 |
 |
 |
| |
Preclinical antiviral drug combination studies utilizing novel orally bioavailable agents for chronic hepatitis B infection: AB-506, a next generation HBV capsid inhibitor, and AB-452, an HBV RNA destabilizer
|
| |
| |
full pdf slide presentation attached
Reported by Jules Levin
EASL - The International Liver Congress 2018,
April 11 -15, 2018, Paris, France
Rene Rijnbrand
Arbutus Biopharma Inc.
Download the PDF here

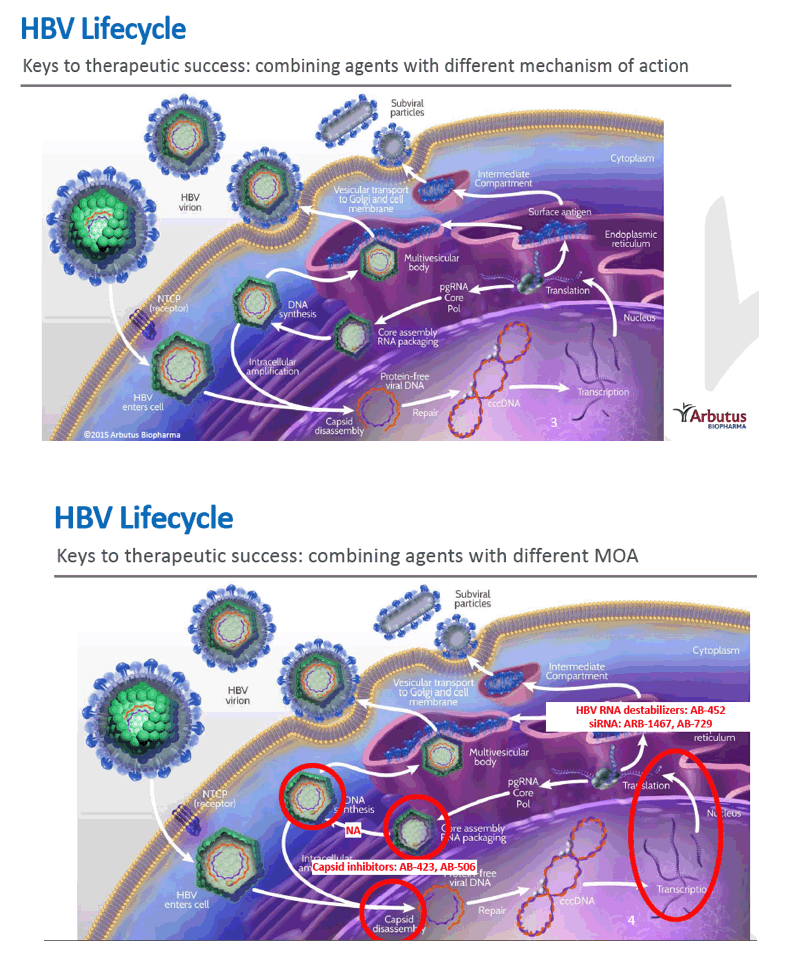
Source: Hepatitis B. WHO (2017) http://www.who.int/mediacentre/factsheets/fs204/en/
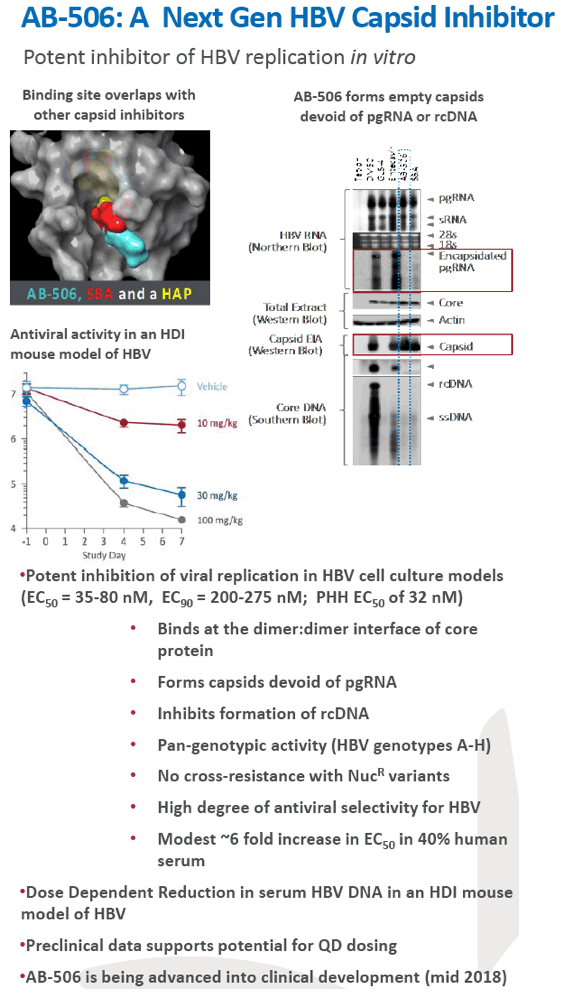
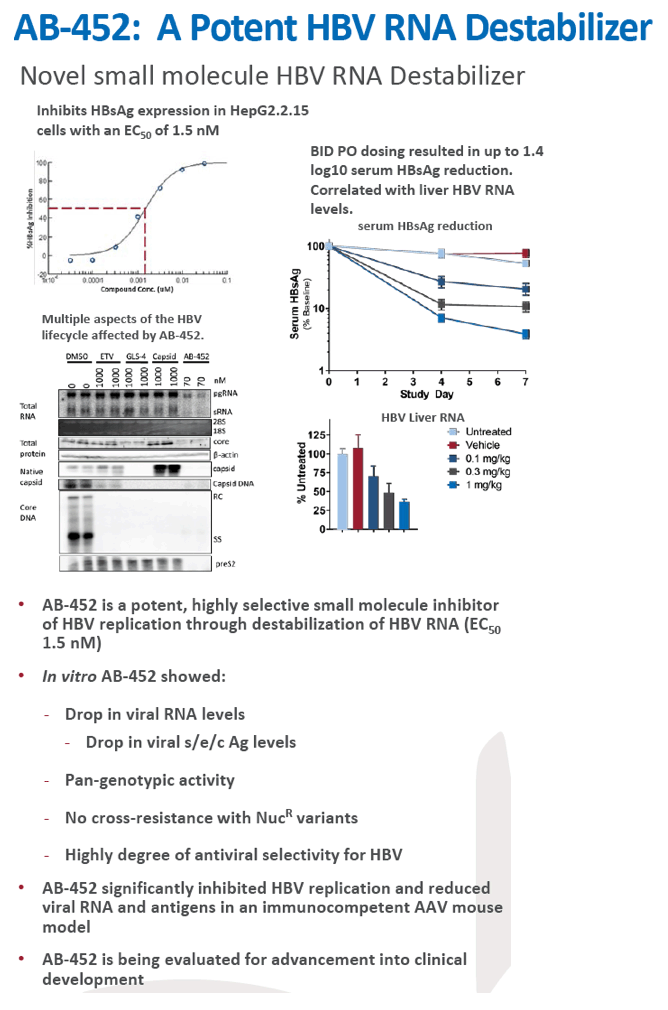

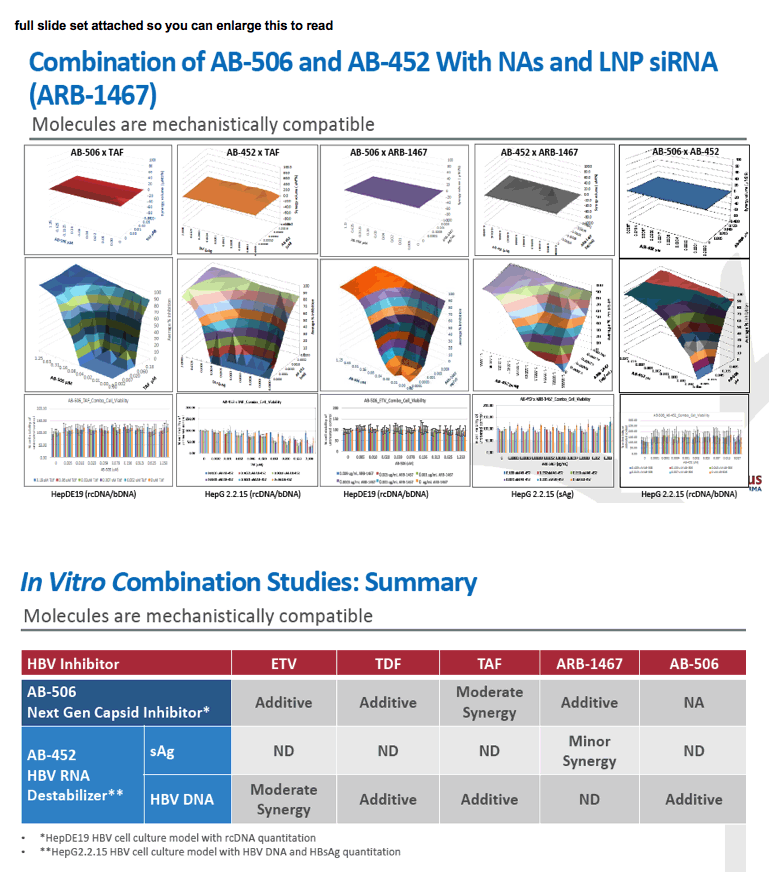
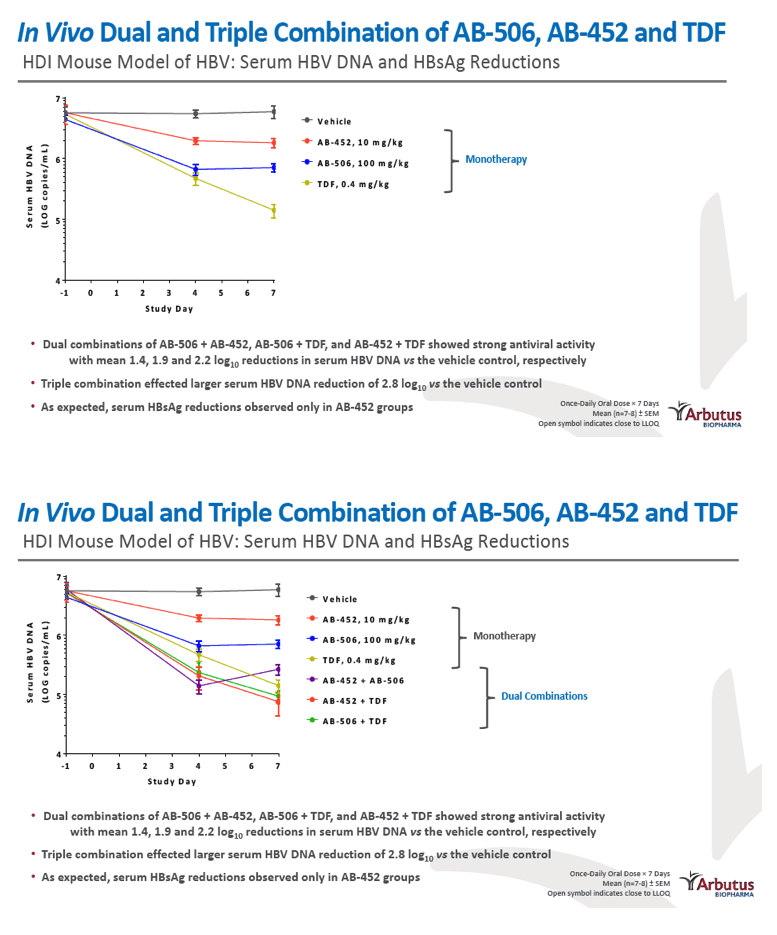
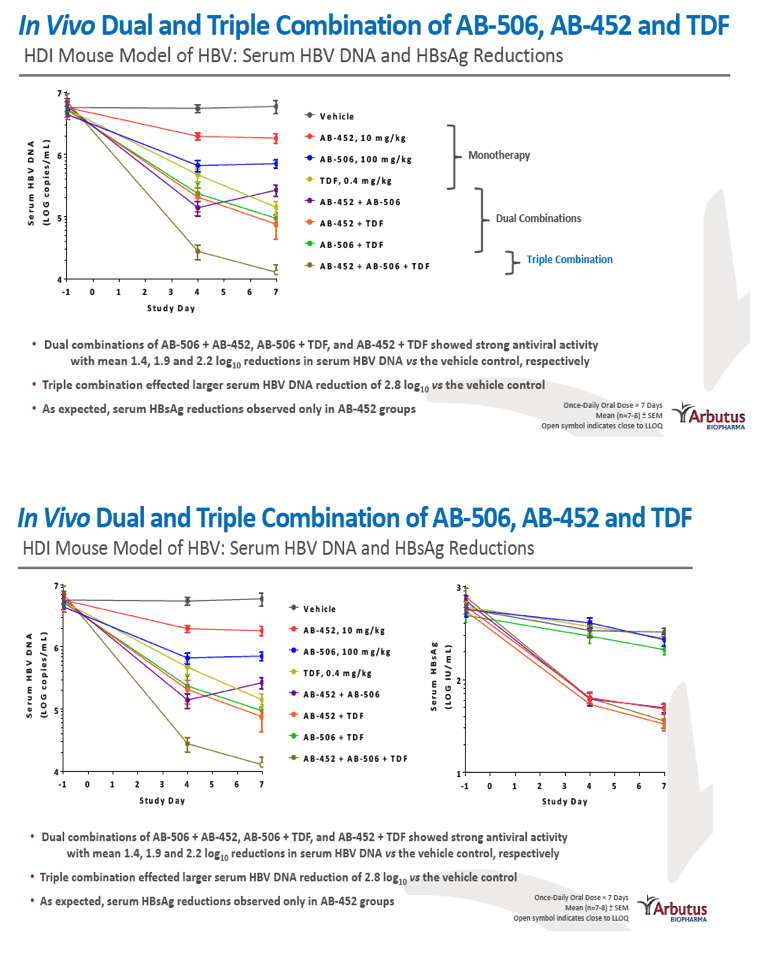
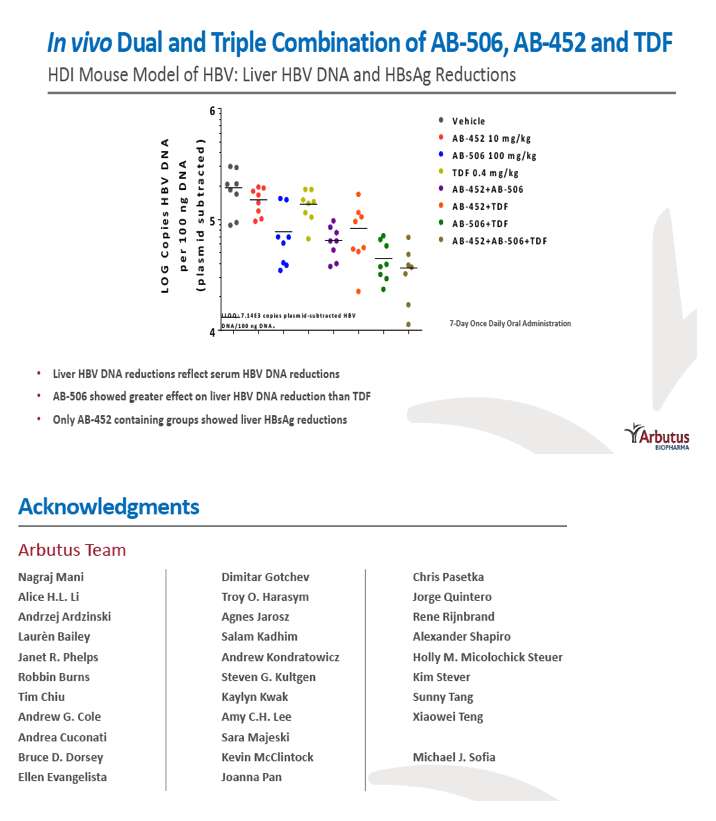
|
| |
|
 |
 |
|
|