 |
 |
 |
| |
IMPACT OF METHOTREXATE ON ARTERIAL
INFLAMMATION IN PERSONS WITH TREATED HIV
|
| |
| |
Reported by Jules Levin
CROI 2018 March 4-7 Boston MA
Ahmed Tawakol1 , Heather Ribaudo2 , Amorina E. Ishai1 , Ashley McKhann2 , Daniel H. Solomon3 , Zahi A. Fayad4 , Steven G. Deeks5 ,
Michael M. Lederman6 , Steven K. Grinspoon1 , James H. Stein7 , Paul M. Ridker3 , Judith S. Currier8 , and Priscilla Hsue5
ORAL - WEBCAST: CROI: Impact of Low Dose Methotrexate on Immune Activation and Endothelial Function in Treated HIV - (03/07/18)

Program Abstract:
Arterial inflammation, a key driver of atherosclerotic cardiovascular disease (ASCVD), is elevated in individuals living with HIV and is hypothesized to underpin the increased incidence of ASCVD in this population. Low-dose methotrexate (LDMTX) is an anti-inflammatory drug that is associated with lower risk of ASCVD in individuals with rheumatoid arthritis. We evaluated the impact of LDMTX on arterial inflammation in treated HIV infection.
We conducted a randomized placebo-controlled trial in ART-treated HIV-infected individuals age ≥40 years with or at increased risk for ASCVD and with CD4+ T-cells >400 cells/mm3. Participants received weekly LDMTX or placebo for 24 weeks; both groups received 1 mg folate daily. Arterial inflammation was assessed using FDG PET/CT imaging at 0 and 24 weeks, and measured as standardized uptake values (SUV). SUV was additionally corrected for background activity, producing target-to-background-ratios (TBR). The primary endpoint was ΔTBR. Additionally, a pre-specified secondary endpoint was ΔSUV (which is less susceptible to variation and may associate better with histological evidence of inflammation). Intent to treat group comparisons used stratified Wilcoxon tests.
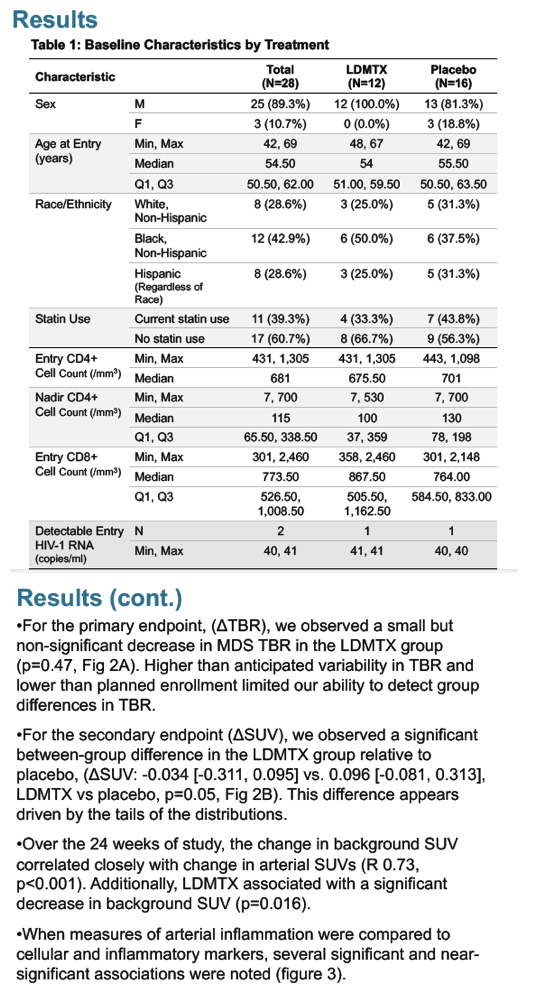
A total of 28 individuals, from 8 sites, provided evaluable image sets. Participants had a median [IQR] age of 55 [51, 62] years, 25 were male, and entry CD4+ T cells of 681 [581, 871] /mm3. For the primary endpoint, (ΔTBR), we observed a small but non-significant decrease in TBR in the LDMTX group ((ΔTBR: -0.126 [-0.41, 0.258]) relative to placebo (0.026 [-0.176, 0.438], p=0.47, Fig 1A). Higher than anticipated variability in TBR and lower than planned enrollment limited our ability to detect group differences in TBR. For the secondary endpoint (ΔSUV), we observed a significant between-group difference in the LDMTX group relative to placebo (ΔSUV: -0.034 [-0.311, 0.095] vs. 0.096 [-0.081, 0.313], LDMTX vs placebo, p=0.05, Fig 1B). This represented an approx. 2% decrease in SUV (from baseline 2.01) in the LDMTX, and a 5% increase in SUV (from baseline of 1.82) in the placebo group.
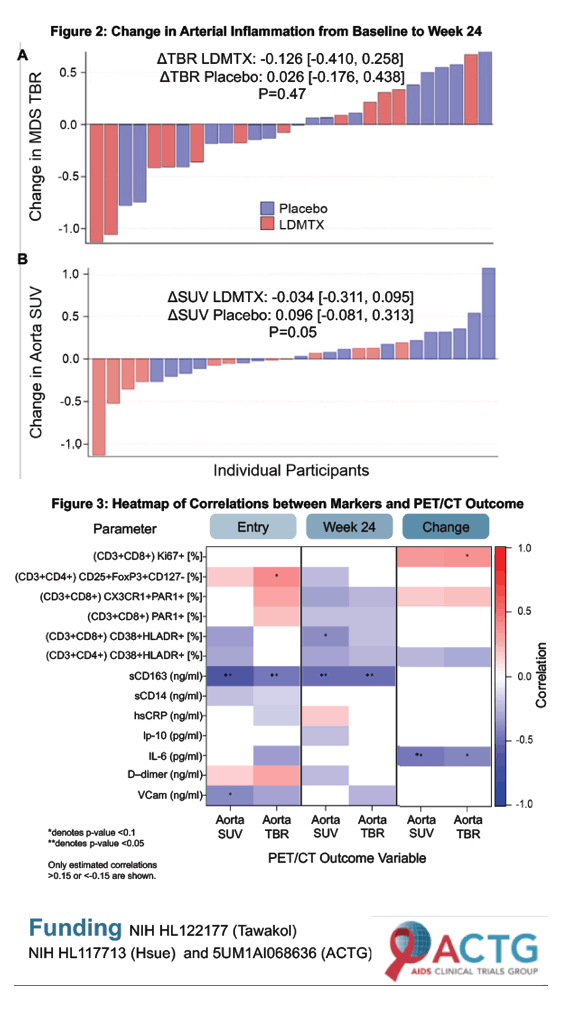
LDMTX may reduce arterial inflammation in HIV-infected adults with or at risk for ASCVD, at least as measured by ΔSUV. This finding may explain the apparent beneficial impact of LDMTX on ASCVD risk in chronic inflammatory diseases. The potential effect of LDMTX on arterial inflammation in HIV should be studied in a larger cohort.

|
| |
|
 |
 |
|
|