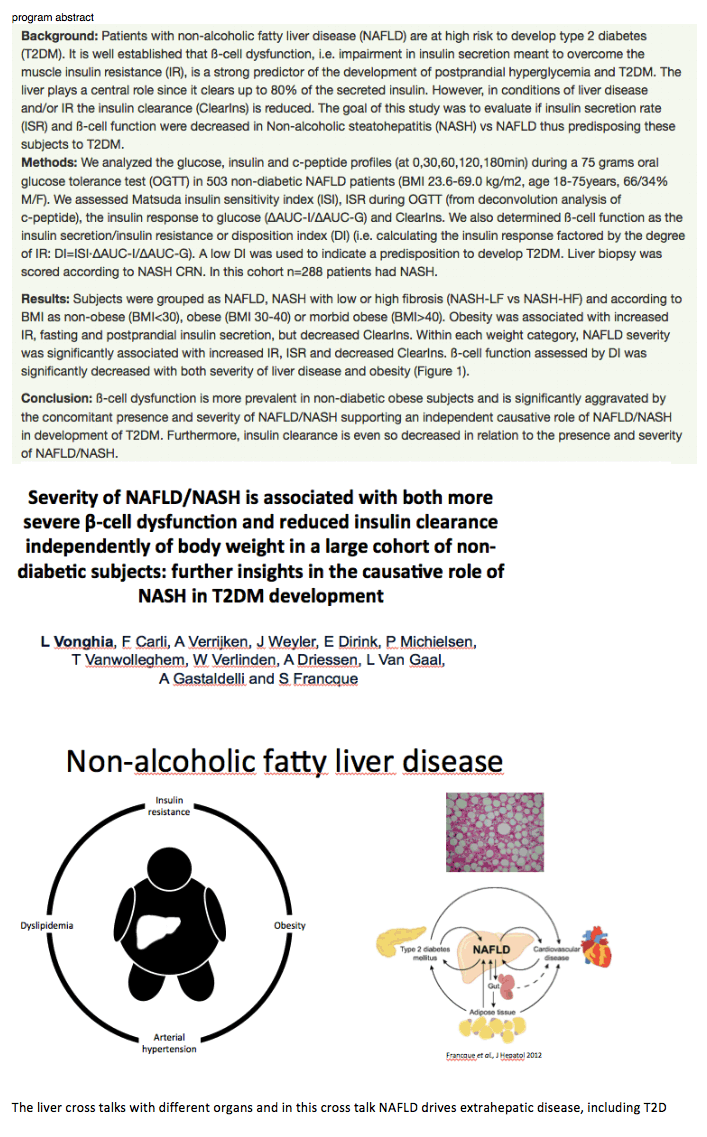
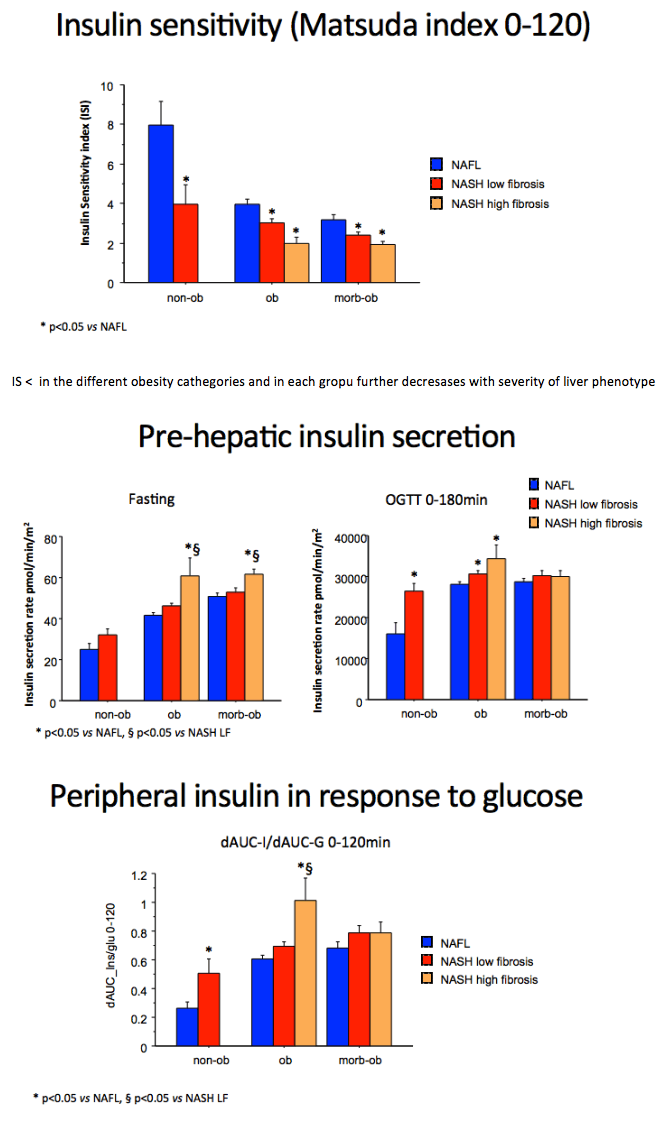
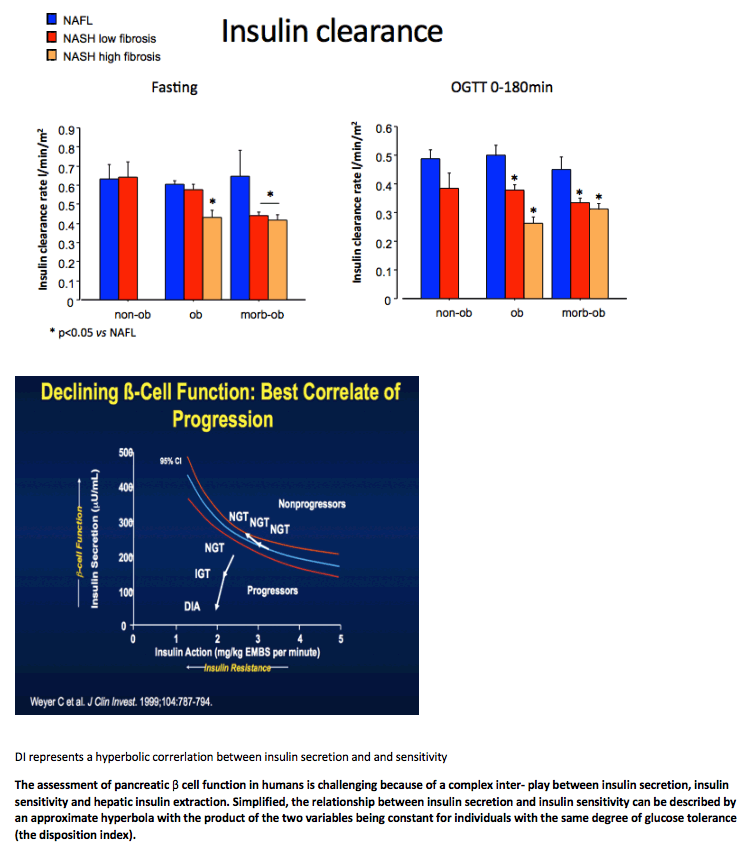
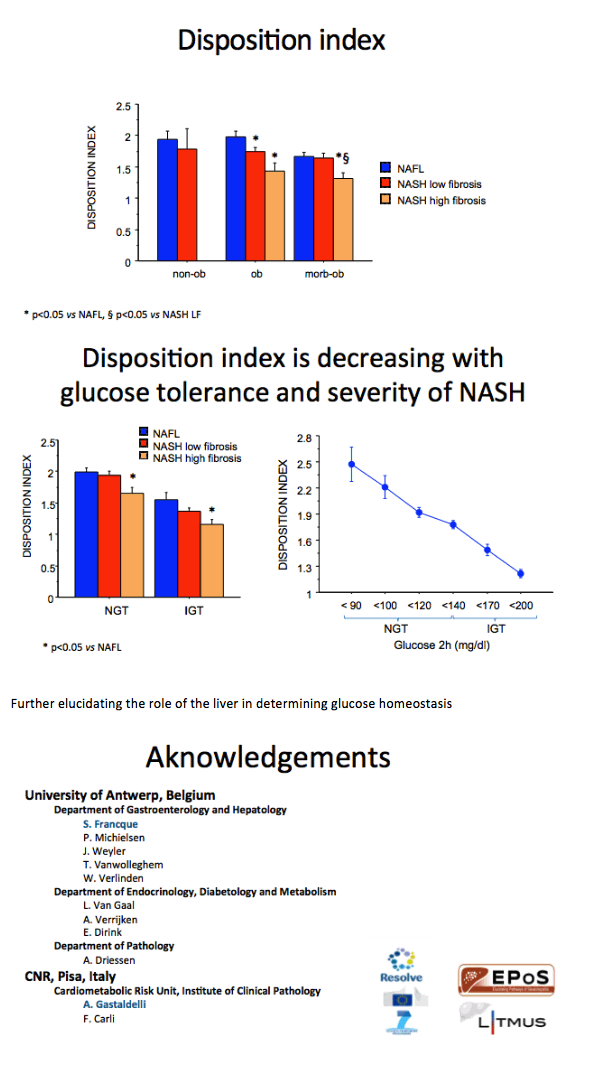
Severity of NAFLD/NASH is associated with both more severe β-cell dysfunction and reduced insulin clearance independently of body weight in a large cohort of non-diabetic subjects: further insights in the causative role of NASH in T2DM development
|
| |
| |
Reported by Jules Levin
AASLD 1028 Nov 9-13 SF
L Vonghia, F Carli, A Verrijken, J Weyler, E Dirink, P Michielsen,
T Vanwolleghem, W Verlinden, A Driessen, L Van Gaal,
A Gastaldelli and S Francque

The primary function of a beta cell is to produce and release insulin. Insulin is a hormone that brings about effects which reduce blood glucose concentration. Beta cells can respond quickly to spikes in blood glucose concentrations by secreting some of their stored insulin while simultaneously producing more
https://en.wikipedia.org/wiki/Beta_cell
In contrast to macrophages and T cells, little is known about the role of B cells in the development of insulin resistance despite evidence that such cells are recruited to adipose tissue shortly after initiation of a high fat diet15 and their activation is increased in patients with T2D16. Here we demonstrate that B cells and IgG are important pathogenic effectors in the development of obesity-associated insulin resistance and glucose intolerance, but not of excess weight gain, in DIO mice. Manipulation of B cells, antibodies or their receptors may yield promising new therapies for the management of insulin resistance and its associated co-morbidities.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3270885/
In pancreatic β cells, mitochondria play a central role in coupling glucose metabolism to insulin exocytosis, thereby ensuring strict control of glucose-stimulated insulin secretion. Defects in mitochondrial function impair this metabolic coupling, and ultimately promote apoptosis and β cell death. Various factors have been identified that may contribute to mitochondrial dysfunction. In this review we address the emerging concept of complex links between these factors. We also discuss the role of the mitochondrial genome and mutations associated with diabetes, the effect of oxidative stress and reactive oxygen species, the sensitivity of mitochondria to lipotoxicity, and the adaptive dynamics of mitochondrial morphology. Better comprehension of the molecular mechanisms contributing to mitochondrial dysfunction will help drive the development of effective therapeutic approaches.
https://www.cell.com/trends/endocrinology-metabolism/pdf/S1043-2760(12)00094-X.pdf
program abstract
|
| |
|
 |
 |
|
|