| |
|
Glecaprevir-Pibrentasvir for 8 or 12 Weeks in HCV Genotype 1 or 3 Infection
|
| |
| |
NEJM Jan 25 2018 - S. Zeuzem, G.R. Foster, S. Wang, A. Asatryan, E. Gane, J.J. Feld, T. Asselah, M. Bourliere, P.J. Ruane, H. Wedemeyer, S. Pol, R. Flisiak, F. Poordad, W.-L. Chuang, C.A. Stedman, S. Flamm, P. Kwo, G.J. Dore, G. Sepulveda-Arzola, S.K. Roberts, R. Soto-Malave, K. Kaita, M. Puoti, J. Vierling, E. Tam, H.E. Vargas, R. Bruck, F. Fuster, S.-W. Paik, F. Felizarta, J. Kort, B. Fu, R. Liu, T.I. Ng, T. Pilot-Matias, C.-W. Lin, R. Trinh, and F.J. Mensa
Abstract
Background
Glecaprevir and pibrentasvir are direct-acting antiviral agents with pangenotypic activity and a high barrier to resistance. We evaluated the efficacy and safety of 8-week and 12-week courses of treatment with 300 mg of glecaprevir plus 120 mg of pibrentasvir in patients without cirrhosis who had hepatitis C virus (HCV) genotype 1 or 3 infection.
Methods
We conducted two phase 3, randomized, open-label, multicenter trials. Patients with genotype 1 infection were randomly assigned in a 1:1 ratio to receive once-daily glecaprevir-pibrentasvir for either 8 or 12 weeks. Patients with genotype 3 infection were randomly assigned in a 2:1 ratio to receive 12 weeks of treatment with either glecaprevir-pibrentasvir or sofosbuvir-daclatasvir. Additional patients with genotype 3 infection were subsequently enrolled and nonrandomly assigned to receive 8 weeks of treatment with glecaprevir-pibrentasvir. The primary end point was the rate of sustained virologic response 12 weeks after the end of treatment.
Results
In total, 1208 patients were treated. The rate of sustained virologic response at 12 weeks among genotype 1-infected patients was 99.1% (95% confidence interval [CI], 98 to 100) in the 8-week group and 99.7% (95% CI, 99 to 100) in the 12-week group.
Genotype 3-infected patients who were treated for 12 weeks had a rate of sustained virologic response at 12 weeks of 95% (95% CI, 93 to 98; 222 of 233 patients) with glecaprevir-pibrentasvir and 97% (95% CI, 93 to 99.9; 111 of 115) with sofosbuvir-daclatasvir; 8 weeks of treatment with glecaprevir-pibrentasvir yielded a rate of 95% (95% CI, 91 to 98; 149 of 157 patients). Adverse events led to discontinuation of treatment in no more than 1% of patients in any treatment group.
Conclusions
Once-daily treatment with glecaprevir-pibrentasvir for either 8 weeks or 12 weeks achieved high rates of sustained virologic response among patients with HCV genotype 1 or 3 infection who did not have cirrhosis. (Funded by AbbVie; ENDURANCE-1 and ENDURANCE-3 ClinicalTrials.gov numbers, NCT02604017 and NCT02640157.)
Hepatitis C virus (HCV) genotypes 1 and 3 are together responsible for approximately 70% of an estimated 71 million to 80 million HCV infections worldwide.1,2 Direct-acting antiviral agents have become the standard-of-care treatment for chronic HCV infection and show high rates of sustained virologic response in most HCV genotypes.3 The efficacy of approved, direct-acting antiviral regimens with 12-week treatment durations ranges from 94 to 99% among patients with HCV genotype 1 infection.4-7 An 8-week course of treatment with sofosbuvir-ledipasvir may be considered for patients with genotype 1 infection who do not have cirrhosis and who have not previously received HCV treatment; however, treatment is restricted according to baseline viral load and fibrosis stage.8,9 For patients with HCV genotype 3 infection who do not have cirrhosis and have not previously received treatment, the standard of care is a 12-week course of sofosbuvir plus either daclatasvir10 or velpatasvir5; both regimens have yielded an overall rate of sustained virologic response of 98% at 12 weeks after treatment.11,12 Most approved regimens recommend 12 weeks of treatment for patients with HCV infection; however, patient adherence can decline during the last 4 weeks of treatment, which suggests that shorter treatment durations may improve adherence.13 Furthermore, a reduced-duration HCV regimen with pangenotypic activity that maintains high efficacy regardless of the characteristics of the patient or of the virus may increase the number of treated patients, especially in resource-limited settings.14
Glecaprevir is a nonstructural (NS) protein 3/4A protease inhibitor that was identified by AbbVie and Enanta; it is coformulated with pibrentasvir, an NS5A inhibitor. The in vitro half-maximal effective concentrations of glecaprevir and pibrentasvir range from 0.85 to 2.8 nmol per liter and 1 to 5 pmol per liter, respectively, across HCV genotypes 1 through 6. Together, these direct-acting antivirals have pangenotypic, synergistic anti-HCV activity with a high barrier to resistance, minimal metabolism, primarily biliary excretion, and negligible renal excretion.15,16 Phase 2 trials of glecaprevir (300 mg) plus pibrentasvir (120 mg) for 8 or 12 weeks in patients without cirrhosis who were infected with HCV genotype 1, 2, 3, 4, 5, or 6 showed rates of sustained virologic response of more than 93% at 12 weeks after treatment, including a rate of 97% among patients with genotype 1 or 3 infection who were treated for 8 weeks.17
In ENDURANCE-1 (A Study to Evaluate the Efficacy and Safety of ABT-493/ABT-530 in Subjects with Genotype 1 Infection), which involved HCV genotype 1-infected patients, including those with human immunodeficiency virus type 1 [HIV-1] coinfection, and ENDURANCE-3 (A Study Comparing Efficacy and Safety of ABT-493/ABT-530 to Sofosbuvir Dosed With Daclatasvir in Adults With HCV Genotype 3 Infection), which involved HCV genotype 3-infected patients who had not previously received treatment for HCV infection, we evaluated the efficacy and safety of an 8-week or 12-week course of treatment with ribavirin-free, once-daily coformulated glecaprevir-pibrentasvir in patients without cirrhosis. In HCV genotype 3-infected patients, a 12-week course of treatment with glecaprevir-pibrentasvir was also compared with a 12-week course of treatment with sofosbuvir-daclatasvir.
Results
Patients and Treatment
In total, 1410 patients were screened between October 21, 2015, and May 4, 2016; 200 patients were excluded from the trial at screening, predominantly because of unmet eligibility criteria (185 of 200 patients; 92%) (Figure 1, and Fig. S1 in the Supplementary Appendix). Overall, 1053 patients underwent randomization, and 1051 of these patients were treated: 351 HCV genotype 1-infected patients received glecaprevir-pibrentasvir for 8 weeks, 352 genotype 1-infected patients and 233 genotype 3-infected patients received glecaprevir-pibrentasvir for 12 weeks, and 115 genotype 3-infected patients received sofosbuvir-daclatasvir for 12 weeks. An additional 157 genotype 3-infected patients received glecaprevir-pibrentasvir for 8 weeks without undergoing randomization.
The characteristics of the patients at baseline were generally similar among the treatment groups (Table 1 and Table 2). The prevalence of stage F3 fibrosis was higher among the genotype 3-infected patients in the 8-week glecaprevir-pibrentasvir group than among patients in the other treatment groups (17%, as compared with 8 to 9%). The prevalence of patients infected with genotype 3 who had baseline HCV RNA levels of 6 million IU per milliliter or higher was higher in the 12-week glecaprevir-pibrentasvir group than in the sofosbuvir-daclatasvir group (28% vs. 12%). A total of 33 patients coinfected with HCV genotype 1 and HIV-1 were enrolled.
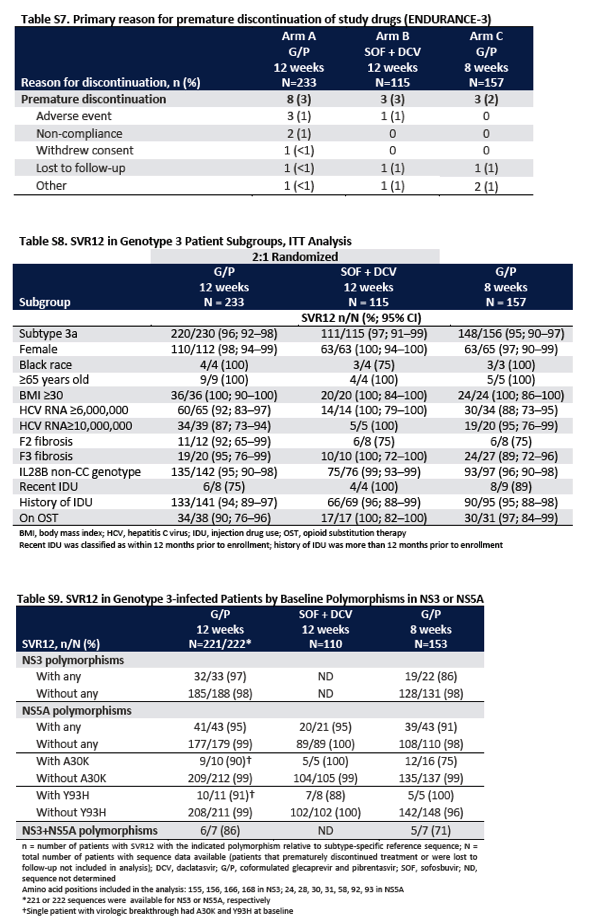
Among patients infected with HCV genotype 1, the prevalence of polymorphisms in NS3 or NS5A was similar in the 8-week and 12-week treatment groups. Genotype 3-infected patients who were treated for 8 weeks had a higher rate of NS5A polymorphisms at baseline than did those who were treated for 12 weeks (Table 2, and Table S4 in the Supplementary Appendix). The most common NS5A polymorphisms were at positions A30 and Y93. Among patients infected with genotype 3, the prevalence of the A30K variant was 10% in the 8-week glecaprevir-pibrentasvir group, as compared with 5% among the patients receiving 12 weeks of treatment with glecaprevir-pibrentasvir and 4% among patients receiving 12 weeks of treatment with sofosbuvir-daclatasvir, and the prevalence of the Y93H variant was 5% in the 12-week glecaprevir-pibrentasvir group, 7% in the 12-week sofosbuvir-daclatasvir group, and 3% in the 8-week glecaprevir-pibrentasvir group.
Efficacy among Patients with HCV Genotype 1 Infection
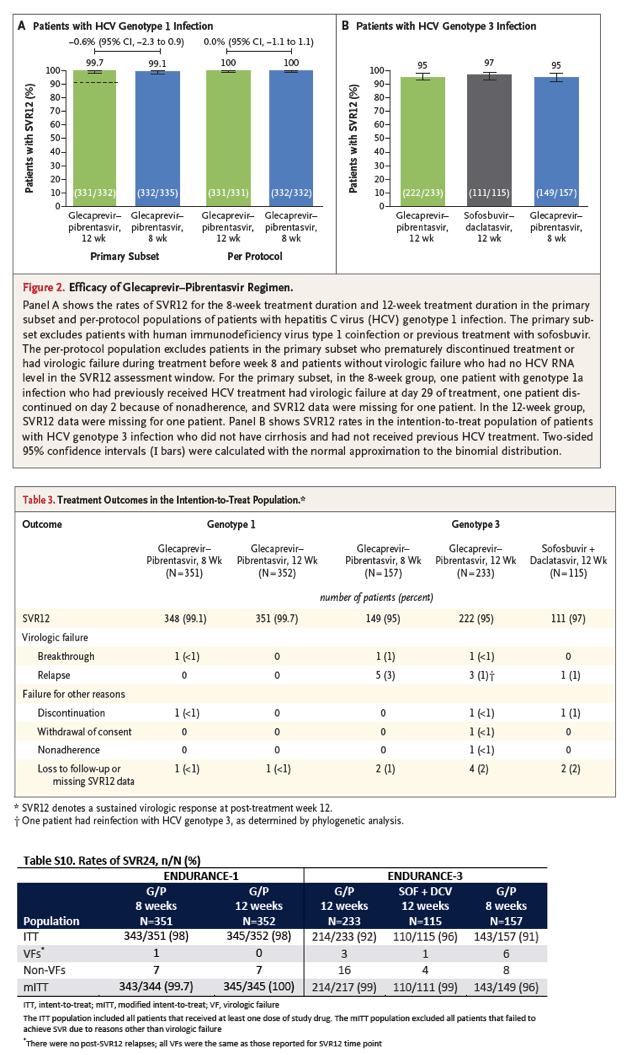
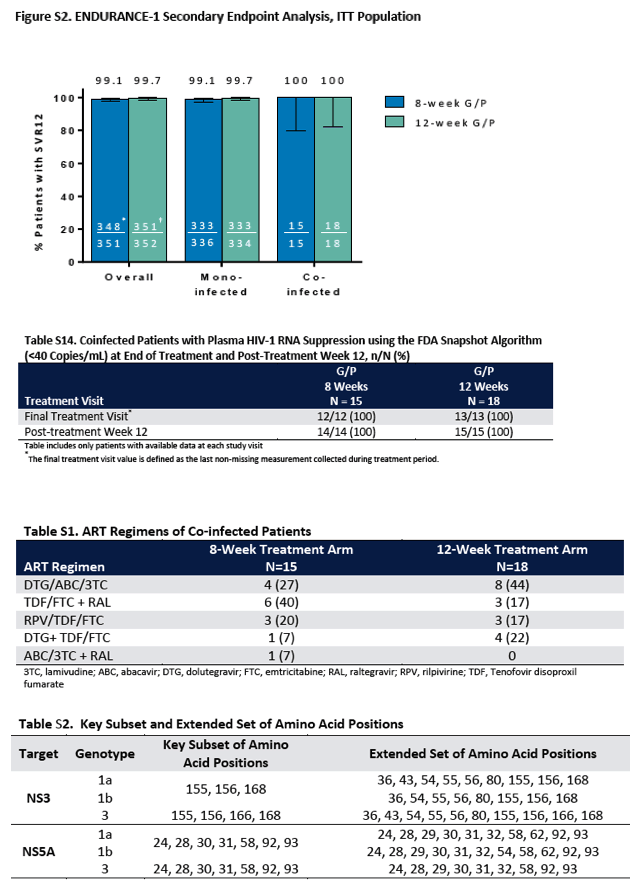
Of the 703 genotype 1-infected patients, 1 had breakthrough infection during treatment (the patient was enrolled in the 8-week treatment group; details are provided in Table S5 in the Supplementary Appendix); there were no relapses. In the primary subset of patients, the rate of sustained virologic response at 12 weeks was 99.1% (95% confidence interval [CI], 98 to 100) in the 8-week group and 99.7% (95% CI, 99 to 100) in the 12-week group. The rate of sustained virologic response at 12 weeks in the per-protocol population was 100% regardless of treatment duration (Figure 2A). All patients with HIV-1 coinfection (33 patients) or previous sofosbuvir treatment (3 patients) had a sustained virologic response at week 12 (Fig. S2 in the Supplementary Appendix).
The results of the three ranked analyses of the primary efficacy end point in this trial all indicated that the 8-week treatment duration was noninferior to the 12-week treatment duration. Noninferiority of the 12-week treatment regimen to the historical control (first ranked primary analysis) was shown by the 95% lower confidence limit for the rate of sustained virologic response at week 12 (99.7%; 95% CI, 99 to 100) in the primary subset exceeding 91%. The noninferiority of the 8-week regimen to the 12-week regimen was shown by the 95% lower confidence bound for the difference in rates of sustained virologic response at week 12 falling above -5 percentage points in both the per-protocol population (difference, 0.0 percentage points; 95% CI, -1.1 to 1.1; second ranked primary analysis) and the primary subset (difference, -0.6 percentage points; 95% CI, -2.3 to 0.9; third ranked primary analysis).
Rates of sustained virologic response at 12 weeks according to baseline factors such as HCV subtype, NS3 or NS5A polymorphisms, plasma HCV RNA level, fibrosis stage, and treatment history are shown in Table S6 in the Supplementary Appendix. The rate of sustained virologic response at 12 weeks among genotype 1-infected patients with a baseline plasma HCV RNA level higher than 6 million IU per milliliter was 100% (95% CI, 93 to 100; 49 of 49 patients) in the 8-week group and 98% (95% CI, 88 to 99.6; 42 of 43 patients) in the 12-week group. There were no relapses between post-treatment week 12 and post-treatment week 24 (final study visit) (Table S10 in the Supplementary Appendix).
Efficacy among Patients with HCV Genotype 3 Infection
Among HCV genotype 3-infected patients, glecaprevir-pibrentasvir treatment for 12 weeks resulted in a rate of sustained virologic response of 95% (95% CI, 93 to 98; 222 of 233 patients), for which the lower bound of the 95% confidence interval was higher than 92%; sofosbuvir-daclatasvir for 12 weeks resulted in a rate of 97% (95% CI, 93 to 99.9; 111 of 115 patients) (Figure 2B). The difference in the rate of sustained virologic response at 12 weeks between the 12-week glecaprevir-pibrentasvir group and the 12-week sofosbuvir-daclatasvir group was -1.2 percentage points (95% CI, -5.6 to 3.1), for which the lower bound of the 95% confidence interval was higher than -6 percentage points. These results showed that the 12-week glecaprevir-pibrentasvir regimen was noninferior to the 12-week regimen of sofosbuvir-daclatasvir. The 8-week glecaprevir-pibrentasvir group had a rate of sustained virologic response at 12 weeks of 95% (95% CI, 91 to 98; 149 of 157 patients). Although the lower bound of the 95% confidence interval in the 8-week glecaprevir-pibrentasvir group was not higher than 92%, according to the prespecified hierarchical testing procedure, noninferiority of the 8-week regimen to the 12-week regimen of glecaprevir-pibrentasvir was shown because the difference in rates between the two groups was -0.4 percentage points (97.5% CI, -5.4 to 4.6), for which the lower bound of the 97.5% confidence interval was above -6 percentage points.
We did not test the superiority of 12 weeks of glecaprevir-pibrentasvir to 12 weeks of sofosbuvir-daclatasvir because of the fixed-sequence statistical procedure, which required both noninferiority criteria to be met for the comparison between the 8-week and 12-week regimens of glecaprevir-pibrentasvir in order to proceed to testing the next ordered comparison. The difference in rates of virologic relapse after 8 weeks and 12 weeks of treatment (3% and 1%, respectively) was 2.0 percentage points, for which the 95% confidence interval overlapped zero (95% CI, -1.2 to 6.3); details for all patients with virologic failure are provided in Table S5 in the Supplementary Appendix. One patient with HCV genotype 3a infection who was treated for 12 weeks with glecaprevir-pibrentasvir had reinfection, as determined by phylogenetic analysis. The reasons for a lack of a sustained virologic response at 12 weeks are shown in Table 3, the reasons for premature discontinuation of treatment are shown in Table S7 in the Supplementary Appendix, and the rates of sustained virologic response at 12 weeks according to baseline factors are shown in Table S8 in the Supplementary Appendix. There were no relapses between post-treatment week 12 and post-treatment week 24 (final study visit) (Table S10 in the Supplementary Appendix).
Glecaprevir-pibrentasvir treatment for 12 weeks yielded a rate of sustained virologic response at 12 weeks of 95% (95% CI, 85 to 99; 41 of 43 patients) among patients with baseline NS5A polymorphisms and 99% (95% CI, 96 to 99.7; 177 of 179 patients) among patients without such polymorphisms (Table S9 in the Supplementary Appendix). Among patients treated with glecaprevir-pibrentasvir for 8 weeks, the rates of sustained virologic response at 12 weeks were 91% (95% CI, 78 to 96; 39 of 43 patients) for patients with NS5A polymorphisms at baseline and 98% (95% CI, 94 to 99.5; 108 of 110 patients) for patients without NS5A polymorphisms at baseline. The rates of sustained virologic response at 12 weeks among patients with the Y93H variant at baseline were 100% (5 of 5 patients) in the 8-week glecaprevir-pibrentasvir group, 91% (95% CI, 62 to 98; 10 of 11 patients) in the 12-week glecaprevir-pibrentasvir group, and 88% (7 of 8 patients) in the 12-week sofosbuvir-daclatasvir group; among patients with the A30K variant at baseline, the rates were 75% (95% CI, 51 to 90; 12 of 16 patients) in the 8-week glecaprevir-pibrentasvir group, 90.0% (95% CI, 60 to 98; 9 of 10 patients) in the 12-week glecaprevir-pibrentasvir group, and 100% (5 of 5 patients) in the 12-week sofosbuvir-daclatasvir group.
Safety
The safety profile of glecaprevir-pibrentasvir was similar among patients treated for 8 or 12 weeks (Table 4, and Table S11 in the Supplementary Appendix). The most common adverse events (occurring in at least 10% of patients) were headache and fatigue. Across both trials, two patients prematurely discontinued trial treatment because of adverse events that were assessed by the investigators as having a reasonable possibility of being related to direct-acting antivirals: one patient (genotype 3, 12-week glecaprevir-pibrentasvir group) discontinued treatment because of abdominal pain, headache, and malaise, and one patient (genotype 3, 12-week sofosbuvir-daclatasvir group) discontinued because of headache. Three additional patients discontinued trial treatment because of adverse events that were assessed by the investigators as unlikely to be related to direct-acting antivirals. Three patients died during the post-treatment period of the trials: two patients died from heroin overdoses, and one died from acute ethanol intoxication and combined methadone toxicity; all three events were deemed unrelated to the trial drugs. Across both trials, serious adverse events were reported in 1 to 2% of patients in each treatment group; none were deemed related to the trial drugs (Table S12 in the Supplementary Appendix).
Clinically significant laboratory abnormalities occurred in less than 1% of patients (Table 4, and Table S13 in the Supplementary Appendix). Four patients had grade 3 elevations in total bilirubin levels; all elevations predominantly involved indirect bilirubin and were not accompanied by increases in alanine aminotransferase levels. None of the patients who were treated with glecaprevir-pibrentasvir had postnadir elevations in alanine aminotransferase levels that were grade 3 or higher (i.e., to >5 times the upper limit of the normal value). All HIV-1-coinfected patients had continued suppression of HIV-1 RNA levels (to <200 copies per milliliter) during the treatment period, and none required a change in baseline antiretroviral therapy regimen. More information on the cohort of patients coinfected with HIV-1 and HCV is provided in Tables S14 through S18 in the Supplementary Appendix.
Discussion
Although the current standard-of-care treatments for patients without cirrhosis who are infected with HCV genotype 1 or genotype 3 result in high rates of sustained virologic response,4-7,11,12 patients could benefit from shorter-duration treatment options that maintain high efficacy irrespective of HCV genotype. Until recently, there were no approved treatment regimens with a duration of less than 12 weeks for patients with HCV genotype 3 infection, and 8-week treatment options for patients with genotype 1 infection were limited according to virus subtype, treatment history, fibrosis stage, or baseline viral load.8,9 In the present trials, 8 weeks of treatment with glecaprevir-pibrentasvir resulted in high rates of sustained virologic response at 12 weeks among patients without cirrhosis who had HCV genotype 1 infection (>99%) or genotype 3 infection (95%). A treatment duration of 8 weeks was noninferior to a duration of 12 weeks with regard to the rate of sustained virologic response at 12 weeks.
Glecaprevir-pibrentasvir treatment was associated with low rates of serious adverse events or clinically significant laboratory abnormalities; there were no elevations of grade 3 or higher in alanine aminotransferase levels. Adverse events that have historically been attributed to HCV protease inhibitors, such as severe rash, decreased neutrophil count, anemia, and elevated alanine aminotransferase levels, were not observed with glecaprevir-pibrentasvir treatment. Although protease inhibitors such as paritaprevir or simeprevir have been associated with hyperbilirubinemia,22across both trials, 4 of 1093 patients (0.4%) who were treated with glecaprevir-pibrentasvir had isolated grade 3 increases in total bilirubin level (all predominantly involving indirect bilirubin), a frequency lower than with other HCV protease inhibitors.22-24 Overall, the frequency and severity of adverse events observed with the NS3/4A protease inhibitor-containing glecaprevir-pibrentasvir regimen was similar to that observed with the NS5B nucleotide polymerase inhibitor-containing regimen of sofosbuvir-daclatasvir. All patients who were coinfected with HIV-1 and HCV had continued HIV-1 suppression throughout the trial.
The ability to shorten treatment duration without compromising efficacy remains an important goal in therapy for all HCV genotypes. In addition to the results with 8 weeks of treatment for patients with HCV genotype 1 or genotype 3 from these two trials, recent phase 3 trials have shown that an 8-week regimen of treatment with glecaprevir-pibrentasvir in patients without cirrhosis who have HCV genotype 2 infection or genotype 4, 5, or 6 infection yielded rates of sustained virologic response at 12 weeks of 98% and 93% (with no virologic failures), respectively.25-27
These trials were not designed or powered to formally compare responses across subgroups defined according to baseline characteristics. Nonetheless, in ENDURANCE-1, there were no relapses among HCV genotype 1-infected patients who were treated for 8 weeks, regardless of their baseline HCV RNA level, and glecaprevir-pibrentasvir treatment yielded high rates of sustained virologic response among genotype 1-infected patients regardless of baseline characteristics that have historically been associated with lower efficacy.28
In ENDURANCE-3, efficacy was assessed with respect to the presence of NS5A polymorphisms at baseline. NS5A polymorphisms, such as A30K and Y93H, can have important clinical implications, particularly for patients infected with HCV genotype 3, because they confer a high degree of resistance to currently available NS5A inhibitors and have been associated with lower rates of response to treatment. For example, the HCV genotype 3 Y93H polymorphism decreases the susceptibility of the virus to velpatasvir and daclatasvir, resulting in half-maximal effective concentrations that are increased by factors greater than 100 and 3700, respectively5,29; the A30K polymorphism reduces the susceptibility of the virus to daclatasvir, resulting in a half-maximal concentration that is increased by a factor of 117.29 In contrast, single HCV genotype 3a A30K and Y93H polymorphisms or substitutions confer minimal decreases in susceptibility to pibrentasvir in vitro (with respective half-maximal concentrations increased by a factor of <2.5).15,30 In ENDURANCE-3, among patients with the Y93H variant at baseline, 15 of 16 patients (94%; 95% CI, 72 to 99) treated with glecaprevir-pibrentasvir for 8 or 12 weeks had a sustained virologic response at 12 weeks, including 5 of 5 patients (100%) who were treated for 8 weeks. Genotype 3-infected patients who were treated with glecaprevir-pibrentasvir and who had the A30K variant in NS5A at baseline had a lower rate of sustained virologic response at 12 weeks than patients without A30K (81% [21 of 26] vs. 99% [344 of 349]). However, the majority of patients who were treated with glecaprevir-pibrentasvir had a sustained virologic response regardless of the presence of A30K. The low prevalence of the genotype 3 A30K variant reported in this trial (<7% overall) and other trials (4.5 to 6%31,32) makes the true importance of this factor with regard to overall efficacy difficult to assess. Despite the higher prevalence of patients with the HCV genotype 3 A30K variant at baseline in the 8-week glecaprevir-pibrentasvir treatment group, the 8-week glecaprevir-pibrentasvir regimen achieved noninferiority to the 12-week regimen in patients with genotype 3 infection.
Two patients with genotype 1 infection and 10 patients with genotype 3 infection did not have a sustained virologic response at 12 weeks because of nonadherence to the trial regimen, withdrawal of informed consent, or loss to follow-up. Specifically, the frequency of treatment failure for nonvirologic reasons among patients with genotype 3 infection appeared to be higher than the frequency reported in phase 3 trials involving similar populations.11,12 The patients infected with HCV genotype 1 and those infected with HCV genotype 3 in our trials differed with regard to several baseline characteristics: patients with genotype 3 infection were younger (median age, 47 to 49 years vs. 52 to 53 years), more likely to be receiving opiate substitution therapy (15 to 20% vs. 3 to 4%), and more likely to be current injection drug users (3 to 6% vs. 1%) than patients with HCV genotype 1 infection. It is notable that 65% of enrolled patients with genotype 3 infection had a history of injection drug use, which is in line with the reported high prevalence of people who inject drugs among those who have HCV genotype 3 (approximately 50 to 65%).33-36
There are some limitations to these trials. Despite the fact that the same eligibility criteria, similar trial sites, and similar sample-size considerations were used in the two trials, the lack of random assignment to the treatment groups for the comparison of the 8-week and 12-week durations of glecaprevir-pibrentasvir treatment in genotype 3-infected patients may explain the imbalances in baseline conditions described above and could have affected the efficacy assessment. Another potential limitation was the open-label nature of ENDURANCE-3, which could have biased the safety comparison between the treatment groups. A third limitation was the small number of patients enrolled in ENDURANCE-1 who had previously received sofosbuvir. However, patients with treatment-associated substitutions after sofosbuvir treatment failure are not expected to be harder to cure than other patients in this trial, given the nonoverlapping mechanism of action of sofosbuvir with those of glecaprevir and pibrentasvir. Finally, there were small numbers of patients in some of the genotype 1 subgroups, particularly the subgroups of patients with F3 fibrosis, HIV-1 coinfection, and black race.
Treatment with glecaprevir-pibrentasvir for 8 weeks yielded rates of sustained virologic response at 12 weeks of 99% and 95% in patients without cirrhosis who had HCV genotype 1 infection or genotype 3 infection, respectively, which indicated that 8 weeks of treatment with glecaprevir-pibrentasvir is an efficacious treatment option for HCV genotype 1 or 3 infection.
Methods
Trial Design
We conducted two phase 3, open-label, multicenter trials (Figure 1). Patients in ENDURANCE-1 were randomly assigned in a 1:1 ratio to receive glecaprevir-pibrentasvir for 8 or 12 weeks. The treatment assignments were not concealed. Randomization was computer-generated and stratified according to screening plasma HCV RNA level and HCV genotype 1 subtype, which were considered potential prognostic factors.
ENDURANCE-3 had an active-controlled, partially randomized design: patients were randomly assigned in a 2:1 ratio to receive either glecaprevir-pibrentasvir or 400 mg of sofosbuvir plus 60 mg of daclatasvir for 12 weeks. After additional phase 2 data that supported the efficacy of an 8-week treatment duration became available, a subsequent protocol amendment enabled the nonrandom assignment of patients into a third group for 8 weeks of treatment with glecaprevir-pibrentasvir. In both trials, glecaprevir-pibrentasvir was administered orally once daily as three tablets (each containing 100 mg of glecaprevir and 40 mg of pibrentasvir), for a total daily dose of 300 mg of glecaprevir and 120 mg of pibrentasvir.
Trial Oversight
All the patients provided written informed consent. The trials were designed in accordance with Good Clinical Practice guidelines, the Declaration of Helsinki, and applicable local regulations, with approval from an independent ethics committee or institutional review board for all trial sites. The sponsor (AbbVie) conducted the data analyses, and the authors had full access to the data; the authors vouch for the accuracy and completeness of the data and for the fidelity of the trials to the protocols, which are available with the full text of this article at NEJM.org. The investigators, participating institutions, and sponsor agreed to maintain the confidentiality of the data. The first draft of the manuscript was generated by all the authors, with assistance from sponsor-employed medical writers.
Patients
Patients who were at least 18 years of age were eligible for participation in the trials if they had chronic HCV genotype 1 or 3 infection with an HCV RNA level higher than 1000 IU per milliliter at screening. Patients with HCV genotype 1 infection could have HIV-1 coinfection and could either have never received HCV treatment or have received treatment with interferon or pegylated interferon with or without ribavirin or treatment with sofosbuvir plus ribavirin with or without pegylated interferon. Patients with genotype 3 infection had to have never received HCV treatment. The absence of cirrhosis in all patients was documented by means of liver biopsy, assessment of serum markers, or transient elastography. Patients who were positive for hepatitis B surface antigen or coinfected with more than one HCV genotype were excluded. Detailed eligibility criteria are provided in the Supplementary Appendix, available at NEJM.org.
Assessments
Plasma HCV RNA was quantified for each sample with the use of the COBAS Ampliprep/TaqMan real-time reverse-transcriptase polymerase-chain-reaction assay, version 2.0 (Roche Molecular Diagnostics), which has a lower limit of quantification of 15 IU per milliliter. Next-generation sequencing was conducted on plasma samples collected from all patients at baseline, and the presence of polymorphisms in NS3 and NS5A at baseline was evaluated with the use of a 15% detection threshold. For patients who had virologic failure, substitutions (relative to the patient’s baseline HCV sequence) that arose during treatment were analyzed (Table S2 in the Supplementary Appendix). Details on genotype and subtype determination, HCV resistance analysis, and monitoring of HIV-1 suppression and of lymphocytes and lymphocyte subsets for HIV-1 coinfected patients are provided in the Supplementary Appendix. Adverse events and clinical laboratory abnormalities were monitored throughout the trial, and safety analyses were performed in the intention-to-treat population.
End Points
The primary efficacy end point for both trials was sustained virologic response at week 12 after treatment, defined as an HCV RNA level below 15 IU per milliliter 12 weeks after the last dose of trial drug without any confirmed quantifiable post-treatment HCV RNA level through the post-treatment week 12 visit. A full list of the secondary efficacy end points is provided in Table S3 in the Supplementary Appendix. Secondary end points included the percentage of patients with virologic failure during treatment (defined as a confirmed increase in HCV RNA level of >1 log10 IU per milliliter above the nadir during treatment, confirmed HCV RNA level ≥100 IU per milliliter after the level had been <15 IU per milliliter during treatment, or an HCV RNA level ≥15 IU per milliliter at the end of treatment [with at least 6 weeks of treatment]) and post-treatment relapse (defined as a confirmed HCV RNA level ≥15 IU per milliliter between the end of treatment and 12 weeks after the last dose of trial drug among patients who both completed treatment and had an HCV RNA level of <15 IU per milliliter at the end of treatment [excluding reinfection]).
Statistical Analyses
In ENDURANCE-1, a fixed-sequence testing procedure was used to conduct three sequentially ranked primary efficacy analyses in the primary subset of the intention-to-treat population, defined as all patients who underwent randomization and who received at least one dose of trial drug, excluding those who had HIV coinfection or who had previously received sofosbuvir treatment. Only if success had been shown for the first primary analysis would the second primary analysis proceed, and only if success had been shown for the second primary analysis would the third primary analysis proceed. Under the assumption of a rate of sustained virologic response of 97% at 12 weeks, we calculated that at least 270 patients monoinfected with HCV genotype 1 who had not received previous treatment with sofosbuvir would be required in each treatment group to provide more than 90% power for the analyses. The first-ranked primary efficacy analysis was the analysis of the efficacy of the 12-week treatment duration, wherein the lower bound of the two-sided 95% confidence interval, calculated with the use of the normal approximation to the binomial distribution for the percentage of patients treated for 12 weeks who had a sustained virologic response at week 12, was compared with a threshold rate of 91%. The threshold was determined with the use of a 6-percentage-point margin, on the basis of the historical rate of sustained virologic response of 97% that has been observed among HCV genotype 1-infected, HCV monoinfected patients who had not previously received direct-acting antiviral treatment who were treated for 12 weeks.4,6
The second-ranked primary efficacy analysis was a noninferiority assessment of an 8-week treatment duration relative to a 12-week treatment duration with regard to the rate of sustained virologic response at 12 weeks. This analysis was conducted with the use of a noninferiority margin of 5 percentage points in the per-protocol population, which excluded patients in the primary subset who prematurely discontinued treatment or had virologic failure before week 8 of treatment and patients without virologic failure for whom data on HCV RNA level during the window for assessment of sustained virologic response at 12 weeks were missing. Since patients in both treatment groups received the same regimen during the first 8 weeks, treatment failure for any reason before week 8 could not be attributed to treatment duration and thus would not inform the noninferiority analysis. If the lower bound of the confidence interval (calculated with the use of the normal approximation to the binomial distribution) for the difference in rates of sustained virologic response at 12 weeks (i.e., the rate in the 8-week group minus the rate in the 12-week group) was above the noninferiority margin of -5 percentage points, then the 8-week regimen would be considered noninferior to the 12-week regimen. To support the second-ranked primary efficacy analysis of noninferiority of the 8-week to the 12-week treatment duration, the third-ranked primary efficacy analysis was the same analysis, conducted in the more inclusive primary subset of patients, which included the patients who were excluded from the per-protocol population.
Sensitivity analyses were performed for the primary efficacy end point of the rate of sustained virologic response at 12 weeks, in which differences in proportions of patients with sustained virologic response at 12 weeks between the 12-week and 8-week groups across strata were calculated on the basis of stratum-adjusted Mantel-Haenszel proportions. The rate of sustained virologic response at week 12 among all HCV genotype 1-infected patients was also assessed in the intention-to-treat population and in patient subgroups with HIV-1 coinfection, HCV monoinfection, or previous sofosbuvir treatment.
ENDURANCE-3 was designed to have 90% power to evaluate the noninferiority of a 12-week regimen of glecaprevir-pibrentasvir to a 12-week regimen of sofosbuvir-daclatasvir, with the use of a noninferiority margin of 6 percentage points in the intention-to-treat analyses. During enrollment for the two randomly assigned treatment groups, phase 2 efficacy data became available that showed a 97% rate of sustained virologic response at 12 weeks among patients infected with HCV genotype 3 after 8 weeks of treatment with glecaprevir-pibrentasvir17; in consultation with regulatory agencies, we added an 8-week glecaprevir-pibrentasvir treatment group to which patients were nonrandomly assigned. We used the same eligibility criteria for this treatment group, and sample-size considerations for direct statistical comparisons with the two 12-week treatment groups were taken into account. All statistical tests were performed in the intention-to-treat population, defined as all patients who received at least one dose of trial drug. Statistical comparisons between the 12-week treatment groups and the 8-week group were added to the protocol and statistical analysis plan and approved by regulatory agencies. Noninferiority of the 8-week regimen to the 12-week regimen of glecaprevir-pibrentasvir with regard to the rate of sustained virologic response at 12 weeks was shown with 80% power if the lower bound of the confidence interval for the differences was above the noninferiority margin of -6 percentage points or if the lower bound of the confidence interval for the within-group rate was higher than 92%. This threshold corresponds to the historical 98% rate of sustained virologic response at 12 weeks associated with sofosbuvir-daclatasvir in the same population, minus the 6-percentage-point noninferiority margin.11 To control the type I error rate, the fixed-sequence testing procedure and Hochberg procedure18 were used during the primary efficacy analyses; details on the use of this procedure in the trial are provided in the Supplementary Appendix.
For both trials, noninferiority margins of 5 or 6 percentage points were selected in consultation with regulatory agencies and in accordance with the principles outlined in guidance on conducting noninferiority trials; the choice of margins was justified on the basis of perceived medical benefit over historical controls and ensured minimal to no loss of efficacy.19-21 More details on the multiplicity adjustment, the study population size, the noninferiority margin, and the calculation of confidence intervals are provided in the Supplementary Appendix.
SAS software, version 9.3, for the UNIX operating system (SAS Institute) was used for all descriptive statistics and analysis. Data were analyzed by the AbbVie Statistics and Statistical Programming and Clinical Pharmacology and Pharmacometrics Departments.
| |
| |
| |
|
|
|