 |
 |
 |
| |
SHORT-TERM ART INTERRUPTION HAS LITTLE EFFECT ON LEVELS OF INTEGRATED PROVIRAL DNA
|
| |
| |
from Jules: some would suggest - As it appears that more than 1 log reduction in a reservoir will be needed to allow control/remission, I do not see any need for ATI until a goal something like that is achieved.
Maybe affects on reservoirs depend on the patient population being studied - so those with a low CD4 nadir or higher viral load prior to initiating HAART have a more dysfunctional immune system that responds differently to ATI/interruption.
Reported by Jules Levin
CROI 2018 March 4-7 Boston MA
Zachary Strongin1, Radwa Sharaf1, Jake VanBelzen2, Jeffrey Jacobson3, Elizabeth Connick4, Paul Volberding5, Daniel Skiest6, Rajesh T. Gandhi7, Daniel R. Kuritzkes1, Una O'Doherty2, Jonathan Z. Li1
1Brigham and Women's Hospital, Boston, MA, USA,2University of Pennsylvania, Philadelphia, PA, USA,3Temple University, Philadelphia, PA, USA,4University of Arizona, Tucson, AZ, USA,5University of California San Francisco, San Francisco, CA, USA,6Baystate Health, Springfield, MA, USA,7Massachusetts General Hospital, Boston, MA, USA

Program Abstract:
HIV analytic treatment interruption (ATI) represents the ultimate test to assess strategies aimed at achieving sustained ART-free remission, but questions remain about the effects of ATI on the viral reservoir. We validated a size-selection-based assay for quantifying integrated proviral HIV DNA as a measure of HIV reservoir size and assessed the impact of ATI on reservoir size after ART resumption.
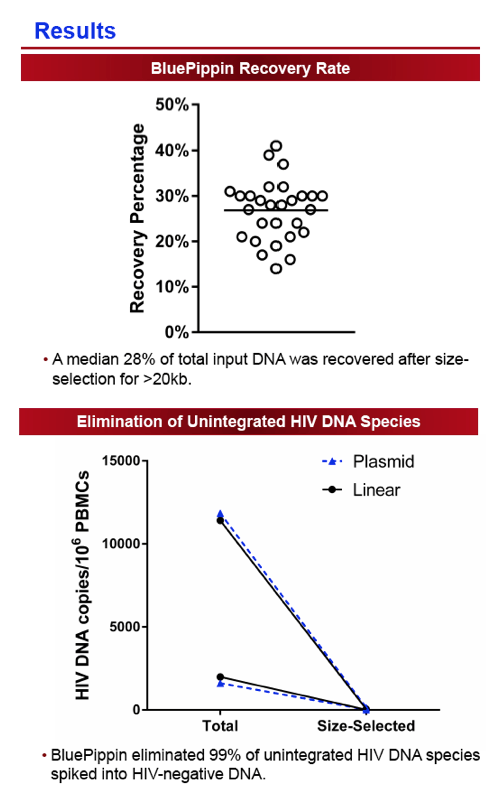
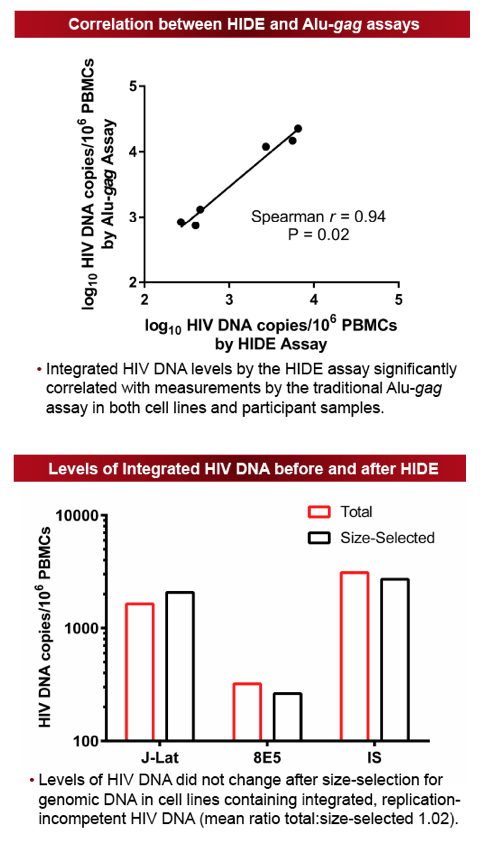
Cryopreserved PBMCs were obtained for 12 participants from previously completed ACTG ATI trials with available samples pre-ATI (T1) and ≥24 weeks after ART resumption (T3). Four participants also had samples available during the ATI (T2). Genomic DNA was separated from episomal DNA by size-selection using the BluePippin pulsed-field gel electrophoresis system and proviral DNA levels quantified by qPCR. Assay validation was first performed by spiking non-integrated HIV DNA into genomic DNA extracted from HIV-uninfected individuals to confirm successful elimination of the episomal HIV fragments. In addition, HIV-infected cell lines and participant samples were used to compare results to the standard Alu-[i]gag[/i] assay.
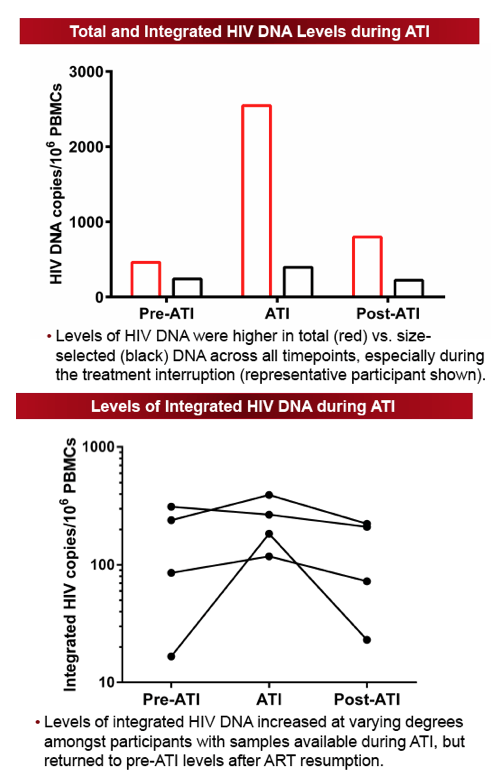
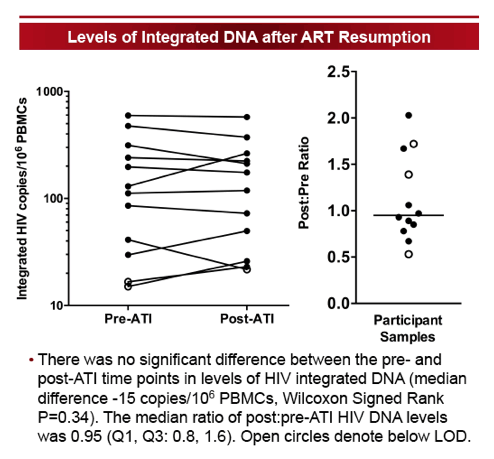
The size-selection-based proviral DNA assay eliminated 99% of non-integrated HIV DNA species and correlated strongly with the established Alu-gag assay (Spearman r=0.94, P=0.02). The median ATI duration was 12 weeks (range 6-67 weeks). For the majority of individuals, integrated DNA levels increased during ATI (median +94 HIV DNA copies/106 PBMCs) and subsequently declined after ≥24 weeks of ART (median -109 HIV DNA copies/106 PBMCs). There was no significant difference in levels of HIV integrated DNA between the pre- and post-ATI time points. The median ratio of post:pre-ATI (T3:T1) HIV DNA levels was 0.95 (Q1, Q3: 0.8, 1.6). In those with higher HIV DNA levels post-ATI, the increase in proviral DNA levels was generally small, with a median change of 10.9 HIV DNA copies/106 PBMCs (Q1, Q3: 6.9, 19.9 HIV DNA copies/106 PBMCs). There was no significant correlation between duration of ATI and the ratio of post:pre-ATI reservoir size.
We validated a size-selection-based proviral DNA assay that is less sample- and labor-intensive than the currently used assays. Levels of integrated HIV DNA showed minimal change after short-term ATI followed by ART resumption, suggesting that short-term ATI can be conducted without a significant impact on levels of integrated proviral DNA.

Program Abstract:
HIV analytic treatment interruption (ATI) represents the ultimate test to assess strategies aimed at achieving sustained ART-free remission, but questions remain about the effects of ATI on the viral reservoir. We validated a size-selection-based assay for quantifying integrated proviral HIV DNA as a measure of HIV reservoir size and assessed the impact of ATI on reservoir size after ART resumption.
Cryopreserved PBMCs were obtained for 12 participants from previously completed ACTG ATI trials with available samples pre-ATI (T1) and ≥24 weeks after ART resumption (T3). Four participants also had samples available during the ATI (T2). Genomic DNA was separated from episomal DNA by size-selection using the BluePippin pulsed-field gel electrophoresis system and proviral DNA levels quantified by qPCR. Assay validation was first performed by spiking non-integrated HIV DNA into genomic DNA extracted from HIV-uninfected individuals to confirm successful elimination of the episomal HIV fragments. In addition, HIV-infected cell lines and participant samples were used to compare results to the standard Alu-[i]gag[/i] assay.
The size-selection-based proviral DNA assay eliminated 99% of non-integrated HIV DNA species and correlated strongly with the established Alu-gag assay (Spearman r=0.94, P=0.02). The median ATI duration was 12 weeks (range 6-67 weeks). For the majority of individuals, integrated DNA levels increased during ATI (median +94 HIV DNA copies/106 PBMCs) and subsequently declined after ≥24 weeks of ART (median -109 HIV DNA copies/106 PBMCs). There was no significant difference in levels of HIV integrated DNA between the pre- and post-ATI time points. The median ratio of post:pre-ATI (T3:T1) HIV DNA levels was 0.95 (Q1, Q3: 0.8, 1.6). In those with higher HIV DNA levels post-ATI, the increase in proviral DNA levels was generally small, with a median change of 10.9 HIV DNA copies/106 PBMCs (Q1, Q3: 6.9, 19.9 HIV DNA copies/106 PBMCs). There was no significant correlation between duration of ATI and the ratio of post:pre-ATI reservoir size.
We validated a size-selection-based proviral DNA assay that is less sample- and labor-intensive than the currently used assays. Levels of integrated HIV DNA showed minimal change after short-term ATI followed by ART resumption, suggesting that short-term ATI can be conducted without a significant impact on levels of integrated proviral DNA.

|
| |
|
 |
 |
|
|