 |
 |
 |
| |
No Uptick in HIV Replication After Switch to DTG/3TC in Randomized Trial
|
| |
| |
HIV Drug Therapy, Glasgow 2018, October 28-31, 2018, Glasgow
Mark Mascolini
Switching from standard 3-drug antiretroviral therapy (ART) to a dolutegravir/lamivudine (DTG/3TC) 2-drug combination in a randomized trial did not boost low-level viral replication: Residual viremia remained stable in both treatment arms. The findings indicate that DTG/3TC does not allow low-level HIV replication typically controlled by 3-drug combos.
Researchers who conducted this study noted that most ART-treated people with a viral load undetectable on standard assays have residual viremia, that is, a few copies detectable by highly sensitive single-copy HIV assays. Bolstering standard ART regimens with additional antiretrovirals has no impact on residual viremia. But questions remains about whether increasingly popular 2-drug antiretroviral combinations control residual viremia as well as standard 3-drug regimens.
To address that question, ASPIRE study investigators analyzed residual viremia when the trial began and after 24 and 48 weeks. This open-label study recruited people taking a stable 3-drug regimen with a viral load below 20 copies and no record of previous virologic failure. Most trial participants, 88%, were men, and median age stood at 47 years.
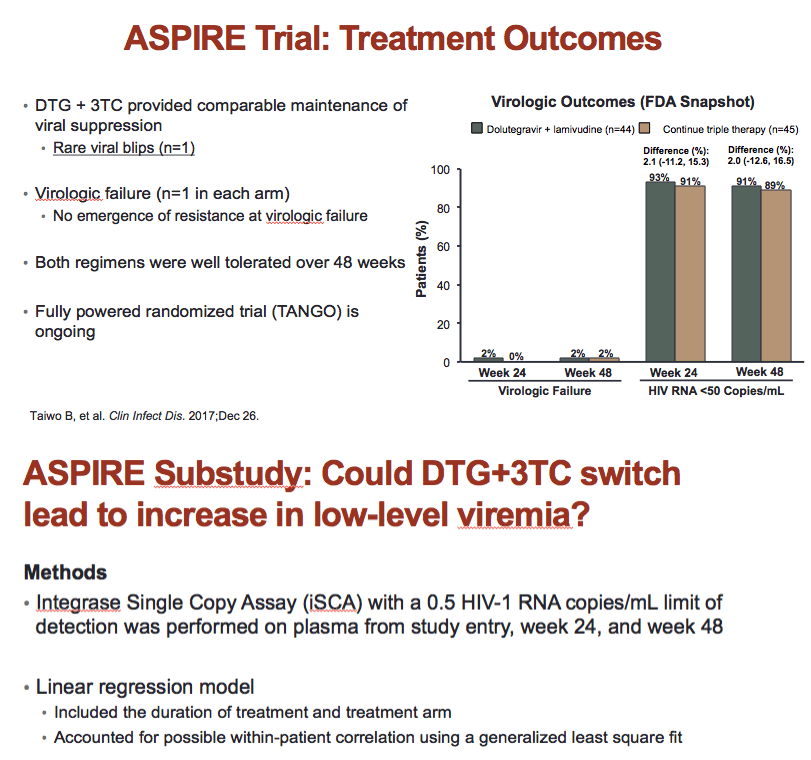
Researchers randomized 89 people to continue their 3-drug combination or to switch to only 2 drugs, 3TC and DTG, an integrase inhibitor, taken once daily. After 48 weeks 91% of participants in the DTG/3TC group and 89% in the 3-drug group had maintained a viral load below 50 copies, a result indicating the noninferiority of DTG/3TC to triple therapy [2]. One person in each group had virologic failure, and resistance mutations did not emerge with either failure.
The residual viremia analysis relied on an integrase single-copy assay with a lower detection limit of 0.5 HIV RNA copies per milliliter. The investigators used this assay on samples collected at study entry, week 24, and week 48. To determine between-group differences in residual viremia, they used linear regression adjusted for time on treatment and treatment arm. The analysis excluded participants who stopped ART during the study.
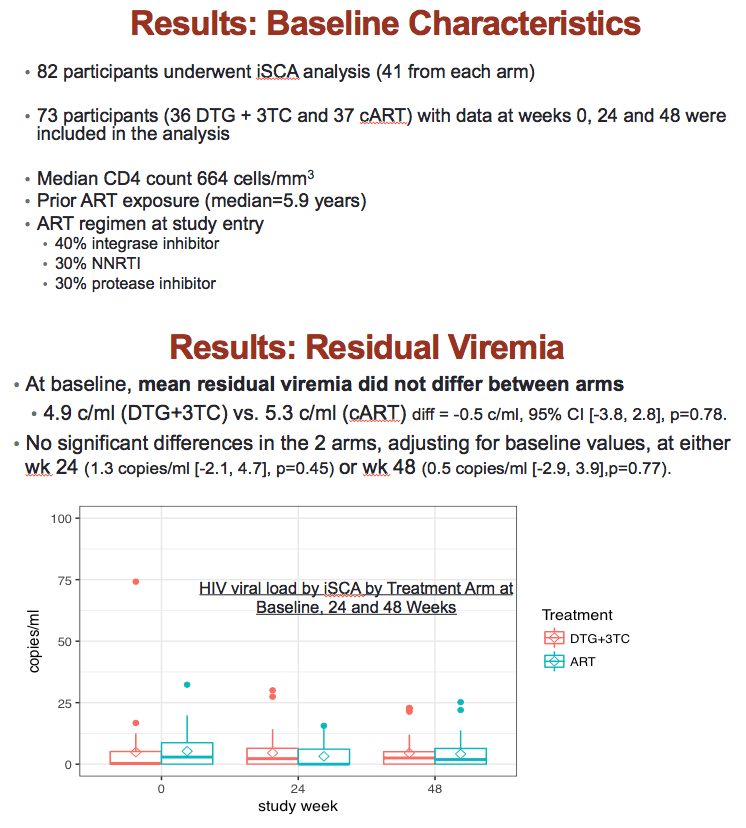
Forty-one people in each treatment arm got tested with the integrase single-copy assay. They had taken ART for a median of 5.9 years and had a median CD4 count of 644. When the study began, 40% were taking an integrase inhibitor, 30% a nonnucleoside, and 30% a protease inhibitor. Thirty-six people randomized to DTG/3TC and 37 continuing triple therapy had single-copy results available from weeks 0, 24, and 48.
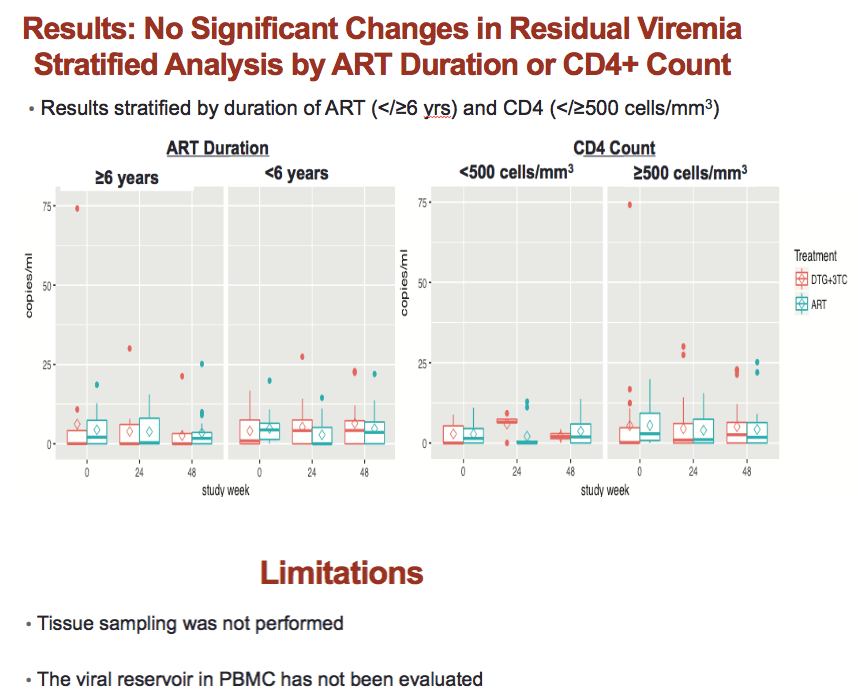
On the initial single-copy measure, residual viremia averaged 4.9 copies/mL in the DTG/3TC group and 5.3 copies/mL in the other group. The resulting 0.5-copy/mL difference was not statistically significant (P = 0.78). In an analysis adjusting for baseline values, no significant difference between treatment arms emerged at week 24 (1.3 copies/mL difference, P = 0.45) or week 48 (0.5 copy/mL difference, P = 0.77). The ASPIRE team saw no residual viremia difference between treatment groups when they divided results according to ART duration (6 or more years versus fewer) or CD4 count (500 or more CD4s versus fewer).
The researchers concluded that in their 48-week trial of DTG/3TC versus standard triple therapy, "there was no evidence for increased viral replication after [the] switch to DTG + 3TC as reflected by stable levels of residual viremia." They believe their findings support continued study of 2-drug therapy with DTG/3TC. TANGO, a larger study of DTG/3TC, is under way [3].
References
1. Li J, Sax P, Marconi V, et al. No significant changes to residual viremia after switch to dolutegravir and lamivudine in a randomized trial. HIV Drug Therapy, Glasgow 2018, October 28-31, 2018, Glasgow. Abstract O145.
2. Taiwo BO, Marconi VC, Berzins B, et al. Dolutegravir plus lamivudine maintain HIV-1 suppression through week 48 in a pilot randomized trial. Clin Infect Dis. 2018;66:1794-1797. https://academic.oup.com/cid/article-abstract/66/11/1794/4775007
3. ClinicalTrial.gov. Switch study to evaluate dolutegravir plus lamivudine in virologically suppressed human immunodeficiency virus type 1 positive adults (TANGO). ClinicalTrials.gov identifier NCT03446573. https://clinicaltrials.gov/ct2/show/NCT03446573
Non-Inferior Efficacy of Dolutegravir (DTG) Plus Lamivudine (3TC) vs DTG Plus Tenofovir/Emtricitabine (TDF/FTC) Fixed-Dose Combination in Antiretroviral Treatment-Naive Adults With HIV-1 Infection-Week 48 Results From the GEMINI Studies - (07/25/18)
Dolutegravir plus lamivudine maintains HIV-1 suppression through week 48 in a pilot randomized trial (ASPIRE) - (11/08/17)



|
| |
|
 |
 |
|
|