| |
Among SVR patients, recent injection cocaine use was associated with rapid eGFR decline
["Hepatitis C virus cure does not impact kidney function decline in HIV co-infected patients"]
|
| |
| |
Download the PDF here
AIDS March 27 2018 - Rossi, Carminea; Saeed, Sahara; Cox, Josephb; Vachon, Marie-Louisec; Martel-laferriere, Valeried; Walmsley, Sharon, L.e; Cooper, Curtisf; Gill, M., Johng; Hull, Markh; Moodie, Erica, E.M.i; Klein, Marina, B.a,bfor the Canadian Co-Infection Cohort Investigators
"Results: We identified 384 co-infected patients who achieved SVR (53% treated with interferon-free antiviral regimens) and 768 propensity-score matched patients with chronic HCV infection. Most patients were men (78%) and white (87%), with a median age of 51 years (interquartile range: 45-56). During 1767 person-years of follow-up, 4041 eGFR measurements were available for analysis. Annual rates of decline in eGFR were similar between patients with SVR [-1.32 (ml/min per 1.73 m2)/year, 95% confidence interval (CI) -1.75 to -0.90] and chronic infection [-1.19 (ml/min per 1.73 m2) per year, 95% CI -1.55 to -0.84]. Among SVR patients, recent injection cocaine use was associated with rapid eGFR decline [-2.16 (ml/min per 1.73 m2)/year, 95% CI -4.17 to -0.16].
Conclusion: SVR did not reduce the rate of kidney function decline among HCV-HIV co-infected patients. Increased risk of chronic kidney disease in co-infection may not be related to persistent HCV replication but to ongoing injection cocaine use."
Fig. 1. Kidney function trajectories in the reference population, by illicit drug use, among patients with sustained virologic
response (nU384). eGFR, estimated glomerular filtration rate; SVR, sustained virologic response.
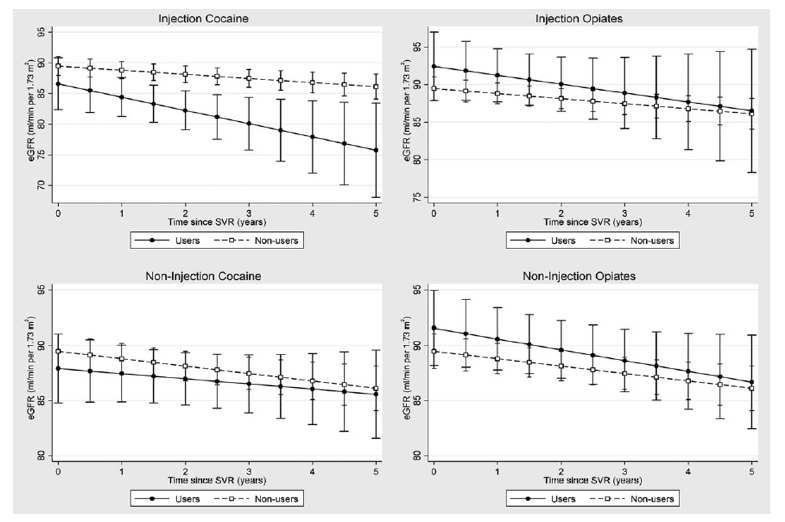
"Among the 384 patients who achieved SVR, 43 (11%) and 46 (12%) patients reported recent injection cocaine and injection opiate use within the last 6 months, respectively, and 61 (16%) and 58 (15%) reported recent noninjection crack/cocaine and opiate use, respectively. After SVR, the adjusted annual rate of change in eGFR was faster among active injection cocaine users [-2.16 (ml/min per 1.73 m2)/year, 95% CI -4.17 to -0.16], as compared with nonusers of any illicit drug [-0.67 (ml/min per 1.73 m2)/year, 95% CI -1.14 to -0.20] but was not statistically significant [difference: -1.49 (ml/min per 1.73 m2)/year, 95% CI -3.49 to 0.51]. No similar accentuation in decline was observed among the other illicit drugs (Supplemental Table 2, http://links.lww.com/QAD/B216). Analyses of eGFR trajectories demonstrated that in the reference population, after 5 years, injection cocaine users will have a lower eGFR (76 ml/min per 1.73 m2, 95% CI 68-83), compared with nonusers of any illicit drug (86 ml/min per 1.73 m2, 95% CI 84-88). There were no significant differences in eGFR trajectories between other illicit drug users and nonusers (Fig. 1)."
Objective: To examine the impact of sustained virologic response (SVR) and illicit (injection and noninjection) drug use on kidney function among hepatitis C virus (HCV) and HIV co-infected individuals.
Design: Longitudinal observational cohort study of HCV-HIV co-infected patients.
Methods: Data from 1631 patients enrolled in the Canadian Co-Infection Cohort between 2003 and 2016 were analyzed. Patients who achieved SVR were matched 1 : 2 with chronically infected patients using time-dependent propensity scores. Linear regression with generalized estimating equations was used to model differences in estimated glomerular filtration rates (eGFR) between chronic HCV-infected patients and those achieving SVR. The relationship between illicit drug use and eGFR was explored in patients who achieved SVR.
Results: We identified 384 co-infected patients who achieved SVR (53% treated with interferon-free antiviral regimens) and 768 propensity-score matched patients with chronic HCV infection. Most patients were men (78%) and white (87%), with a median age of 51 years (interquartile range: 45-56). During 1767 person-years of follow-up, 4041 eGFR measurements were available for analysis. Annual rates of decline in eGFR were similar between patients with SVR [-1.32 (ml/min per 1.73 m2)/year, 95% confidence interval (CI) -1.75 to -0.90] and chronic infection [-1.19 (ml/min per 1.73 m2) per year, 95% CI -1.55 to -0.84]. Among SVR patients, recent injection cocaine use was associated with rapid eGFR decline [-2.16 (ml/min per 1.73 m2)/year, 95% CI -4.17 to -0.16].
Conclusion: SVR did not reduce the rate of kidney function decline among HCV-HIV co-infected patients. Increased risk of chronic kidney disease in co-infection may not be related to persistent HCV replication but to ongoing injection cocaine use.
Results
Of the 1726 co-infected patients who were enrolled in the CCC since 2003, we excluded 42 spontaneous clearers and 53 patients who were previously treated and developed SVR (Supplemental Figure, http://links.lww.com/QAD/B216). Characteristics of the base cohort at enrollment, as well as those with spontaneous clearance and previous SVR are shown in Supplemental Table 1, http://links.lww.com/QAD/B216. During the study period, 562 patients were treated for chronic HCV. Of those treated, a treatment response was not available for 53 patients (9%), most of whom had just recently completed therapy (42/53; 79%). Of the 509 patients with known treatment responses, 45 (9%) had not yet completed any posttreatment study follow-up. Of the 457 patients with an available treatment result and posttreatment follow-up, 384 (84%) developed SVR.
Measured baseline covariate balance between patients who achieved SVR (n = 384) and propensity score-matched chronically infected patients (n = 768) are shown in Table 1. Covariates appeared balanced between the two populations, with no substantial imbalances between the two study groups. More than half of all patients in the SVR group were treated with an all-oral DAA regimen (202/384; 53%) and 70% had HCV genotype 1 (268/384).
In the primary analysis, we included 1152 HCV-HIV co-infected participants who were followed for a total of 1767 person-years of follow-up (PYFU). A total of 4041 post-SVR eGFR measurements were available for analysis. The mean follow-up time was 1.6 years in the SVR group and 1.5 years in the chronically infected group. Annual rates of decline in eGFR were similar between SVR [-1.32 (ml/min per 1.73 m2)/year, 95% confidence interval (CI) -1.75 to -0.90] and chronically infected patients [-1.19 (ml/min per 1.73 m2)/year, 95% CI -1.55 to -0.84; Table 2]. Our results were similar after additionally adjusting for the propensity score to account for measured baseline covariate imbalances. After including time-dependent covariates in the outcome model, rates of eGFR decline were slightly attenuated, though there remained no statistically or clinically significant difference between the two groups.
Among the 384 patients who achieved SVR, 43 (11%) and 46 (12%) patients reported recent injection cocaine and injection opiate use within the last 6 months, respectively, and 61 (16%) and 58 (15%) reported recent noninjection crack/cocaine and opiate use, respectively. After SVR, the adjusted annual rate of change in eGFR was faster among active injection cocaine users [-2.16 (ml/min per 1.73 m2)/year, 95% CI -4.17 to -0.16], as compared with nonusers of any illicit drug [-0.67 (ml/min per 1.73 m2)/year, 95% CI -1.14 to -0.20] but was not statistically significant [difference: -1.49 (ml/min per 1.73 m2)/year, 95% CI -3.49 to 0.51]. No similar accentuation in decline was observed among the other illicit drugs (Supplemental Table 2, http://links.lww.com/QAD/B216). Analyses of eGFR trajectories demonstrated that in the reference population, after 5 years, injection cocaine users will have a lower eGFR (76 ml/min per 1.73 m2, 95% CI 68-83), compared with nonusers of any illicit drug (86 ml/min per 1.73 m2, 95% CI 84-88). There were no significant differences in eGFR trajectories between other illicit drug users and nonusers (Fig. 1).
Among the 384 patients who achieved SVR, 43 (11%) and 46 (12%) patients reported recent injection cocaine and injection opiate use within the last 6 months, respectively, and 61 (16%) and 58 (15%) reported recent noninjection crack/cocaine and opiate use, respectively. After SVR, the adjusted annual rate of change in eGFR was faster among active injection cocaine users [-2.16 (ml/min per 1.73 m2)/year, 95% CI -4.17 to -0.16], as compared with nonusers of any illicit drug [-0.67 (ml/min per 1.73 m2)/year, 95% CI -1.14 to -0.20] but was not statistically significant [difference: -1.49 (ml/min per 1.73 m2)/year, 95% CI -3.49 to 0.51]. No similar accentuation in decline was observed among the other illicit drugs (Supplemental Table 2, http://links.lww.com/QAD/B216). Analyses of eGFR trajectories demonstrated that in the reference population, after 5 years, injection cocaine users will have a lower eGFR (76 ml/min per 1.73 m2, 95% CI 68-83), compared with nonusers of any illicit drug (86 ml/min per 1.73 m2, 95% CI 84-88). There were no significant differences in eGFR trajectories between other illicit drug users and nonusers (Fig. 1).
Discussion
In this analysis of HCV-HIV co-infected patients who were treated in both the interferon and interferon-free eras, we found that annual rates of change in eGFR were similar between those who were successfully treated and comparably matched patients who were chronically infected. These findings suggest that there is no immediate benefit of successful HCV treatment for kidney function and that HCV cure may not reduce the overall incidence of CKD in co-infected patients.
Our results differ from previous reports in cohort studies of HIV-negative populations. In a clinical cohort of 650 HCV mono-infected patients with cirrhosis treated with interferon in Japan, SVR was associated with a 63% reduction in CKD incidence [24]. Given this population was cirrhotic, this finding may be explained by the impact of SVR on liver disease progression, which reduces the risk of hepatorenal syndrome, and not specifically an effect of HCV elimination on renal function [35]. A large administrative-linked, population-based study in Taiwan reported that initiation of HCV treatment was associated with an 85% decline in ESRD incidence compared with untreated HCV-infected individuals [25,36]. This study excluded patients with advanced liver disease and specific comorbidities, such as those with psychiatric diagnoses, which would be a contraindication to interferon-based treatment and did not control for lifestyle risk factors, such as alcohol and drug use, which would be less common in people who initiate therapy with interferon. The protective association of interferon treatment may, therefore, be overestimated, though because of the large effect, further investigation is warranted.
Our findings, however, are consistent with two recent cohort studies examining the association between HCV viral eradication and CKD in co-infected populations treated in the interferon era. In an analysis of 340 HCV-HIV co-infected patients treated in Italy, Leone et al. [37] report that achieving a SVR was not associated with a shorter time to incident CKD. Similarly, an analysis of 2503 co-infected patients in the Swiss HIV Cohort Study found no evidence that successful treatment with interferon was associated with a reduced risk of ESRD, as compared with those who were chronically infected [38]. In contrast, a recent Spanish cohort study, which directly compared 1625 HCV-HIV co-infected patients with SVR to those who failed treatment in the interferon-era, reported a clinically significant protective effect of SVR on CKD [39]. The latter study, however, did not adjust for baseline eGFR and if low eGFR is an important predictor for failing to achieve SVR, then the non-SVR group would have shorter time to CKD and the protective effect of successful treatment may be overestimated [24].
HCV has been implicated as an important risk factor for metabolic, cardiovascular, kidney, and neurological extrahepatic manifestations [17,40]. Although the pathophysiologic effect of HCV on extrahepatic comorbidities are not completely understood, they are likely related to a combination of direct HCV effects, such as viral replication in extrahepatic cells, or immune activation resulting in chronic inflammation, and indirect effects such as drug and alcohol use, poor nutrition, and HIV-related issues [23]. For some comorbidities, such as diabetes, wherever HCV viral replication is known to directly affect insulin-signaling pathways[17], the effect of successful treatment has resulted in a reduction of clinical outcomes observed in several epidemiologic studies [38,39,41]. For other extrahepatic comorbidities, such as cardiovascular disease and CKD, the mechanisms are likely multifaceted and HCV viral eradication alone may not reduce the incidence of all clinical outcomes. For example, on the one hand, chronic HCV has been associated with favourable lipid profiles [42], but has also been associated with greater prevalence of atherogenic risk factors [43]. In epidemiologic studies, SVR has been strongly associated with a significant reduction in cerebrovascular events and acute coronary syndrome in a HCV mono-infected population [25,44], but produced conflicting results in HIV co-infected populations [38,39]. For kidney diseases, it is clear that HCV treatment reduces complications associated with mixed cryoglobulinemia, which is associated with the development of membranoproliferative glomerular nephritis (MPGN) disease, though it is less clear if HCV treatment has an impact on kidney function in patients without any underlying renal diseases [45].
There are several factors that may explain our null findings. First, given that most co-infected patients have been infected with HCV for more than two decades, it is possible that successful HCV treatment may have a limited impact on the kidney after HCV replication has continued unabated during this long period. This is unlikely, however, as most patients had normal kidney function (≥90 ml/min per 1.73 m2) at the time of SVR, suggesting that HCV itself may not be an important contributor to CKD in HIV-infected populations and that other exposures such as cocaine use may explain the increased risk of chronic renal impairment [20]. Second, it may be possible that SVR may have a stronger effect on kidney function among subpopulations of patients who are more likely to rapidly progress to ESRD after developing CKD, such as HIV-infected African Americans, who carry the APOL1 or MYH9 genetic variants, or patients with cirrhosis or diabetes [46]. We were underpowered, however, to test this hypothesis in our study.
Few studies have examined the relative contributions of HCV viremia, lifestyle factors, and alternative mechanisms to the development of CKD in patients with chronic viral infections [19-21]. We reported earlier that frequent and cumulative exposure to injection crack/cocaine was associated with chronic renal impairment in HCV-HIV co-infected patients, independent of HCV viral replication [20]. Garg et al. [19] have also reported an association between crack/cocaine use and acute kidney injury among a small cohort of chronic HCV-infected patients. Recently, in a large cohort of HCV mono-infected veterans and matched controls, Rogal et al. [21] found that both alcohol and drug dependence, as measured with administrative billing codes, were associated with an increased risk of CKD. We extend these findings to HIV co-infected patients who achieved SVR and note the increasing role that substance use, namely injection cocaine, has on short-term kidney function. Whether these effects persist after long-term substance use or whether kidney function normalizes after cessation remains to be investigated. This is the first study to examine the effect that lifestyle risk factors have on kidney function after SVR, and we encourage the development of further long-term studies assessing the impact of substance use on CKD and other extrahepatic illnesses in HIV or HCV-infected populations [23].
Our analysis adds to the developing body of literature on extrahepatic outcomes following successful HCV treatment in the co-infected population and has several strengths. First, we included a large number of patients who were treated with DAAs, which have replaced interferon-based therapy for both treatment-naive and treatment-experienced chronic HCV patients, regardless of genotype. Compared with previous research in this area, our study population is, then, more generalizable to those co-infected patients initiating care in low-incidence settings. Second, our use of time-dependent propensity scores reduces the impact that unmeasured confounding, which varies with time would have on influencing both why patients would initiate HCV treatment and subsequent changes in their kidney function. This is particularly important, as our research encompasses several distinct HCV treatment periods, with an increasing number of HCV-HIV co-infected patients with comorbid conditions initiating treatment. Lastly, our use of routinely measured eGFR values allows us to model annual rates of change in kidney function, which are clinically relevant for those treating HIV-infected patients on stable antiretroviral therapy. Furthermore, the use of repeated eGFR measurements allow us to increase statistical power, as compared with previous analyses, and reduce biases associated with irregular eGFR measurements leading to CKD diagnosis in patients at risk for the outcome.
Our study has several potential limitations. First, we were limited by a relatively short follow-up, particularly among those who were recently treated with new DAA regimens. Therefore, we had insufficient follow-up time to allow incident CKD events to occur. Second, we could not directly compare SVR patients with patients who failed treatment, given the small numbers of co-infected patients who failed (n = 73). Comparison of these two groups would provide a less biased estimate of the effect of SVR on kidney function, as it would control for factors related to treatment initiation, but such analyses have become increasingly difficult as HCV treatment becomes much more effective. Our method of using time-dependent propensity scores with incidence density sampling provided a matched sample with comparable baseline characteristics. It is possible, however, that unmeasured characteristics, such as poor nutrition or the use of concomitant nephrotoxic medications, may have been more prevalent in the chronically infected group and these factors may also have influenced eGFR [47] however, the groups were well balanced with respect to HIV disease stage, comorbid conditions, and recent hospitalizations suggesting exposure to such medications would likewise be balanced. Furthermore, we were unable to assess if urinary biomarkers of kidney disease, such as proteinuria, were differentially distributed between our study groups. Lastly, we relied on patient self-report to assess illicit drug-use exposures. Although this may be affected by social desirability biases, nurses and clinicians maintain good relationships with study participants in the CCC, yielding little incentive to falsely reporting data.
In conclusion, we found that SVR was not associated with a short-term reduction in the rate of change in eGFR among HCV-HIV co-infected patients receiving clinical care. Regardless, HCV treatment should continue to be promoted, given its clear benefits in reducing the risk of liver-disease progression, premature mortality, and HCV transmission. For co-infected individuals who achieved SVR, monitoring eGFR in those who continue to use cocaine and referral to substance-use services would be beneficial, given their higher risk of experiencing kidney injury over time.
| |
| |
| |
|
|
|