 |
 |
 |
| |
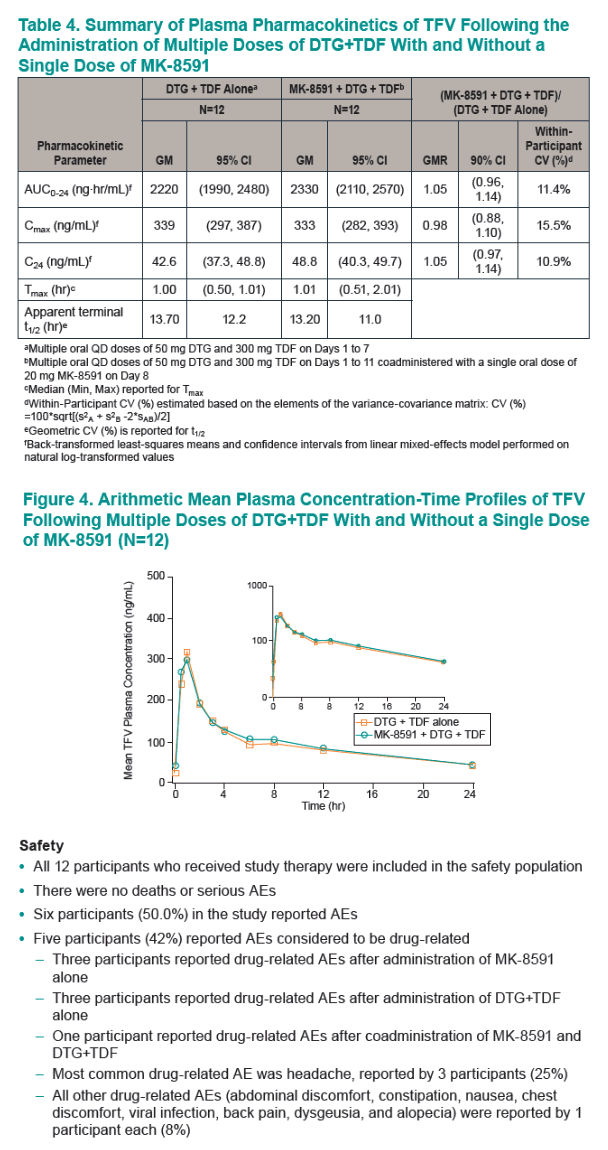
Pharmacokinetics of MK-8591, Dolutegravir and Tenofovir Disoproxil Fumarate Are Not Altered After Coadministration When Compared to Single Agent Administration
|
| |
| |
Reported by Jules Levin
HIV Glasgow; Glasgow, UK; 28-31 October 2018.
D. Jackson Rudd1; S. Zhang1; K. L. Fillgrove1; S. Fox-Bosetti1; R. Matthews1; E. Friedman1; D. Armas2; S. A. Stoch1; M. Iwamoto1
1Merck & Co., Inc., Kenilworth, NJ, USA; 2Celerion, Tempe, AZ, USA
MK-8591 is a nucleoside reverse transcriptase translocation inhibitor.
New Study (DRIVE2Simplify): MK-8591 With Doravirine and Lamivudine in Participants Infected With Human Immunodeficiency Virus Type 1 (MK-8591-011)
MK-8591 Protects 8 of 8 Macaques From Rectal SHIV 6 Days After Dosing
GLASGOW: Efficacy of MK-8591 against diverse HIV-1 subtypes and NRTI-resistant clinical isolates - (11/01/18) oral
GLASGOW: Efficacy of MK-8591 Against Diverse HIV-1 Subtypes and NRTI-Resistant Clinical Isolates - (10/31/18) poster
IAC/2018: Understanding the Resistance Profile of the HIV-1 NNRTI Doravirine in Combination With the Novel NRTTI MK-8591
CROI/2018: MULTIPLE DAILY DOSES OF MK-8591 AS LOW AS 0.25 MG ARE EXPECTED TO SUPPRESS HIV
Antiviral Activity of EFdA [MK-8591] Against NRTI-Sensitive and -Resistant Strains of HIV-2......."potential pre-exposure prophylaxis agent to prevent HIV transmission in women and their infants......MK-8591 (or EFdA) is a NRTI with very potent in vitro activity against HIV....Viral suppression was maintained for at least 7 days after the last dose. This study provides proof of concept for a once weekly oral dosing strategy of MK-8591....."Our data demonstrated that EFdA efficiently prevents both vaginal and oral HIV transmission. Together with EFdA's relatively low toxicity and high potency against drug-resistant HIV strains, these data support further clinical development of EFdA as a potential pre-exposure prophylaxis (PrEP) agent to prevent HIV transmission in women and their infants."
EFdA / MK-8591 - HIV pre-exposure prophylaxis for women and infants prevents vaginal and oral HIV transmission in a preclinical model of HIV infection - (08/05/16)
Pre-exposure Prophylaxis with EFdA Offers Strong Protection against High Dose Mucosal HIV Challenges - [Long-Acting New NRTI for Treatment & PrEP] - (10/24/16)



|
| |
|
 |
 |
|
|