| |
Do people living with HIV experience greater age advancement than their HIV-negative counterparts? Yes-Biological Age greater by 13 Years in HIV+
|
| |
| |
Download the PDF here
Download the PDF here
De Francesco, Davidea; Wit, Ferdinand W.b,c; Bürkle, Alexanderd; Oehlke, Sebastiand; Kootstra, Neeltje A.e; Winston, Alanf; Franceschi, Claudiog; Garagnani, Paolog; Pirazzini, Chiarag; Libert, Claudeh,i; Grune, Tilmanj; Weber, Danielaj; Jansen, Eugène H.J.M.k; Sabin, Caroline A.a; Reiss, Peterb,c on behalf of the the Co-morBidity in Relation to AIDS (COBRA) Collaboration
All HIV-positive COBRA participants had plasma HIV RNA less than 50 copies/ml and were on antiretroviral therapy at study visit, they had a median (IQR) CD4+ T-cell count of 618 (472-806) cells/μl, and 31% had a prior clinical AIDS diagnosis (Table 1).- average age 55 - The number and proportion of people who had ever been exposed to each antiretroviral drug and the median (IQR) duration of exposure are reported in Supplementary Table 2, http://links.lww.com/QAD/B389.
Biological age was significantly greater than chronological age by a mean of 13.2 (95% CI 11.6-14.9) years in HIV-positive COBRA participants and by 5.5 (3.8-7.2) years in HIV-negative participants (P < 0.001 for each). In contrast, biological age was a mean of 7.0 (4.1-9.9) years lower than chronological age in blood donors (P < 0.001, Fig. 1a). Whilst age advancement was greater in both COBRA groups compared with blood donors (P < 0.001 for each), the HIV-positive COBRA participants also demonstrated greater age advancement than the HIV-negative participants (P < 0.001). Age advancement was also negatively correlated with chronological age (Pearson's r = -0.17, P = 0.08) with no significant interaction with HIV-status/group (P = 0.66, Fig. 1b)......Factors associated with advancement: Overall, men had greater age advancement than women even after adjustment for HIV-status/group. However, among COBRA participants only, the difference (95% CI) between men and women was only 0.09 (-5.04 to 5.22) years (P = 0.97) and the difference between COBRA groups and blood donors remained significant even after adjusting for sex (both Ps <0.001). Viral co-infections such as CMV and chronic HBV, as well as CD4+ and CD8+ T-cell count and their ratio appeared to be associated with increased age advancement. However, only chronic HBV, total and high avidity anti-CMV IgG antibody titer and CD8+ T-cell count showed significant associations, which were also independent of HIV-status/group.
In conclusion, our results suggest that PLWH with undetectable plasma HIV RNA may experience accentuated aging - [of course, I disagree, I think that accelerated & accentuated both are in play-the data supports this too . Jules] - compared with HIV-negative individuals with similar lifestyles, as estimated using a set of validated biomarkers of aging. This age advancement appears to be related to viral co-infections such as CMV and chronic HBV, but also to historic severe immunosuppression and possibly exposure to particular antiretroviral drugs. Future longitudinal studies are required to further help clarifying the effect of HIV and its treatment on the natural aging process and the functional and clinical consequences in the millions of PLWH worldwide.
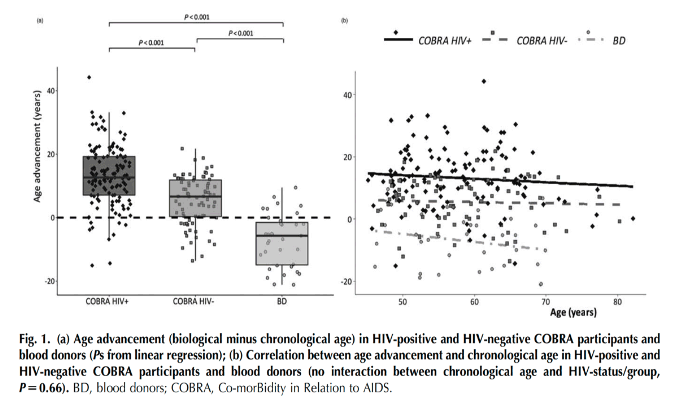
Both successfully treated PLWH and people without HIV with similar lifestyles show signs of age advancement compared with healthy blood donors who, conversely, appear younger than their chronological age. This may be explained by the strict requirements around blood donation in the Netherlands. This hypothesis is supported, for example, by the observation that the prevalence of CMV infection was higher in HIV-negative participants (79.8%), and lower in blood donors (22.9%), than in the general Dutch population (50% prevalence at 50 years of age) [27]......Whilst both groups of COBRA participants exhibited age advancement, this was significantly greater in HIV-positive than in HIV-negative participants. Whilst this difference in age advancement did not appear to be explained by differences in participant characteristics, chronic viral co-infections such as CMV and HBV, prior immunosuppression and exposure to some antiretroviral drugs may have contributed to this age advancement. CMV and HBV co-infections may cause premature aging of the immune system in PLWH by their chronic antigenic stimulation, inducing systemic immune activation [28]. In particular, CMV reactivation and concurrent immune responses to control infection are associated with aging and increased morbidity and mortality in both the general population and PLWH [29,30]. In the same group of people reported here, we previously found that increased anti-CMV IgG levels are associated with a higher proportion of terminally differentiated CD4+ and CD8+ T cells as well as CD4+ T-cell activation [31], which may, in turn, have contributed to the observed greater age advancement.......Among antiretroviral drugs, we found a significant association between age advancement and duration of past exposure to saquinavir, which was independent of concomitant exposure to other drugs generally considered to have the greatest mitochondrial toxicities and potential effects on aging such as didanosine, stavudine, zalcitabine and zidovudine (data not shown). Whereas saquinavir has been shown to directly induce vascular endothelial toxicity in vitro [34], other HIV protease inhibitors were shown to be able to induce vascular smooth muscle cell senescence by downregulating ZMPSTE24, leading to prelamin A accumulation and potential premature vascular aging, changes, which were also observed in peripheral blood mononuclear cells from HIV-infected patients who were treated with the same protease inhibitors [35]. The reason why we only observed an association between age advancement and exposure to saquinavir, but not other HIV protease inhibitors, remains unclear, and suggests this observation should be interpreted with caution. Importantly, having experienced more pronounced immunodeficiency in HIV-positive participants was also linked to an increased age advancement suggesting that a greater effect on aging is likely to occur as the infection remains untreated for longer......Our findings are consistent with previous reports of brain aging in the COBRA study [18] and with other studies of cellular and molecular markers of biological aging.
Indeed, studies of telomere length and CDKN2A [14,36], CD8+ T-cell senescence [37], and DNA methylation profiles ('epigenetic clock') [16,38] showed similar indications of age advancement in PLWH. Other studies reported evidence of accentuated aging only in PLWH with low nadir CD4+ T-cell counts compared with HIV-negative individuals [39,40]. Of note, however, these previous studies often included untreated PLWH (some with detectable HIV RNA) and HIV-negative controls that differed with regards to some lifestyle behaviors, such as smoking and alcohol consumption. In our study, effectively treated PLWH with high CD4+ T-cell counts and highly comparable HIV-negative controls were purposely recruited in order to reduce the influence of sociodemographic and lifestyle confounding factors. The importance of an appropriately chosen control group of HIV-negative individuals is also highlighted by the finding that HIV-negative COBRA participants showed greater age advancement compared with blood donors. Furthermore, these studies only focus on one single tissue or body system, and are therefore unable to reflect the intrinsic multicausal and multisystem nature of the aging process [41]. Chronic HIV may differently affect the rate of aging at the levels of cells, tissues or body systems within the same organism and this complexity is more likely reflected by a method used in our study, which integrates multiple sources of molecular, cellular and physiologic data.
Aging can be defined as the time-dependent decline of functional capacity and stress resistance associated with increased risk of disability, morbidity and mortality [5]. There is clear evidence that the rate of aging differs significantly between individuals, because of genetic heterogeneity and environmental factors [6]. Therefore, chronological age may not represent the best way of measuring aging and may not accurately reflect an individual's position in his/her total lifespan [7]. This has led to a search for reliable biomarkers of aging, defined as biological parameters that capture the age-related changes in body function or composition. These biomarkers could serve to measure 'biological' age, a hypothetical value denoting the extent of age-related changes in function and composition of a human body, and predict the onset of age-related diseases and/or expectant residual lifetime more accurately than chronological age [8].
Many candidate biomarkers of aging have been proposed in the scientific literature and have been used to investigate the association between HIV and aging [9]. Among these, there are markers of chronic systemic immune activation (soluble CD14+ and CD163+ [10,11]), inflammation (C-reactive protein and interleukin-6 [12]), coagulation (D-dimer [13]), leukocyte telomere length [14], somatic mitochondrial DNA mutations [15], expression levels of the cell cycle regulator CDKN2A [14], DNA methylation levels [16], ophthalmological parameters [17] and age-related brain atrophy [18].
A 10-item panel of biomarkers of aging identified by the MARK-AGE project [21,22] were measured and are listed in Supplementary Table 1, http://links.lww.com/QAD/B389. Briefly, these biomarkers have been selected as best predictors of chronological age among nearly 400 candidate biomarkers in a population of approximately 3300 individuals aged between 30 and 74 years (mean age was 56 years) recruited from eight European countries.
ABSTRACT
Objectives: Despite successful antiretroviral therapy, people living with HIV (PLWH) may show signs of premature/accentuated aging. We compared established biomarkers of aging in PLWH, appropriately chosen HIV-negative individuals, and blood donors, and explored factors associated with biological age advancement.
Design: Cross-sectional analysis of 134 PLWH on suppressive antiretroviral therapy, 79 lifestyle-comparable HIV-negative controls aged 45 years or older from the Co-morBidity in Relation to AIDS (COBRA) cohort, and 35 age-matched blood donors.
Methods: Biological age was estimated using a validated algorithm based on 10 biomarkers. Associations between 'age advancement' (biological minus chronological age) and HIV status/parameters, lifestyle, cytomegalovirus (CMV), hepatitis B (HBV) and hepatitis C virus (HCV) infections were investigated using linear regression.
Results: The average (95% CI) age advancement was greater in both HIV-positive [13.2 (11.6-14.9) years] and HIV-negative [5.5 (3.8-7.2) years] COBRA participants compared with blood donors [-7.0 (-4.1 to -9.9) years, both P's < 0.001)], but also in HIV-positive compared with HIV-negative participants (P < 0.001). Chronic HBV, higher anti-CMV IgG titer and CD8+ T-cell count were each associated with increased age advancement, independently of HIV-status/group. Among HIV-positive participants, age advancement was increased by 3.5 (0.1-6.8) years among those with nadir CD4+ T-cell count less than 200 cells/μl and by 0.1 (0.06-0.2) years for each additional month of exposure to saquinavir.
Conclusion: Both treated PLWH and lifestyle-comparable HIV-negative individuals show signs of age advancement compared with blood donors, to which persistent CMV, HBV co-infection and CD8+ T-cell activation may have contributed. Age advancement remained greatest in PLWH and was related to prior immunodeficiency and cumulative saquinavir exposure.
|
|
| |
| |
|
|
|