| |
|
SVR & Comorbidities [Kidney/CVD/Bone/Cancer/Diabetes] in HCV/HIV+ - Hepatitis C Infection and the Risk of Non-Liver-Related Morbidity and Mortality in HIV-Infected Persons in the Swiss HIV Cohort Study
|
| |
| |
Download the PDF here
CROI-2016: Hepatitis C and the Risk of Non-Liver-Related Morbidity and Mortality in HIV+ Persons....kidney, bone, cardiovascular - (04/4/16)
"Compared with HCV-negative controls, HCV-seropositive participants had an increased risk of liver events (incidence rate ratio [IRR], 6.29 [95% confidence interval {CI}, 3.52-11.22]), liver-related death (IRR, 8.24 [95% CI, 3.61-18.83]), kidney events (IRR, 2.43 [95% CI, 1.11-5.33]), and osteoporosis/fracture (IRR, 1.43 [95% CI, 1.03-2.01]). Among HCV-seropositive individuals, treated participants without SVR vs those with SVR had a higher risk of liver events (IRR, 6.79 [95% CI, 2.33-19.81]), liver-related death (IRR, 3.29 [95% CI, 1.35-8.05]), and diabetes mellitus (IRR, 4.62 [95% CI, 1.53-13.96]). Similar but not statistically significant differences were found between untreated HCV RNA-positive patients and those with SVR. While HCV exposure was associated with an increased risk of kidney disease and osteoporosis/fracture, this risk did not seem to be dependent of persistent HCV RNA. Successful HCV treatment was associated with a lower incidence of liver disease, liver-related death, and diabetes mellitus, whereas the other conditions studied were less affected.....Compared with HCV-negative controls, HCV-seropositive participants had an increased risk of liver events (incidence rate ratio [IRR], 6.29 [95% confidence interval {CI}, 3.52-11.22]), liver-related death (IRR, 8.24 [95% CI, 3.61-18.83]), kidney events (IRR, 2.43 [95% CI, 1.11-5.33]), and osteoporosis/fracture (IRR, 1.43 [95% CI, 1.03-2.01])..... Among HCV-seropositive individuals, treated participants without SVR vs those with SVR had a higher risk of liver events (IRR, 6.79 [95% CI, 2.33-19.81]), liver-related death (IRR, 3.29 [95% CI, 1.35-8.05]), and diabetes mellitus (IRR, 4.62 [95% CI, 1.53-13.96]). Similar but not statistically significant differences were found between untreated HCV RNA-positive patients and those with SVR"
HCV-Associated Risks of Comorbidities and Death
Comparing HCV-Seropositive Groups Combined to HCV-Seronegative Controls
HCV-seropositive persons had an increased risk of liver disease (adjusted incidence rate ratio [IRR], 6.29 [95% confidence interval {CI}, 3.52-11.22]), liver-related death (IRR, 8.24 [95% CI, 3.61-18.83]), kidney disease (IRR, 2.43 [95% CI, 1.11-5.33]), and osteoporosis/fracture (IRR, 1.43 [95% CI, 1.03-2.01]), compared with HCV-seronegative controls. No evidence for an increased risk in HCV-seropositive persons was found for diabetes mellitus (IRR, 1.27 [95% CI, .83-1.93]), cardiovascular disease (IRR, 0.90 [95% CI, .60-1.34]), non-AIDS malignancy (IRR, 1.07 [95% CI, .75-1.52]), CDC HIV category B/C events (IRR, 1.12 [95% CI, .79-1.60]), and non-liver-related death (IRR, 0.90 [95% CI, .68-1.21]).
Comparing Each of the 4 HCV-Seropositive Groups to HCV-Seronegative Controls
The adjusted IRRs for the development of comorbidities and death for the different HCV-seropositive groups compared to HCV-seronegative controls are shown in Figure 2. HCV-treated non-SVR patients had the highest incidence of liver disease, liver-related death, kidney disease, diabetes mellitus, and CDC HIV category B/C events compared with HCV-seronegative controls. They also had the highest risk of cardiovascular events and non-AIDS malignancies but without reaching statistical significance.
The risk of osteoporosis/fracture was significantly increased in HCV spontaneous clearers and patients with SVR compared to HIV-monoinfected controls. The incidence of non-liver-related death was similar in all of the HCV-seropositive groups compared with HIV-monoinfected controls.
Figure 2. Incidence rate ratios and 95% confidence intervals for the development of clinical events and deaths in multivariable analysis according to hepatitis C virus (HCV) serostatus and HCV stage during a follow-up time period of 8.2 years (mean). Multivariable analysis adjusted for HIV acquisition category, age, HIV-1 RNA, smoking, alcohol use, active intravenous drug use, and duration of HIV and HCV infection. Abbreviations: CDC, Centers for Disease Control and Prevention; CI, confidence interval; HCV, hepatitis C virus; HIV, human immunodeficiency virus; SVR, sustained virologic response; Tx, treatment.
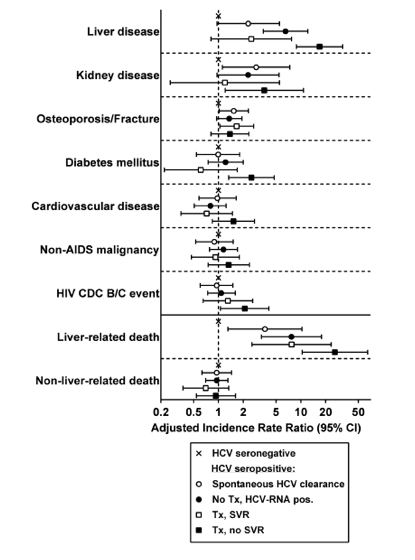
We found that HCV seropositivity was associated with increased risk of chronic kidney disease in accordance with others [18]. HCV-treated non-SVR participants, but not those with SVR, had an increased risk of kidney events compared with HIV-monoinfected patients. When we compared non-SVR participants with those with SVR, the incidence in kidney disease was elevated but did not reach statistical significance. In accordance with our findings, two large cohort collaboration groups [19, 20] found an increased risk in replicating but not cleared HCV infection compared with HIV-monoinfected participants. In a similar US and Canadian cohort study, results were contradictory, perhaps due to considerable differences between cohorts regarding patient characteristics [21]. Taken together, these results suggest that in HIV/HCV-coinfected persons, HCV exposure is associated with increased kidney disease risk, most probably due to a high prevalence of traditional renal risk factors in this patient group.
HCV exposure was an independent risk factor for osteoporosis and fracture. However, replicating compared with resolved HCV infection was not associated with bone-related events. Accordingly, several observational studies have shown that HIV/HCV-coinfected patients have an increased fracture incidence compared with HIV-monoinfected and HCV- and HIV-uninfected persons [22, 23]. Hansen et al demonstrated that fracture risk did not differ between viremic vs nonviremic HCV infection [24]. This suggests that osteoporosis and fracture risk in HIV/HCV-coinfected patients is multifactorial and mainly determined by lifestyle-related risk factors associated with HCV exposure, including illicit drug and alcohol abuse, poor nutrition, increased risk of trauma, and HIV and ART, rather than by HCV itself, and that achieving viral cure of chronic HCV infection will not significantly improve bone health. [from Jules: or, that HCV damage to bone may not be reparable by SVR].
We detected a trend of an increased risk of cardiovascular events in HCV nonresponders compared with successfully treated patients. In the current literature, reports regarding the association between chronic HCV infection and cardiovascular disease are conflicting. Although there is evidence that HCV should be considered a risk factor for carotid atherosclerosis, stroke, and heart failure (reviewed in [5, 25]), the association between HCV infection and coronary artery disease remains unclear (reviewed in [26]).
In line with a Spanish HIV/HCV cohort that found an increased frequency of new AIDS-defining events and deaths in HCV treatment nonresponders vs responders [7], we noted an increased risk of CDC HIV category B/C events in non-SVR compared with HIV-monoinfected patients. Non-SVR compared to those with SVR had a higher incidence of HIV/AIDS events, but without reaching statistical significance. As CD4 cell levels and suppression of HIV replication were similar between non-SVR and HIV-monoinfected participants at time of HIV/AIDS events, respectively, at last follow-up (Table 1), this finding cannot be explained by CD4 cell recovery, splenic sequestration in cirrhotic non-SVR patients [27], or control of HIV infection. One might speculate whether failure to achieve SVR might be due to an immunodeficiency predisposing also for HIV/AIDS-related opportunistic conditions.
Extrahepatic mortality was high in HCV-coinfected persons, consistent with our previous observations [2]. In HCV-seropositive participants, only one-third of deaths were liver related. Main causes of extrahepatic death were sepsis, substance abuse, and HIV/AIDS, indicating the important contribution of social and behavioral factors to mortality in this patient group. However, the risk of non-liver-related death in HCV-seropositive vs HCV-seronegative, and HCV-viremic vs nonviremic patients, was similar in adjusted analysis.
The treated non-SVR participants had the highest risk of liver-related and also of non-liver-related events. The prevalence of advanced liver fibrosis was by far the highest in this group (Table 1). When we adjusted for advanced fibrosis, most IRRs decreased, indicating that liver fibrosis is an important contributor to non-liver-related morbidity and mortality.
We conclude that HCV-exposed, HIV-infected individuals have an increased risk of kidney disease and bone-related events that does not seem to be related to persistent viral replication. In addition to a significant decrease of liver-related disease and death, therapeutic viral eradication leads to a reduction of diabetes mellitus. Prospective large-scale cohort collaborations are needed to further describe the impact of HCV eradication with DAAs on non-liver-related morbidity and mortality.
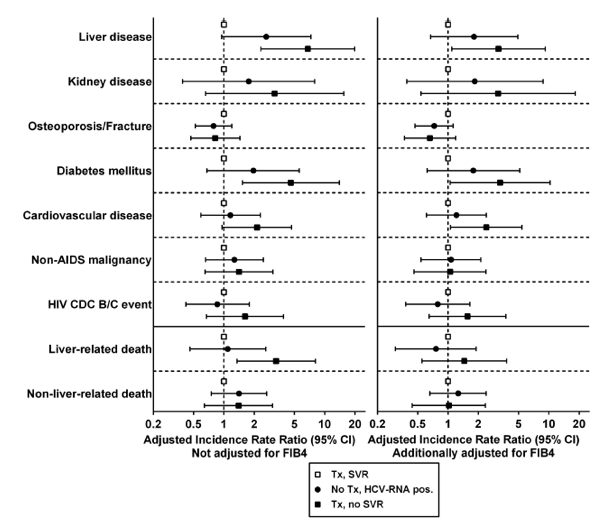
Comparing HCV-Viremic Patients (Untreated HCV RNA Positive and Treated Non-SVR) to Those With SVR
Treated patients with non-SVR vs those with SVR had a higher risk of liver events, liver-related death, and diabetes mellitus. To investigate whether the increased risks in non-SVR participants could be related to advanced liver fibrosis, we additionally adjusted for FIB-4 score. As expected, the risk of liver-related death in non-SVR patients did not remain increased. The difference in incidence of liver events and diabetes remained. HCV-viremic patients (both untreated HCV RNA positive and non-SVR) compared to those with SVR had a trend for an elevated incidence of kidney disease, cardiovascular events, and non-AIDS malignancies but without reaching statistical significance (Figure 3; Supplementary Table 1).
Figure 3. Incidence rate ratios and 95% confidence intervals for the development of clinical events and deaths in multivariable analysis according to hepatitis C virus (HCV) viremia in treated patients during a follow-up time period of 8.2 years (mean). Left: Multivariable analysis adjusted for HIV acquisition category, age, HIV-1 RNA, smoking, alcohol use, active intravenous drug use, and duration of HIV and HCV infection. Right: Additionally adjusted for liver fibrosis stage measured by FIB-4 score. Abbreviations: CDC, Centers for Disease Control and Prevention; CI, confidence interval; HCV, hepatitis C virus; HIV, human immunodeficiency virus; SVR, sustained virologic response; Tx, treatment.
| |
| |
| |
|
|
|