 |
 |
 |
| |
Efficacy of human papillomavirus-based screen-and-treat for cervical cancer prevention among HIV-infected women - Cervical Outcomes Similar With Immediate Cryotherapy and Standard Cytology in HIV+ Women
|
| |
| |
Download the PDF here
Download the PDF here
25th Conference on Retroviruses and Opportunistic Infections (CROI), March 4-7, 2018, Boston
Mark Mascolini
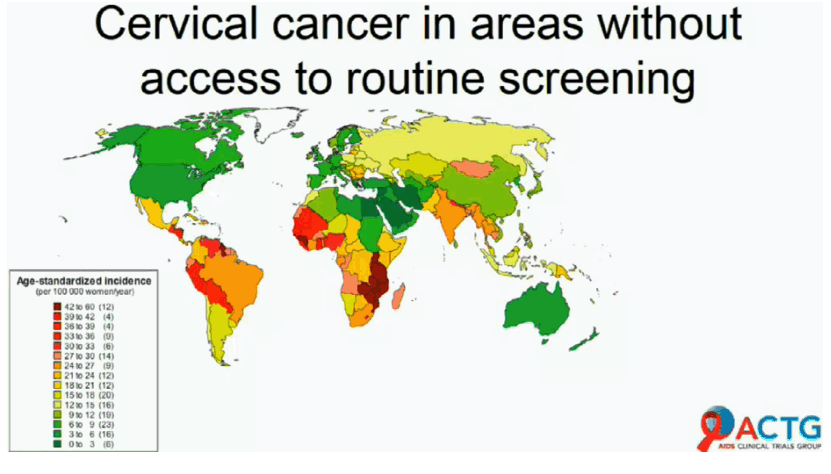
A high-risk HPV (hrHPV) test-and-treat strategy did no better than standard cytology-based screening in preventing cervical intraepithelial neoplasia grade 2 or worse (CIN2+) in women with HIV, according to results of the 288-woman AIDS Clinical Trials Group (ACTG) A5282 study [1]. But ACTG investigators believe their findings show the test-and-treat approach is feasible for lower-resource countries.
Standard cytology-based cervical cancer screening involves a 3-step process: (1) PAP smear, followed by (2) colposcopy and biopsy if the PAP smear is abnormal, and (3) treatment of cervical intraepithelial neoplasia grade 2 or 3 (the precursor to cervical cancer) with loop electrosurgical excision (LEEP) or other treatments. But that approach remains too resource-intensive in many regions affected by HIV. An alternative screening approach is to test for hrHPV and immediately perform cryotherapy in hrHPV-positive women--the test-and-treat strategy. In a large South African trial, women randomized to hrHPV test-and-treat had the lowest CIN2/3+ rate in follow-up, including a subgroup with HIV [2].
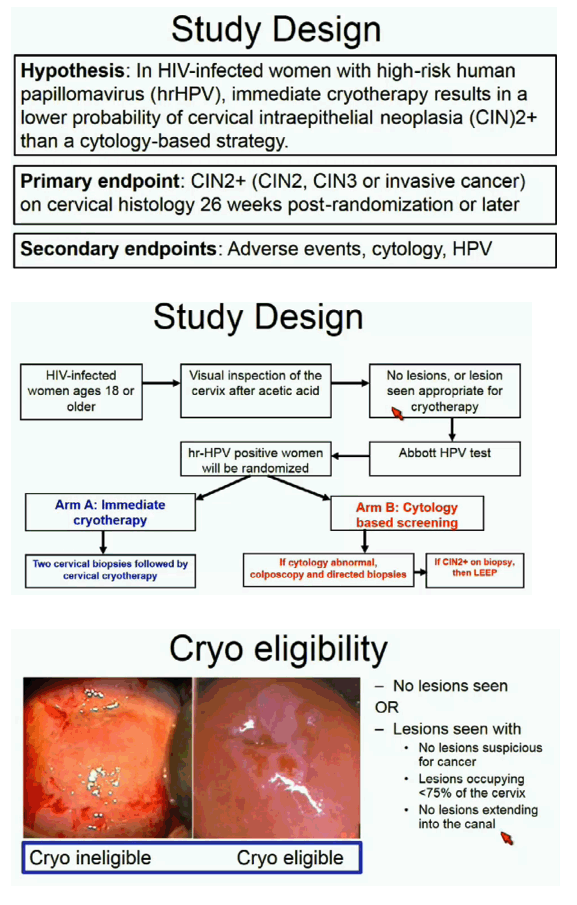
ACTG investigators hypothesized that HIV-positive women with hrHPV would have a lower probability of CIN2+ with immediate cryotherapy (test-and-treat) than with a cytology-based approach. Women randomized to immediate cryotherapy had two cervical biopsies followed by cryotherapy; women randomized to cytology-based screening had colposcopy and directed biopsies if cytology was abnormal, then LEEP if biopsy confirmed CIN2+ [3]. The primary endpoint was CIN2+ (CIN2, CIN3, or invasive cancer) on cervical histology 26 weeks or more after randomization.
The trial enrolled nonpregnant HIV-positive women at least 18 years old with any CD4 count or HIV load. No one had a history of cervical, vulvar, or vaginal cancer, and no one had lesions that might be cancer. Women had cytology and samples stored for HPV testing every 6 months. Colposcopy and directed biopsies occurred at weeks 26, 78, and 130.
The 288 study participants (145 test-and-treat, 143 cytology) had a median age of 35 (interquartile range 31 to 41) and a median CD4 count of 501. Almost two thirds of women, 62%, had an undetectable HIV load. While 74% of women were black, 19% were Asian and 7% Hispanic. Women lived in Africa, India, Haiti, or Peru. While 237 women (82%) completed protocol-defined follow-up, 51 (18%) dropped out early.
Among 145 women randomized to test-and-treat, 143 successfully underwent cervical biopsies and cryotherapy. Thirteen of these 145 (9%) had no result, 93 (64%) had normal biopsies or CIN1, 38 (26%) had CIN2 or CIN3, and 1 (1%) had cervical cancer. Among 143 women in the cytology group, 92 (64%) had abnormal cytology, 85 (92%) of whom had colposcopy and biopsies. Of the 51 women with normal cytology, 22 (43%) had colposcopy and biopsies. Twenty-two women (15%) in the cytology group got diagnosed with CIN2/3, but only 12 of those 22 had LEEP.
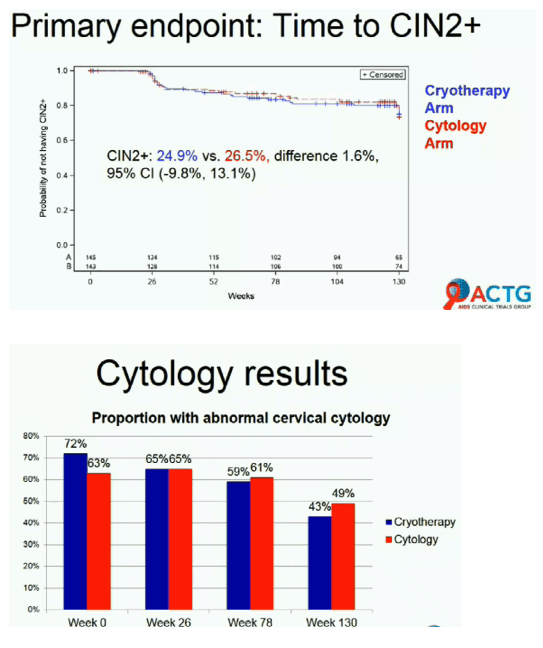
Development of CIN2+ did not differ significantly between the test-and-treat group (24.9%) and the cytology group (26.5%) (difference 1.6%, 95% confidence interval -9.8% to 13.1%). Proportions of women with abnormal cervical cytology were similar in the test-and-treat arm and the cytology arm at week 0 (72% and 63%), week 26 (65% and 65%), week 78 (59% and 61%), and week 130 (43% and 49%). Rates of hrHPV at week 26 were 61% in the test-and-treat arm and 70% in the cytology arm.
The ACTG team concluded that the hrHPV test-and-treat strategy does not improve CIN outcomes 2.5 years after therapy when compared with single-round cytology-based screening. They suggested the results may reflect worse-than-expected response to cryotherapy in these women. But the investigators proposed that the test-and-treat approach to screening "is feasible for lower resource settings and leads to similar outcomes as the resource-intensive cytology-based approach."
References
1. Wilkin T, Matining R, Sahasrabuddhe V, et al. ACTG A5282: HPV test & treat vs. cytology-based cervical cancer prevention in HIV+ women. 25th Conference on Retroviruses and Opportunistic Infections (CROI). March 4-7, 2018. Boston. Abstract 134.
2. Kuhn L, Wang C, Tsai WY, Wright TC, Denny L. Efficacy of human papillomavirus-based screen-and-treat for cervical cancer prevention among HIV-infected women. AIDS. 2010;24:2553-2561.
3. ClinicalTrials.gov. HPV test-and-treat-strategy versus cytology-based strategy for prevention of CIN2+ in HIV-infected women. ClinicalTrial.gov identifier NCT01315353.
----------------------------
therapeutic HPV vaccines in development - 2 pdfs attached
Unlike prophylactic HPV vaccines, which are used to generate neutralizing antibodies against viral particles, therapeutic HPV vaccines are used to stimulate cell-mediated immune responses to specifically target and kill infected cells.
The identification of high-risk HPV as the etiological factor for many diseases provides justification for the development of therapeutic HPV vaccines. The recent developments in the field as well as those discussed in this review have helped contribute to the foundational movement to eradicate HPV and HPV-associated diseases and malignancies. In this review we discussed the various methods of targeting HPV oncoproteins E6 and E7, which represent tumor-specific antigens and excellent targets for therapeutic HPV vaccines. Based on our own previous studies, and those conducted by other investigators in the field, we believe that the current therapeutic HPV vaccines mentioned in this review each possess advantages and limitations. Additional clinical studies are still necessary to further verify the antitumor efficacy of therapeutic HPV vaccines.
With continued efforts to improve and develop therapeutic treatment strategies, we anticipate the continued success of therapeutic HPV vaccines over the next few years, and beyond. We believe that therapeutic HPV vaccines will become clinically available in the near future and be offered alongside other available therapies for the control of HPV-associated diseases.
-----------------------------
excerpts


|
| |
|
 |
 |
|
|