 |
 |
 |
| |
Mixed Signals With Cardiovascular Markers in Dolutegravir Switch Study
|
| |
| |
HIV Drug Therapy, Glasgow 2018, October 28-31, 2018, Glasgow
Mark Mascolini
NEAT trial participants with high cardiovascular risk who switched from a boosted protease inhibitor (PI) to dolutegravir had one biomarker change suggesting cardiovascular benefit and one suggesting greater cardiovascular risk [1]. This 48-week biomarker analysis underlines the complexity of biomarker interpretation and cardiovascular risk in people with HIV.
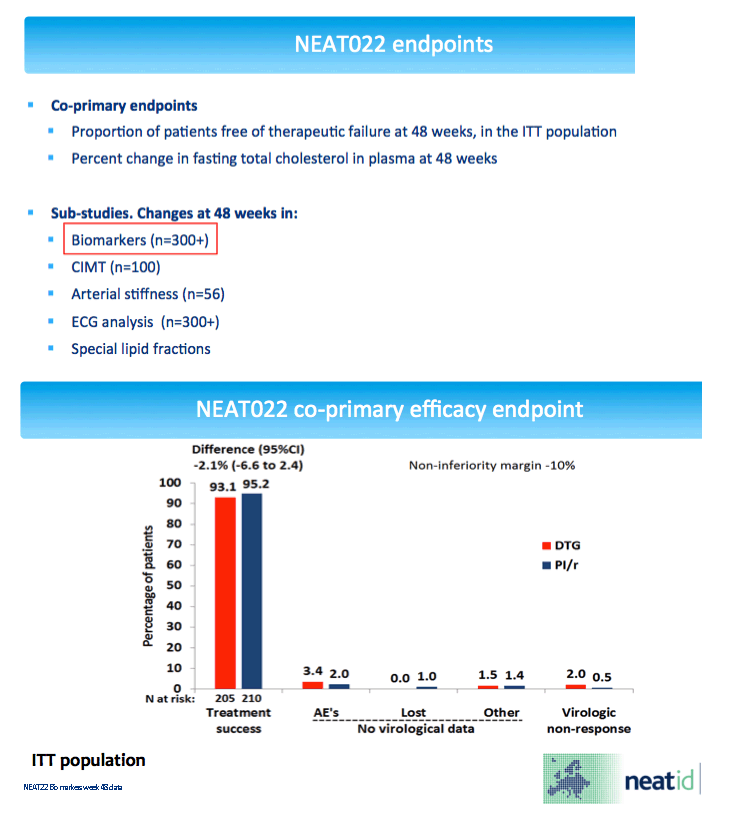
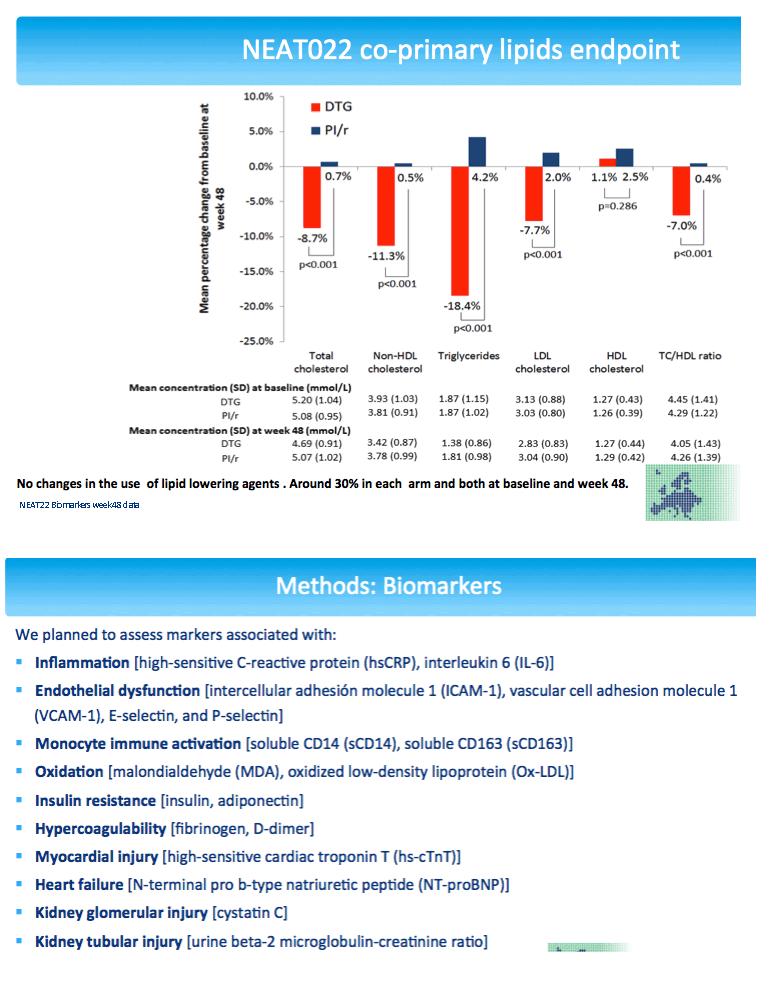
NEAT randomized virologically suppressed people taking a boosted PI to continue that PI or switch to dolutegravir, an integrase inhibitor. Everyone was older than 50 and/or had a Framingham cardiovascular risk score above 10% at 10 years. After 48 weeks people randomized to dolutegravir had a 7.7% drop in "bad" low-density lipoprotein (LDL) cholesterol [2], a change linked to reduced risk of major cardiovascular events in the general population.
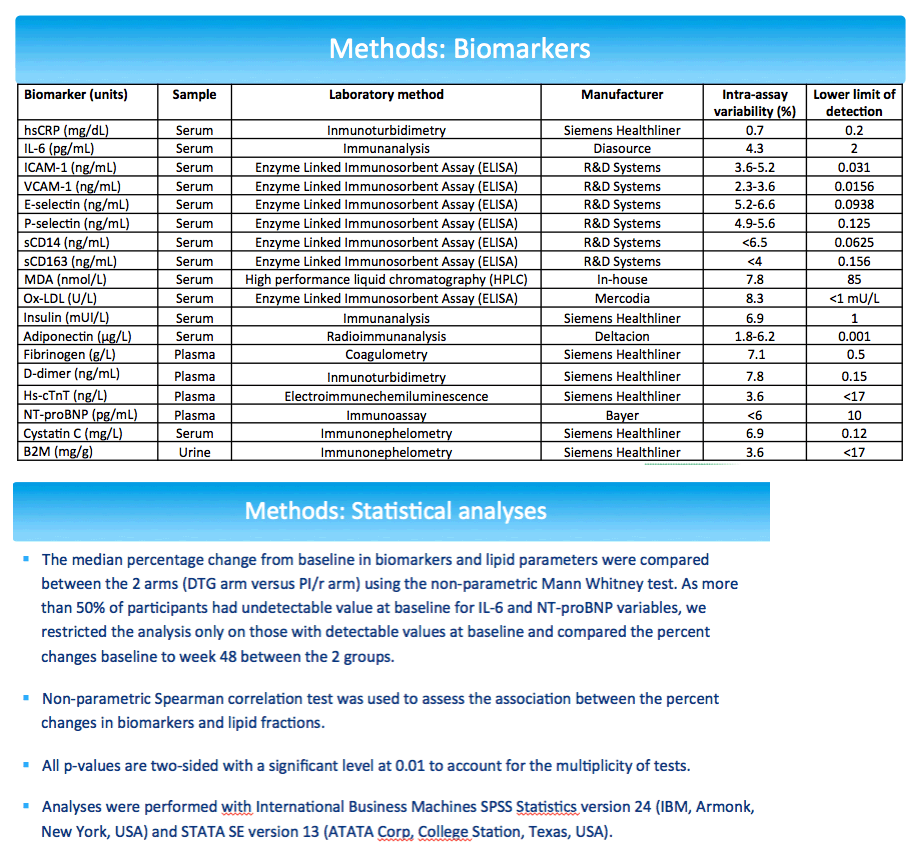
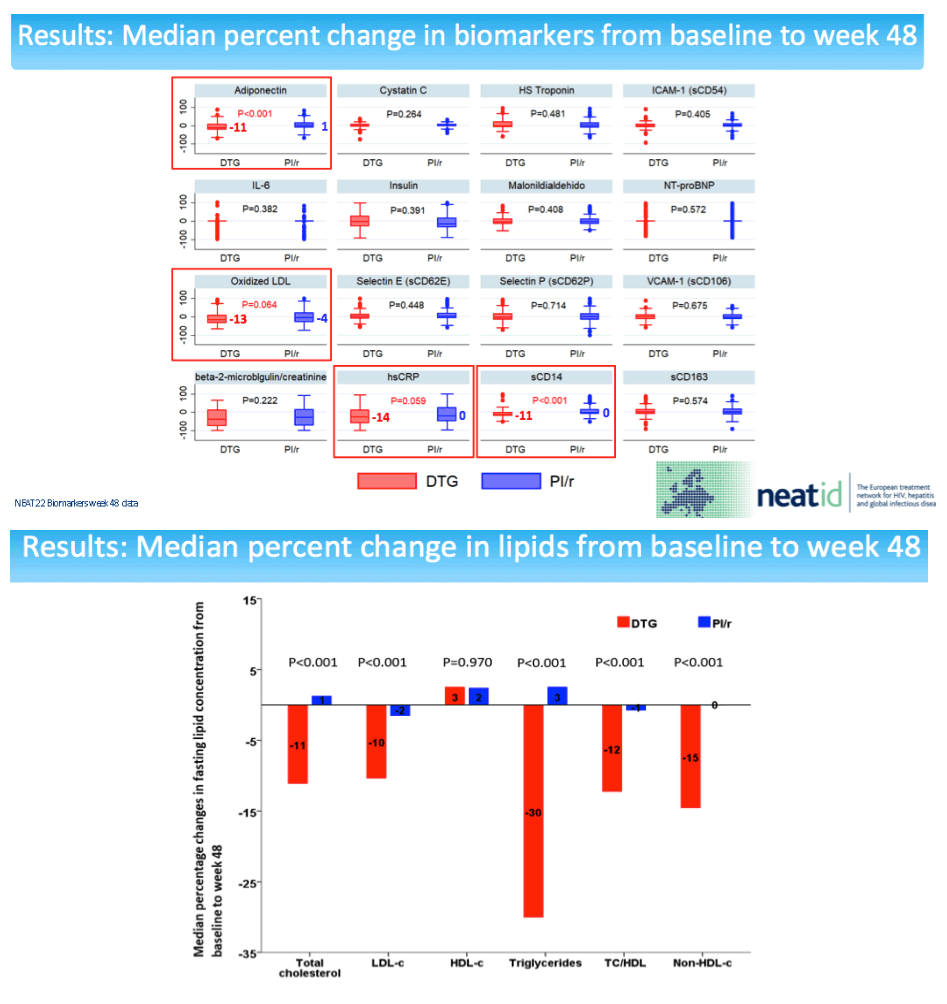
NEAT investigators conducted a new study to see if analysis of markers related to cardiovascular disease pathogenesis would shed more light on the cardiovascular impact of switching from a boosted PI to dolutegravir. The analysis involved participants who stayed with their assigned regimen through 48 weeks. The NEAT team calculated median percent change from baseline in an array of biomarkers: (1) inflammation: high-sensitivity C-reactive protein (hsCRP) and interleukin-6 (IL-6); (2) endothelial dysfunction: intercellular adhesion molecule-1 (ICAM-1), vascular cell adhesion molecule-1 (VCAM-1), selectin E and P; (3) monocyte activation: soluble CD14 (sCD14) and CD163 (sCD163); (4) oxidation: oxidized LDL, malondialdehyde (MDA); (5) insulin resistance: adiponectin, insulin; (6) hypercoagulability: fibrinogen, D-dimer; (7) myocardial injury: high-sensitivity cardiac troponin T; (8) heart failure: NT-proBNP; (9) kidney glomerular injury: cystatin C; and (10) kidney tubular injury: urine beta-2 microglobulin-creatinine ratio.
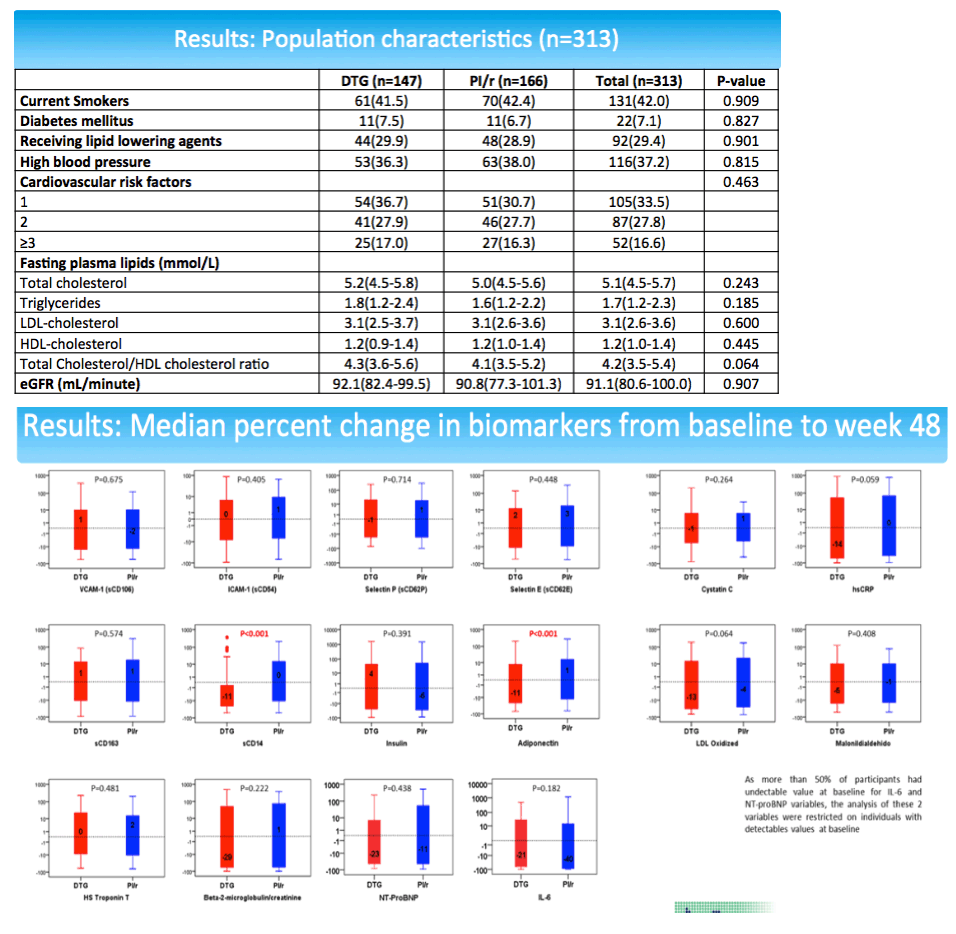
The study group included 147 people randomized to a dolutegravir switch and 166 randomized to a continued PI. Median age stood at 54, 87.5% were older than 50, and 75% had a Framingham risk score above 10%. Median time with an undetectable viral load stood at 4.7 years. Study arms did not differ substantially in any of these measures.
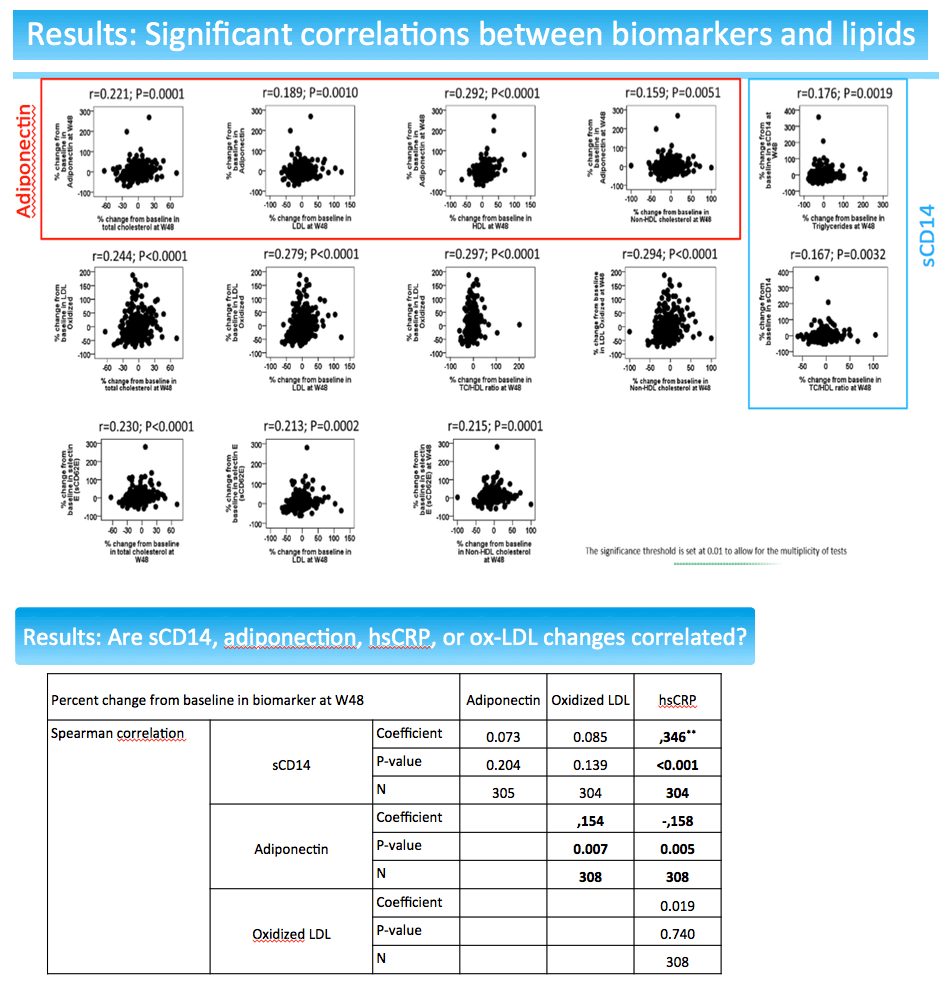
Compared with the PI group, through 48 weeks people switching to dolutegravir had significant drops in sCD14 (-11%, P < 0.001) and adiponectin (-11%, P < 0.001). There were trends to lower hsCRP (-14%, P = 0.059) and oxidized LDL (-13%, P = 0.064) in the dolutegravir group relative to the PI group through 48 weeks.
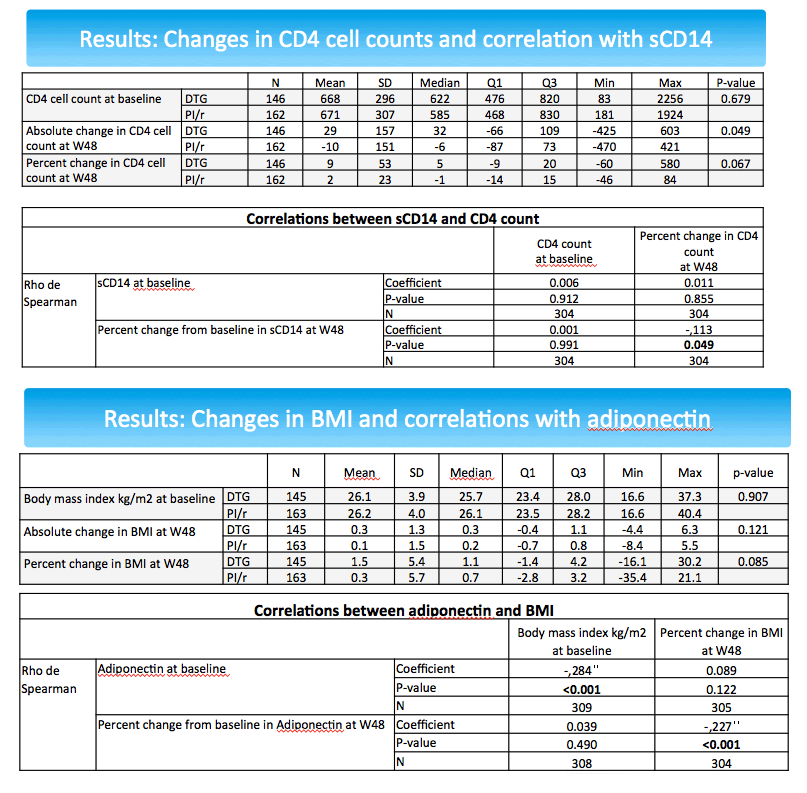
The sCD14 and adiponectin findings pose difficulties in interpretation. Declining sCD14 suggests decreasing cardiovascular effects [3], while falling adiponectin suggest worsening cardiovascular effects [4]. In the NEAT analysis percent change in adiponectin correlated inversely with percent change in body mass index (coefficient -0.227, P < 0.001). In other words, falling adiponectin meant rising body mass index, which would heighten cardiovascular risk. At the same time, percent change in sCD4 correlated inversely with percent change in CD4 count (coefficient -0.113, P = 0.049). So falling sCD4 meant a rising CD4 count.
The NEAT team stressed that the overall cardiovascular impact of switching from a PI to dolutegravir appears to be beneficial [2]. But this study's finding that falling adiponectin correlates with climbing body mass index after the dolutegravir switch "highlights the importance of further assessing the potential impact of dolutegravir therapy on the mechanisms involved in body weight." Analysis of 462 people who stopped dolutegravir in France found that 20% gained more than 10% in weight with the integrase inhibitor [5]. But in the NEAT biomarker analysis, median 48-week body mass index gain was 0.3 kg/m2 with dolutegravir and 0.2 kg/m2 with PIs, a nonsignificant difference (P = 0.121).
References
1. Martinez E, Assoumou L, Moyle G, et al. 48-week changes in biomarkers in subjects with high cardiovascular risk boosted switching from ritonavir-protease inhibitors to dolutegravir: the NEAT022 study. HIV Drug Therapy, Glasgow 2018, October 28-31, 2018, Glasgow. Abstract O113.
2. Gatell JM, Assoumou L, Moyle G, et al. Switching from a ritonavir-boosted protease inhibitor to a dolutegravir-based regimen for maintenance of HIV viral suppression in patients with high cardiovascular risk. AIDS. 2017;31:2503-2514.
3. Longenecker CT, Jiang Y, Orringer CE, et al. Soluble CD14 is independently associated with coronary calcification and extent of subclinical vascular disease in treated HIV infection. AIDS. 2014;28:969-977.
4. Ketlogetswe KS, Post WS, Li X, et al. Lower adiponectin is associated with subclinical cardiovascular disease among HIV-infected men. AIDS. 2014;28:901-909.
5. Menard A, Meddeb L, Tissot-Dupont H, et al. Dolutegravir and weight gain: an unexpected bothering side effect? AIDS. 2017;31:1499-1500.
https://journals.lww.com/aidsonline/fulltext/2017/06190/Dolutegravir_and_weight_gain___an_unexpected.18.aspx




|
| |
|
 |
 |
|
|