 |
 |
 |
| |
Lopinavir/ritonavir Induces Mitochondrial Toxicity in HIV-exposed Uninfected Children
|
| |
| |
Reported by Jules Levin
CROI 2018 March 4-7 Boston MA
Audrey Monnin1, Nicolas Nagot1,2, Marianne Peries1, Roselyne Vallo1, Nicolas Meda3, Mandisa Singata-Madliki4, James K. Tumwine5, Chipepo Kankasa6, Pascal Reynier7, Thorkild Tylleskar8, Philippe Van de Perre1,2, Jean-Pierre Molès1, for the PROMISE PEP consortium ANRS 12174
1University of Montpellier, INSERM, EFS, Montpellier, France, 2CHU Montpellier, Montpellier, France, 3University of Ouagadougou, Ouagadougou, Burkina Faso, 4University of Fort Hare, East London, South Africa, 5University of Makerere, Kampala, Uganda, 6University Teaching Hospital, Lusaka, Zambia, 7CHU Angers, Angers, France, 8University of Bergen, Bergen, Norway


Program abstract:
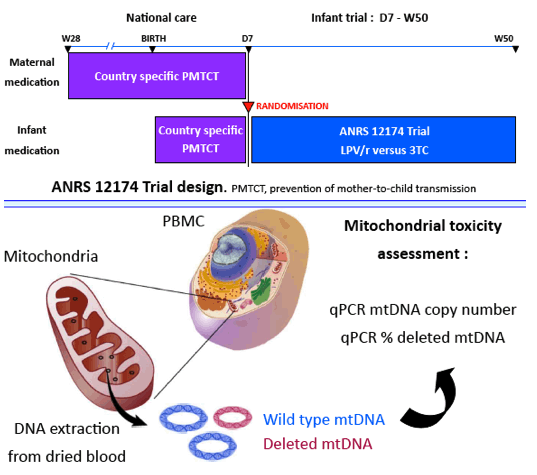
Long-term ART, such as lopinavir-ritonavir (LPV/r) and lamivudine (3TC) can be given to newborns either for HIV treatment or prophylaxis (PrEP) to prevent maternal transmission through breastfeeding. The potential mitochondrial toxicity of these drugs has never been evaluated without the concurrent confounding effect of HIV. This study aimed to quantify the mitochondrial toxicity of 3TC or LPV/r regimens after 1 year of PrEP among HIV-exposed uninfected infants.
We have randomly selected 151 participants having completed the ANRS 12174 randomized controlled trial which enrolled HIV-uninfected children from untreated HIV-infected mothers. We showed that infant 3TC and LPV/r PrEP given from day 7 until 1 year could reduce postnatal HIV transmission to 1.3%. The number of mitochondrial DNA copies per cell (MCN) and the percentage of mitochondrial DNA carrying deletions (MDD) were assessed by real time PCR on stored dried blood spots collected at 7 days after birth and at 1 year. PCR protocols were performed according to the criteria for the diagnostic of mitochondriopathies from the French reference center. A clinically-relevant mtDNA depletion was defined as a reduction of 70% of MCN at 1 year.
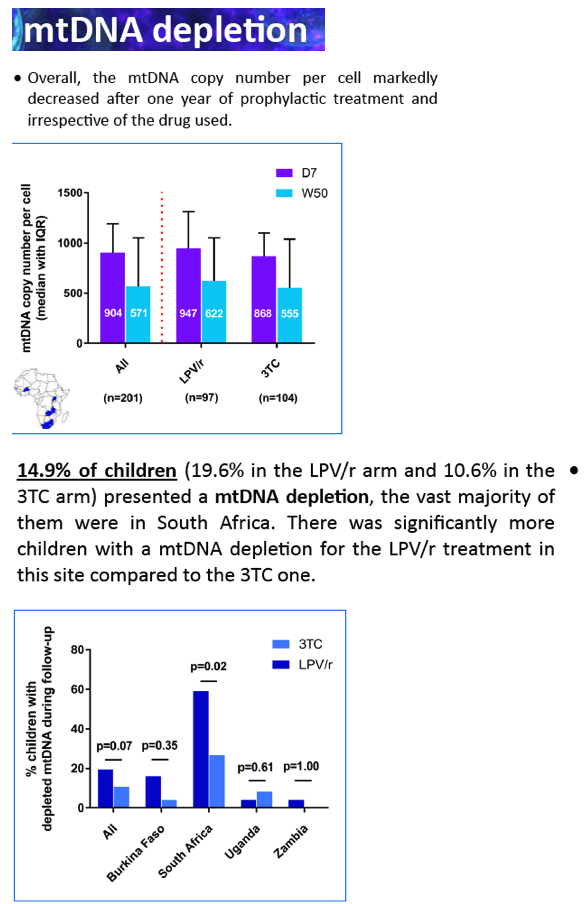
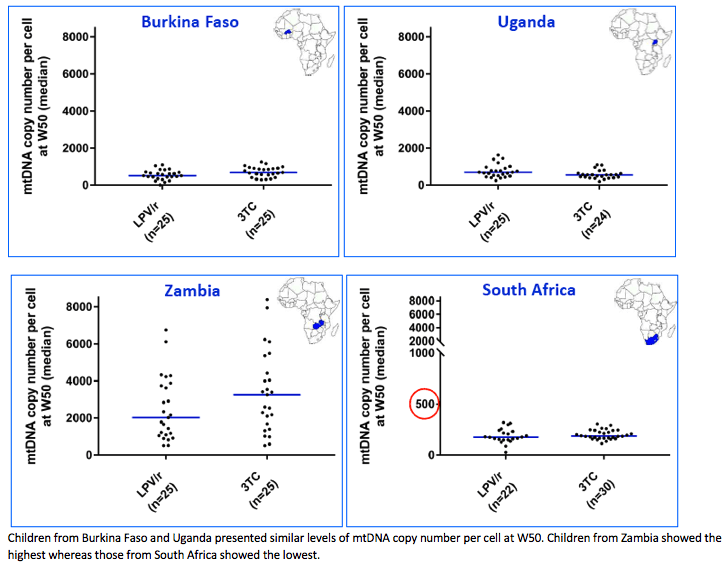
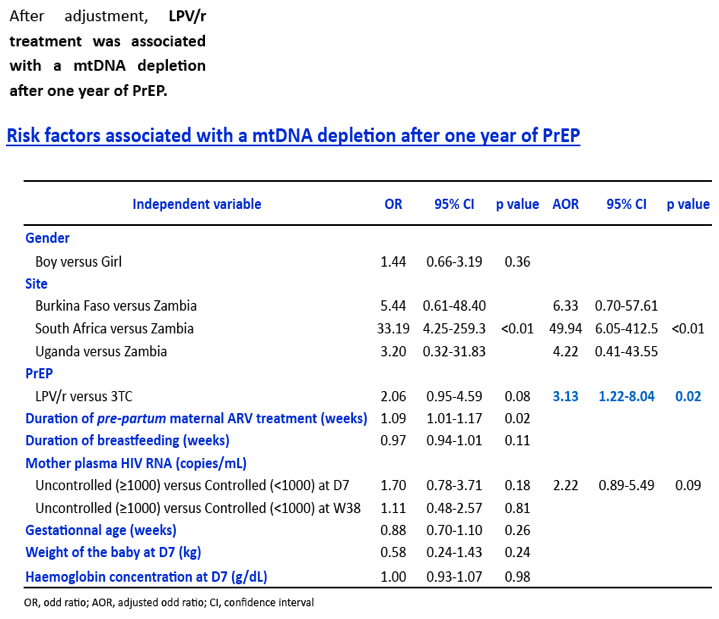
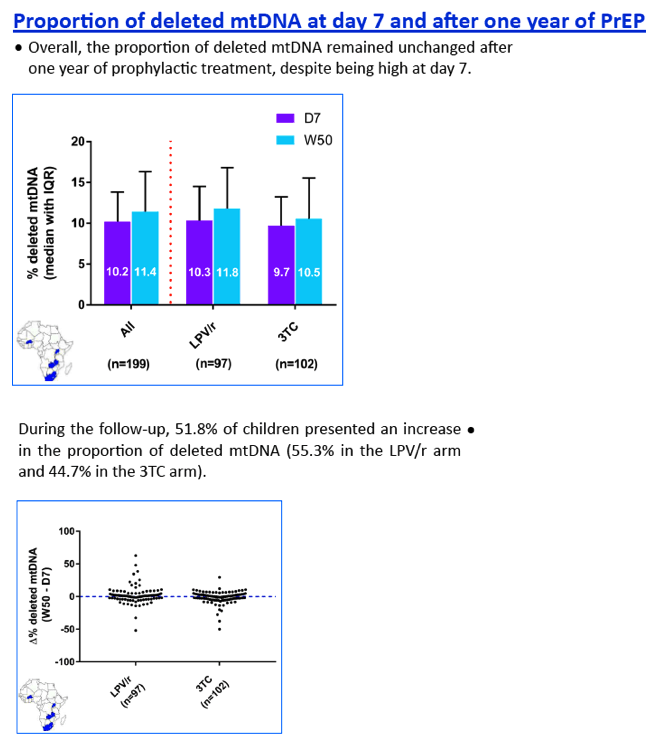
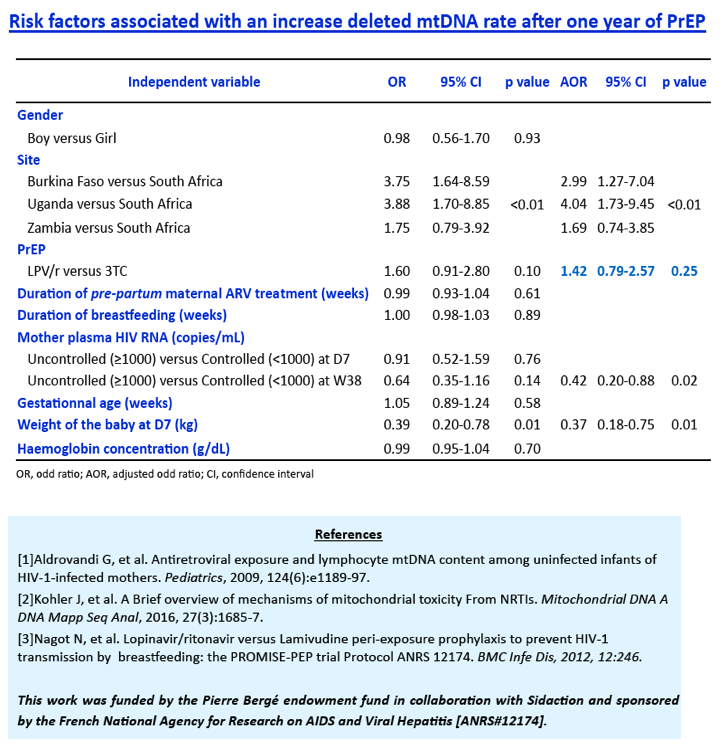
Before PrEP initiation (D7), the median MCN was within normal range (823, IQR:555-1076), while any MDD was unexpectedly detected in 147/149 children, with a median 11.2% (IQR:8.5-15.1) of mtDNA deleted, without difference between arms. After 1 year of PrEP, overall median MCN dropped by 41.2% (IQR:6.9-64.2) without difference between arms (p=0.59). Twenty-nine (19.2%) children showed a mtDNA depletion, without difference between arms (p=0.19). After adjustment for gender, duration of breastfeeding, duration of pre-partum maternal prophylaxis and gestational age, LPV/r tented to be associated with mtDNA depletion (reference = 3TC; OR=1.98, IC95%:0.81-4.87; p=0.14). Similarly, the rate of MDD remained high in both arms (13.7%, IQR:3.2-16.5 for 3TC and 14.6%, IQR:9.6-19.1 for LPV/r) but without difference between them (p=0,42).
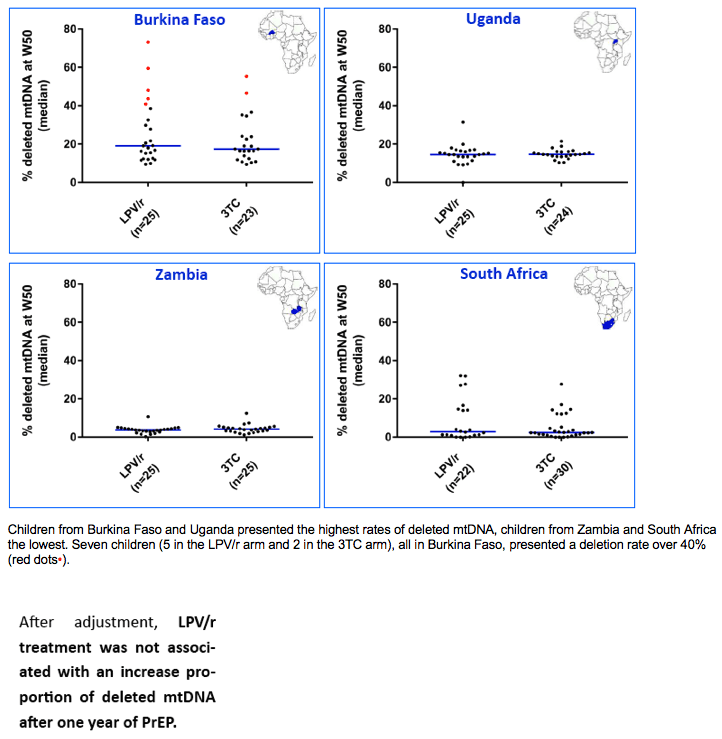
At D7, the mtDNA deletion rate among HIV-exposed infants was close to rates observed in some inherited mitochondrial diseases. After 1 year of PrEP, an unexpected and important mitochondrial toxicity was observed (mtDNA depletion) for LPV/r, which compares to the expected effect of 3TC. The severity of these defects contrasts with the paucity of clinical sign and symptoms related to mitochondriopathies.
|
| |
|
 |
 |
|
|