| |
Ten Take-Home Lessons From Top HIV-Aging Articles in 2017
|
| |
| |
Concise reviews of 10 articles with clinical insights into managing older adults with HIV
1. New HIV Diagnosis Rate Rising 2% Yearly in People Over 50 in Europe
2. Immediate ART Cuts All-Cause and Non-AIDS Mortality in People 50 to 70
3. Frailty Tied to Recurrent Falls in Older Men and Women With HIV
4. Fracture Rate Doubled at Age 50-59 in US Men With vs Without HIV
5. One in 5 Older People With HIV Has an (Often Modifiable) IADL Disability
6. Higher Risk of Heart Failure in Aging Vets With HIV, Regardless of Ejection Fraction
7. Greater Brain Atrophy With HIV Over Age 60 Despite Viral Suppression
8. HIV Tied to Worse Health Quality of Life and Higher Chance of Depression
9. More Psychiatric Symptoms in People Older Than 60 With vs Without HIV
10. Almost 40% of Aging HIV/HCV Group Have Potential DAA Interaction - 77% ≥60 Have Potential DAA Interaction
NATAP Aging & HIV Section
From Jules: older aging HIV+ and their clinics need numerous different types of special support services including unique reimbursement. Yet federal officials remain in the dark, I do not think they understand the problem and have not been discussing these problems much at all. This problem will worsen as HIV+ in the USA age further, frailty & disability increase, long term care demands increase, and the socio-economic situation worsens. HIV healthcare is uniquely funded by $1 billion per year to the Ryan White Care Act by tax payer money designed to address HIV needs, yet the aging problems in the healthcare clinics remains ignored. It is as if they want them to die and go away, so as not to deal with this.
By Mark Mascolini
New HIV Diagnosis Rate Rising 2% Yearly in People Over 50 in Europe
Report: Tavoschi L, Gomes Dias J, Pharris A, on behalf of the EU/EEA HIV Surveillance Network. New HIV diagnoses among adults aged 50 years or older in 31 European countries, 2004-15: an analysis of surveillance data. Lancet HIV. 2017;4:e514-e521.
Background: The HIV population--and indeed the global population--are aging. UNAIDS estimates that 4.2 million people 50 or older had HIV worldwide in 2013. HIV prevalence in older people has risen in the past few decades, particularly in Western Europe, the United States, and Canada. This climbing HIV rate in older people reflects (1) longer survival with HIV infection and (2) a rising incidence of new HIV infections in people 50 or older.
Studies suggest that older adults infected with HIV are more likely to present late6,7,10 and are at increased risk of short-term mortality than are younger adults. Older adults living with HIV and health professionals caring for them face unique challenges.
Objective: This analysis of surveillance data tracks trends and describe factors explaining HIV diagnoses in European people 50 or older from 2004 through 2015.
Methods: Analyzing routine annual surveillance data from January 2004 through December 2015 in 31 European countries, researchers tracked new HIV diagnoses in people 15 or older. The investigators divided this group into younger people (15 to 49) and older people (50 or older). Besides age at diagnosis, researchers collected data on sex, migration status, HIV transmission route, and CD4 count.
The researchers defined late diagnosis as diagnosis below a CD4 count of 350. They used linear regression analysis to determine temporal trends.
Study population: Among 29,419 new HIV diagnoses in 2015, 5076 (17.3%) involved people 50 or older. In 2015 about 77% of both the older and younger groups were men. A significantly higher proportion of older than younger people acquired HIV heterosexually (42.4% versus 30.8%), while lower proportions of older people acquired HIV through sex between men (30.3% versus 45.1%) or by injecting drugs (2.6% versus 4.6%) (P < 0.0001).
Median CD4 count at diagnosis in 2015 was significantly lower in the older group (240 versus 394, P < 0.0001). A significantly higher proportion of older than younger people were born in the reporting country (63.7% versus 53.8%) and a significantly lower proportion were not born in a European country (17.2% versus 25.6%) (P < 0.0001).
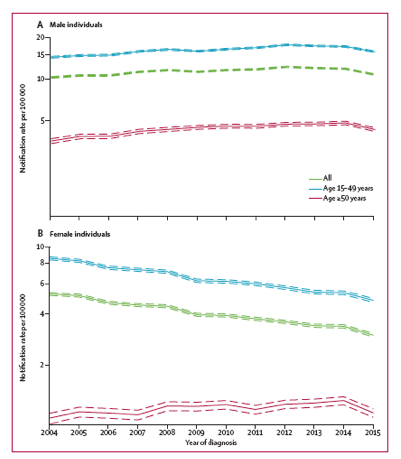
Results: In 2015 the new HIV diagnosis rate was 2.5 per 100,000 in people 50 or older versus 10.4 per 100,000 in younger people. In 2015 the male-to-female diagnosis rate was slightly higher in older people than younger people (3.5 versus 3.3). Across the 2004-2015 study period, in older people the new HIV rate was 4.4 per 100,000 men and 1.2 per 100,000 women.
From 2004 through 2015 there were 312,501 new HIV diagnoses in people younger than 50 (11.4 per 100,000) and 54,102 in people 50 or older (2.6 per 100,000). The new diagnosis rate did not change substantially from year to year in younger people (average annual change -0.4%, 95% confidence interval [CI] -1.0 to 0.2, P = 0.1716) but increased significantly in older people (average annual change 2.1%, 95% CI 1.1 to 3.1, P = 0.0009). Across the 2004-2015 study period, new HIV diagnoses rose significantly from year to year in both older men (average annual change 2.2%, 95% CI 1.2 to 3.3, P = 0.0006) and in older women (average annual change 1.3%, 95% CI 0.2 to 2.4, P = 0.025).
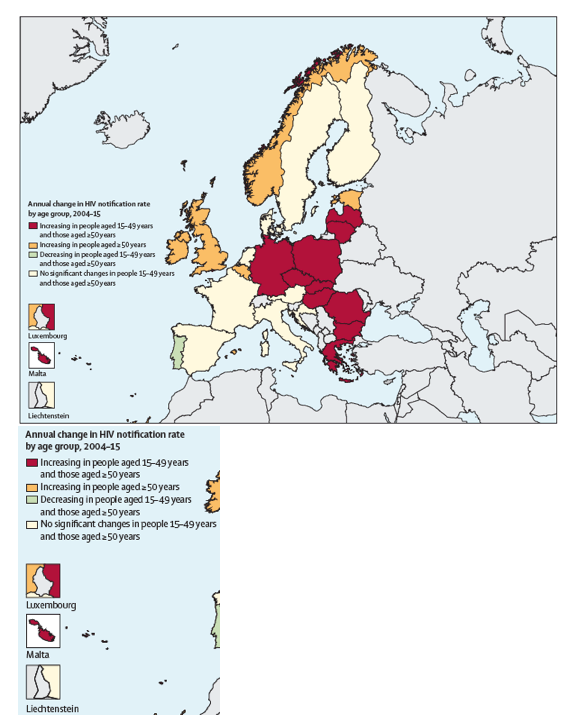
From 2004 through 2015, new HIV diagnoses among older people rose significantly in 16 countries: Belgium, Bulgaria, Czech Republic, Estonia, Germany, Greece, Hungary, Ireland, Latvia, Lithuania, Malta, Norway, Poland, Romania, Slovakia, and the UK. The new HIV rate in older people fell significantly across those years in only one country, Portugal.
Among younger people the new HIV diagnosis rate rose significantly from 2004 through 2015 in 12 countries: Bulgaria, Croatia, Czech Republic, Germany, Greece, Hungary, Latvia, Lithuania, Malta, Poland, Romania, and Slovakia. Across those years the new HIV rate fell significantly among younger people in 6 countries: Austria, France, Netherlands, Norway, Portugal, and the UK. Thus new HIV diagnoses rose in older people but not younger people in 5 countries: Belgium, Estonia, Ireland, Norway, and the UK.

Figure 2: Average annual percentage change in new HIV diagnoses in people aged 50 years or older in the European Union and European Economic Area, 2004-15*
Over the study period new HIV infections resulting from sex between men rose significantly in both older and younger people, but more in older people (average annual change 5.8% versus 2.3%). New HIV resulting from heterosexual sex remained stable in older people (average annual change 1.1%, P = 0.08) while falling significantly in younger people (average annual change -5.7%, P < 0.0001). New HIV resulting from injecting drugs rose significantly in older people (average annual change 7.4%, P < 0.0001) while falling significantly in younger people (average annual change -4.5%, P = 0.0087).
Discussion/implications: Among Europeans 50 or older, new HIV diagnoses rose steadily over a 12-year period (2004-2015).
Figure 3: Annual notification rate of new HIV diagnoses per 100 000 men and boys (A) and women and girls (B) in the European Union and European Economic Area, 2004-15
The y axes are on a log scale. The dotted lines represent 95% CIs.
HIV diagnosis at a CD4 count below 350 was significantly associated with older age in this analysis and in previous reports. These findings could reflect (1) late diagnosis and/or (2) more rapidly declining CD4 counts after infection in older people.
Presenting with a late diagnosis was significantly associated with older age in this analysis and in most
previously published work, with some exceptions. Late presentation is particularly worrying among older people, because it further increases the already higher mortality in this age group compared with younger individuals
Several studies found that HIV testing is uncommon among older people. That trend could reflect low HIV risk perception in older people or "scant knowledge" about HIV and HIV transmission in older people.
Among gay and bisexual men in the United States, the numbers of new HIV diagnoses in men 55 and older rose 18% from 2010 to 2014. In 2013 people 55 or older accounted for one quarter of all diagnosed or undiagnosed HIV infections in the United States.
Research "suggests that the most significant factor affecting [HIV] testing patterns in older adults is an active offer of an HIV test by service providers." Opportunities for offering HIV tests are more frequent in older than younger people because older people use health services more often.
Newly diagnosed older adults were mainly native to the reporting country, in line with findings from national studies. By contrast with trends among younger adults, the study showed a significant increase in new diagnoses among older men and women. Transmission via sex between men rose in both age groups between 2004 and 2015. However, the study noted stable or increasing trends for transmission modes such as heterosexual contact and injecting drug use among older people, compared with significant decreases among younger people.
These findings revealed a slightly increased male-to female ratio in the older age group compared with the younger age group, although not as pronounced as that in other published work, which suggests that diagnoses in men predominate in the older age group.
In line with published evidence, in this analysis, transmission of HIV among older adults was mainly sexual, and acquisition of the infection as a result of heterosexual sex was more common than that as a result of sex between men. More in-depth analysis in national level studies suggests some evidence of risks related to commercial sex and travel abroad among heterosexual men.
Key take-home: "Increasing new HIV diagnoses among older adults point towards the compelling need to heighten awareness among health-care providers and deliver more targeted prevention interventions for this age group and the total adult population."
"Our findings, alongside those from previous studies, show that older adults in the EU/EEA are a group at potential risk for HIV and that older individuals are likely to be diagnosed late across the region. Taken together, the existing evidence points towards the compelling need to consider older adults as a relevant target for HIV prevention programmes."
Additional information:
⋅ UNAIDS. HIV and Aging.
⋅ Centers for Disease Control and Prevention (CDC). HIV among people aged 50 and over.
⋅ US Department of Health and Human Services. HIV and older adults.
⋅ Adekeye OA, Heiman HJ, Onyeabor OS, Hyacinth HI. The new invincibles: HIV screening among older adults in the U.S. PLoS One. 2012;7:e43618.
⋅ Ellman TM, Sexton ME, Warshafsky D, Sobieszczyk ME, Morrison EA. A forgotten population: older adults with newly diagnosed HIV. AIDS Patient Care STDS. 2014;28:530-536.
--------------------------------------------
Immediate ART Cuts All-Cause and Non-AIDS Mortality in People 50 to 70
Report: Lodi S, Costagliola D, Sabin C, et al. Effect of immediate initiation of antiretroviral treatment in HIV-positive individuals aged 50 years or older. J Acquir Immune Defic Syndr. 2017;76;311-318.
Background: Randomized trials determined that starting antiretroviral therapy (ART) at a high CD4 count lowers mortality and risk of serious AIDS and non-AIDS complications. As a result, US and other guidelines recommend ART immediately upon HIV diagnosis, regardless of CD4 count. But trials of ART initiation enrolled mostly younger and middle-aged people and had low death rates, so the impact of immediate ART on mortality in older adults remains uncertain.
Objective: This study estimated 5-year risk of all-cause and non-AIDS mortality in 50- to 70-year-old antiretroviral-naive adults in a general HIV population and the US Veterans Affairs Cohort Study (VACS), which has higher mortality than a general high-income-country HIV population.
Methods: Two groups provided participants for the study, the HIV-CAUSAL Collaboration (including people in Europe, Brazil, and Canada) and the US Veterans Aging Cohort Study (VACS). Participants were 50 to 70 years old, free of AIDS, and naive to antiretrovirals after December 31, 2004. All had at least one CD4 count and viral load measured within 3 months of each other. Baseline was the earliest date at which participants met all these criteria.
Researchers estimated 5-year survival for three groups: (1) immediate ART (starting within 3 months of baseline), (2) starting ART at a CD4 count below 500, or (3) starting ART at a CD4 count below 350. They estimated 5-year survival with a regression model, the parametric g-formula. Follow-up began at baseline and ended at death, 12 months after the most recent lab assessments, date when the participant started a non-ART regimen, or 5 years after baseline, whichever came first.
Study population: The population included 9599 people, 2672 of them (28%) veterans. Most participants in the general HIV population were men (79%), and almost all veterans were men (98%). Median age stood at 55 in the general population (interquartile range [IQR] 52 to 59) and 56 among veterans (IQR 53 to 60). Median baseline CD4 count was 354 (IQR 203 to 530) in the general HIV population and 284 (IQR 128 to 471) in veterans.
Results: During follow-up 74% in the general HIV population and 78% in the veterans group began ART. While 295 people (4.3%) in the general population died, 339 veterans (12.7%) died. Rates of all-cause and non-AIDS mortality were 12.3 and 6.3 per 1000 in the general population and 42.4 and 9.7 per 1000 in veterans.
Estimated 5-year risk of all-cause and non-AIDS mortality was consistently (and usually significantly) higher in 50- to 70-year-olds who started ART below 500 CD4s or below 350 CD4s than in those who started immediately. Compared with people who started ART immediately in the general population, those starting below 500 CD4s had a 3% higher risk of all-cause mortality (significant) and a 3% higher risk of non-AIDS mortality (nearly significant) (see Table). Compared with immediate starters, general-population participants starting below 350 CD4s had a 7% higher risk of all-cause mortality (significant) and a 6% higher risk of non-AIDS mortality (not significant).
The median (interquartile range) CD4 count at baseline was 354 cells/mm3 (203-530) in the general
HIV population and 284 cells/mm3 (128-471) in US Veterans
Compared with 50- to 70-year old veterans who started ART immediately, those starting below 500 CD4s had a 5% higher risk of all-cause mortality and a 6% higher risk of non-AIDS mortality (see Table). Compared with veterans starting ART immediately, those starting below 350 CD4s had an 11% higher risk of all-cause mortality and a 15% higher risk of non-AIDS mortality. All these differences were statistically significant.
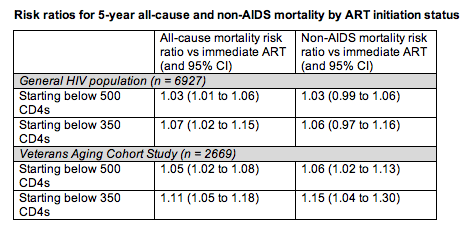
Among people in the general HIV population with a baseline CD4 count at or above 500, estimated risk of all-cause mortality was 2.8% (95% CI 1.5 to 4.5) with immediate ART, 3.7% (95% CI 2.5 to 4.5) when starting ART below 500 CD4s, and 4.4% (95% CI 3.1 to 5.9) when starting ART below 350 CD4s.
Discussion/implications: Starting ART immediately after HIV diagnosis, as guidelines recommend, appears to reduce all-cause and non-AIDS mortality in people 50 to 70 years old.
In a hypothetical 50- to 70-year-old HIV cohort of 1000 people, starting ART immediately rather than below 350 CD4s could prevent between 4 and 16 deaths over 5 years.
These findings on the benefits of immediate ART are compatible with results of a similar 3500-person US study using routinely collected data.
Key take-home: Health care providers and policy makers should put more effort into "diagnosing HIV earlier, particularly in older patients to ensure timely initiation of treatment and follow-up for concomitant comorbidities, thereby maximizing the benefit of early treatment for HIV. Immediate initiation of ART seems to be beneficial in reducing all-cause mortality in AIDS-free patients aged 50 years or older, despite their low baseline CD4 count."
Additional information:
⋅ The INSIGHT START Study Group. Initiation of antiretroviral therapy in early asymptomatic HIV infection. N Engl J Med. 2015;373:795-807.
⋅ Edwards JK, Cole SR, Westreich D, et al. Age at entry into care, timing of antiretroviral therapy initiation, and 10-year mortality among HIV-seropositive adults in the United States. Clin Infect Dis. 2015;61:1189-1195.
⋅ US Department of Health and Human Services Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents. October 2017.
⋅ US Department of Health and Human Services. HIV and older adults.
--------------------------------------
Frailty Tied to Recurrent Falls in Older Men and Women With HIV
Report: Tassiopoulos K, Abdo M, Wu K, et al. Frailty is strongly associated with increased risk of recurrent falls among older HIV-infected adults. AIDS. 2017;31:2287-2294.
Background: Test-measured frailty is highly prevalent in HIV populations, affecting up to one quarter of people. In the general population 1 in 5 falls results in fracture, and the fall-related death rate in older Americans has been rising for a decade. Both frailty and falls occur at earlier-than-expected ages among HIV-infected individuals. But little is known about potential links between frailty and falls in people with HIV. Among middle-aged and older HIV-infected individuals, including those with well suppressed infection, frailty, and impairments in physical function are common . One study found that the prevalence of falls among middle-aged HIV-infected adults with a mean age of 52 years paralleled that of HIV-uninfected adults 65 years or older.
Objective: This prospective, multicenter cohort study assessed associations between frailty and falling in middle-aged and older men and women with HIV.
Methods: The study involved members of AIDS Clinical Trials Group (ACTG) study A5322 (HAILO), all of whom were 40 or older and started antiretroviral therapy in an ACTG trial. In this prospective observational study, participants made twice-yearly visits in which they reported falls in the preceding months.
Participants also had a standard frailty assessment, including grip strength, gait speed (4-meter timed walk), self-reported unintentional weight loss, self-reported exhaustion, and self-reported limitations in physical activities. Researchers rated participants according to the baseline frailty assessment as frail (3 to 5 frailty components), prefrail (1 or 2 components), and nonfrail (no components). They also determined results of two individual frailty tests, gait speed (1 meter/second or slower versus faster) and grip strength (by sex- and BMI-based cutoffs).
The researchers used separate multinomial logistic regression models to assess associations between the three frailty outcomes and single or recurrent falls in the 12 months after the frailty assessment.
Study population: The analysis included 967 ACTG A5322 members who had a baseline frailty assessment and subsequent fall reports. Most participants, 81%, were men, and median age stood at 51 years (interquartile range 46 to 56). Almost half of the study group, 48%, were white, while 30% were black and 22% Hispanic. Most participants, 85%, had at least a high-school education, and only 18% had no health insurance.
Results: Among the 967 study participants, 174 (18%) fell at least once in the past 12 months--106 (11%) reported 1 fall and 68 (7%) reported 2 or more falls. Among the 68 recurrent fallers, 40 (59%) fell 3 or more times. Among 174 fallers, 37 (21%) sought medical care for the fall and 9 (5%) broke a bone.
Fifty-nine people (6%) met frailty criteria and 373 (39%) were prefrail.
Almost half of the people with frailty (49%) had one or more falls, while 22% of prefrail people fell and 12% of nonfrail people fell.
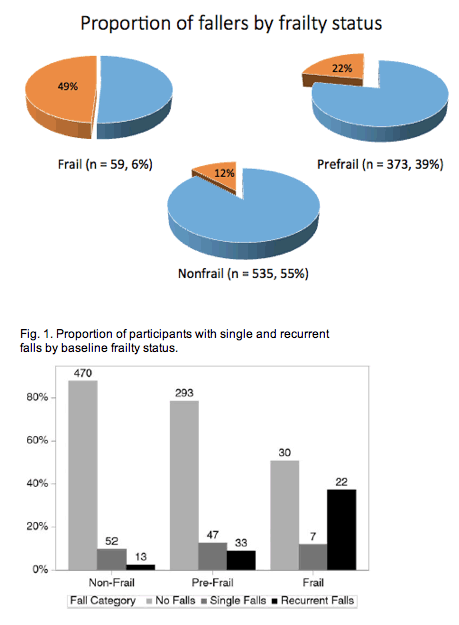
Compared with nonfrail people, prefrail individuals had almost 4-fold higher adjusted odds of recurrent falls (adjusted odds ratio [aOR] 3.80, 95% confidence interval [CI 1.87 to 7.72) and frail people had more than 17-fold higher odds of recurrent falls (aOR 17.3, 95% CI 7.03 to 42.6). Prefrail and frail people had higher odds of a single fall, but these associations did not reach statistical significance.
People who failed either of the two objective frailty measures--grip strength or gait speed--had independently higher odds of recurrent falls: for weak grip strength, aOR 3.77, 95% CI 2.17 to 6.54; for slow gait speed, aOR 2.93, 95% CI 1.59 to 5.38.
Among the 967 people studied, 373 (39%) had peripheral neuropathy. Compared with nonfrail people with peripheral neuropathy, frail participants with neuropathy had almost 18-fold higher odds of falling (aOR 17.7, 95% CI 6.89 to 45.5). In this study over a third of the cohort reported peripheral neuropathy.
"Three hundred and seventy-three participants (38.6%) had peripheral neuropathy. Fifty-five (14.7%) had moderate to severe loss of vibration bilaterally and 278 (74.5%) had absent or hypoactive reflexes bilaterally (35 had both moderate/severe vibration loss and absent/hypoactive reflexes bilaterally). Most of the others had mild loss of vibration bilaterally with normal reflexes. Among individuals with peripheral neuropathy, frail participants were significantly more likely to experience falls (aOR 17.7, 95% CI 6.89, 45.5) than nonfrail individuals, whereas among those without peripheral neuropathy, the association between frailty and falls was weaker and not significant (aOR 2.16, 95% CI 0.76, 6.11; P value for interaction term = 0.003).
Peripheral neuropathy, manifested in part through sensory loss, has deleterious effects on gait, balance, and postural stability. Frail individuals, who are defined by their limited reserves, lower limb weakness, and diminished ability to respond to stressors, are likely unable to recover quickly after losing their balance.
The protective association of slow walk speed on the incidence of single falls was unexpected. However, in a population-based cohort study of community-dwelling older individuals, those who were classified as normal to slow walkers had a lower overall risk of falling than did individuals classified as fast walkers [26], and may suggest that the former category of individuals have already employed compensatory mechanisms to minimize fall risk."
Discussion/implications: This is the first study to link prefrailty to recurrent falls in people with HIV. Frailty also independently predicted recurrent falls.
The association between frailty and falls appears to be greater in people with than without HIV.
The association between frailty and falls proved more pronounced in men than women, but the difference lacked statistical significance.
People with HIV may have several comorbidities that affect fall risk, including weakness, impaired balance, neuropathy, and polypharmacy.
The combined effect of peripheral neuropathy and frailty on fall risk may be greater than the sum of their individual effects.
Key take-home: Either of two simple frailty tests--grip strength or gait speed--may be enough to predict recurrent falls in middle-aged to older people with HIV. "The magnitude of the overall association we identified between frailty and falls is substantially greater than previously observed in studies of older HIV-uninfected individuals."
Additional information:
⋅ Centers for Disease Control and Prevention. Important facts about falls.
⋅ National Council on Aging. Falls prevention.
⋅ Mayo Clinic. Fall prevention: Simple tips to prevent falls.
⋅ Erlandson KM, Schrack JA, Jankowski CM, Brown TT, Campbell TB. Functional impairment, disability, and frailty in adults aging with HIV-infection. Curr HIV/AIDS Rep. 2014;11:279-290.
⋅ Gonciulea A, Wang R, Althoff KN, et al. An increased rate of fracture occurs a decade earlier in HIV+ compared with HIV- men. AIDS. 2017;31:1435-1443.
--------------------------------
Fracture Rate Doubled at Age 50-59 in US Men With vs Without HIV
Report: Gonciulea A, Wang R, Althoff KN, et al. An increased rate of fracture occurs a decade earlier in HIV+ compared with HIV- men. AIDS. 2017;31:1435-1443.
Background: Low bone mineral density (BMD) and, as a result fractures, have become more common with HIV infection than in the general population. Fragility fractures possibly attributed to low BMD are more frequent in men and women with than without HIV. US experts recommend early fracture risk screening in people with HIV, but the age when screening should begin remains uncertain.
"Among HIV+ patients, several risk factors have been associated with bone loss and higher incidence of fracture, including traditional risk factors such as age, sex, race, BMI, smoking, alcohol and drug use. and specific antiretroviral therapy (ART) agents, especially protease inhibitors and tenofovir disoproxil fumarate (TDF) containing combination. Coinfection with hepatitis C virus (HCV) increased fracture risk in several reports, whereas others found no association.
Objective: This prospective study aimed to determine fracture incidence in aging men with or without HIV and to identify fracture risk factors.
Methods: The study involved HIV-positive men who have sex with men (MSM) and behaviorally similar HIV-negative MSM in the ongoing observational Multicenter AIDS Cohort Study (MACS) at 4 centers in the United States. MACS members make twice-yearly visits for evaluation. Researchers collected self-reported fracture data from the MACS database. Starting in 2001, men also specifically reported whether they had a fracture.
This analysis involved men at least 40 years old in 2010 who had at least one follow-up visit. The analysis focused on (1) all fractures except those of the face, skull, or digits and (2) fragility fractures of the vertebral column, femur, wrist, and humerus. The researchers used Poisson regression to estimate crude incidence rate ratio (IRR) and adjusted incidence rate ratio (aIRR).
Study population: The study included 1221 men with HIV and 1408 HIV-negative men. Men with and without HIV were similar in age, body mass index (BMI), estimated glomerular filtration rate (eGFR), and moderate/heavy alcohol drinking. The HIV group included a higher proportion of nonwhites (41% versus 27%, P < 0.001), a higher proportion of current smokers (38% versus 31%, P < 0.001), and a higher proportion of HCV-infected (10% versus 6%, P<0.001). "The presence of comorbidities such as
diabetes and hypertension was similar by HIV serostatus."
At the index visit (the start of observation for fractures), men with HIV had a median CD4 count of 490 and a median viral load of 342 copies/mL. At last follow-up 61% of men with HIV were taking tenofovir disoproxil fumarate (TDF).
Results: Incidence of all fractures proved slightly higher in men with than without HIV (12.8 versus 10.0 per 1000 person-years). Among HIV-negative men, all-fracture incidence was similar at ages 40-49 (9 per 1000 p-y) and 50-59 (8.5 per 1000 p-y) then jumped among men 60 or older (15.1 per 1000 p-y). In contrast, among men with HIV fracture incidence more than doubled from ages 40-59 (8.1 per 1000 p-y) to 50-59 (18.2 per 1000 p-y) and remained at that level at age 60 and older (16.2 per 1000 p-y).
A similar pattern applied to fragility fracture incidence. Overall incidence was moderately higher with than without HIV (4.6 versus 3.4 per 1000 p-y). For HIV-negative men incidence was 2.9 per 1000 p-y at ages 40-49, 2.9 at ages 50-59, and 5.1 at age 60 or older. For HIV-positive men fragility fracture incidence was 2.6 per 1000 at ages 40-49, 6.3 at ages 50-59, and 7.6 at age 60 or older.
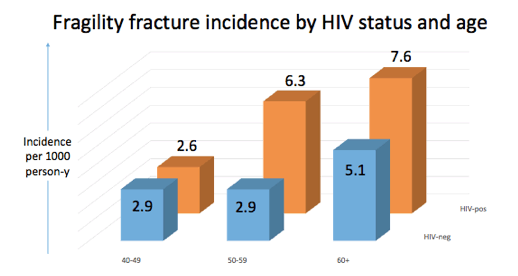
Compared with 40-49-year-old HIV-negative men, adjusted incidence rate ratio (aIRR) for all fractures was significantly higher in 50-59-year-old HIV-positive men (aIRR 1.92, 95% confidence interval [CI] 1.41 to 2.61, P < 0.001). Compared with 40-49-year-old HIV-negative men, adjusted fragility fracture incidence was significantly higher for 50-59-year-old HIV-positive men (aIRR 2.1, 95% CI 1.24 to 3.55, P = 0.006) and for 60-or-older HIV-positive men (aIRR 2.51, 95% CI 1.26 to 5, P = 0.009).
Cumulative use of TDF or protease inhibitors did not predict incidence of all fractures or fragility fractures.
"When current HIV-1 RNA more than 400 copies/ml was replaced by VCY (viremia copy years) in
the multivariable model, higher VCY was associated with all fractures [IRR: 1.14 per log10 increase in VCY (1.01, 1.30); P=0.042]. Neither HIV-1 RNA more than 400 copies/ml nor VCY were associated with higher incidence of fragility fractures"
All-fracture incidence was twice higher in 50-59-year-old men with HIV than in HIV-negative men the same age (aIRR 2.06, 95% CI 1.49 to 2.84). The same held for fragility fractures in this age group (aIRR 2.06, 95% CI 1.21 to 3.50).
Among other risk factors, only hypertension independently predicted all fractures (aIRR 1.32, 95% CI 1.04 to 1.69, P = 0.025). Unassociated factors were non-Caucasian race, BMI at or above 25, diabetes, HCV, eGFR below 60, current smoking, and moderate/heavy/binge drinking. None of these factors predicted fragility fractures.
Discussion/implications: All-fracture and fragility-fracture rates were twice higher in 50-59-year-old men with than without HIV, a finding leading the authors the propose that fracture incidence increases a decade earlier in men with HIV.
Results of this study confirm findings in the US HIV Outpatient Study linking age older than 47 in men and women to increased fracture risk after adjustment for possible confounders (hazard ratio 1.43 per 10 years for fragility fractures).
The authors suggest that the link between hypertension and fractures could reflect (1) loss of bone calcium with hypertension, (2) older age and greater likelihood of falling in people with hypertension, and/or (3) direct effects of antihypertensives on bone and in worsening orthostatic hypotension. "antihypertensive medications may increase the risk of fall injuries by causing or worsening orthostatic hypotension".
Key take-home: The researchers believe their findings support current US and European guidelines to conduct DXA bone mineral density screening for HIV-positive men 50 and older.
"We found no associations between the incidence of fractures and other factors like BMI, race, current
smoking, moderate-heavy or binge alcohol consumption, diabetes or HCV. Several studies have reported significantly higher rates of fractures in patients with HIV and HCV coinfection compared with those with HIV monoinfection, whereas others have not reproduced this finding. HCV has been shown to be a marker of intravenous drug use, and the higher risk of fracture in HIV-HCV coinfected patients has thus been attributed to direct consequences of drug use such as higher risk of trauma, falls and nutritional deficiencies. The small percentage of MACS participants reporting use of intravenous drugs (2%) might explain why no association was detected in our analysis."
"The role of HIV-specific factors in fracture risk remains uncertain. Although no association with ART exposure has been reported in several studies [3,9], others found higher rates of fractures associated with ART exposure. Using data from the ACTG Longitudinal-Linked Randomized Trial, Yin et al. found a significantly higher fracture rate in the first 2 years after ART initiation that declined in subsequent years. We found that current ART use was associated with an increased risk of fracture.
These findings are consistent with results from the Strategies for Management of Antiretroviral Therapy
substudy in which continuous administration of ART results in losses in BMD, whereas ART interruption was associated with BMD stabilization or increases [16]. Taken together, these findings suggest that ART treatment, regardless of the ART regimens used, has detrimental effects on bone health." [from Jules: this raises the question if integrase inhibitor use is different.]
Additional information:
⋅ McComsey GA, Tebas P, Shane E, et al. Bone disease in HIV infection: a practical review and recommendations for HIV care providers. Clin Infect Dis. 2010;51:937-946.
⋅ Young B, Dao CN, Buchacz K, Baker R, Brooks JT; HIV Outpatient Study (HOPS)
Investigators. Increased rates of bone fracture among HIV-infected persons in the HIV Outpatient Study (HOPS) compared with the US general population, 2000-2006. Clin Infect Dis. 2011;52:1061-1068.
⋅ Triant VA, Brown TT, Lee H, Grinspoon SK. Fracture prevalence among human immunodeficiency virus (HIV)-infected versus non-HIV-infected patients in a large U.S. healthcare system. J Clin Endocrinol Metab. 2008;93:3499-3504.
⋅ Shiau S, Broun EC, Arpadi SM, Yin MT. Incident fractures in HIV-infected individuals: a systematic review and meta-analysis. AIDS. 2013;27:1949-1957.
---------------------------------------------
One in 5 Older People With HIV Has an (Often Modifiable) IADL Disability - 2-3 Times higher rate of IADL in HIV+ Especially as HIV+ Age
"Our findings complement these other studies of older HIV-infected persons and highlight the 2 to 3 times higher rate of IADL impairment observed across cohorts compared to the general population, especially among middle-aged adults."
Report: Johs NA, Kunling Wu K, Tassiopoulos K, et al. Disability among middle-aged and older persons with human immunodeficiency virus infection. Clin Infect Dis. 2017; 65:83-91.
Background: "Older human immunodeficiency virus (HIV)-infected adults may experience higher rates of frailty and disability than the general population", as studies show they do. As people with HIV age, a growing burden of comorbidities may contribute to frailty and an escalating risk of disability. Test-measured frailty includes 5 domains--4-meter walk time, grip strength, and self-reported weight loss, exhaustion, and low activity. Disability can be assessed by 8 instrumental activities of daily living (IADLs)-- housekeeping, money management, cooking, transportation, telephone use, shopping, laundry, and medication management.
Objective: This cross-sectional analysis aimed to assess prevalence, overlap, and predictors of frailty and disability in an aging population of antiretroviral-treated people.
Methods: The study involved HIV-positive adults in AIDS Clinical Trials Group (ACTG) Study A5322 (HAILO), all of whom began antiretroviral therapy in an ACTG trial. HAILO enrolled participants in 2013-2014, all of them 40 or older.
At the HAILO baseline visit, participants completed the Lawton-Brody IADL Questionnaire, which assesses limitations in 8 tasks listed above. HAILO researchers defined frailty as limitations in 3 of the 5 domains listed above, prefrailty as limitations in 1 or 2 domains, and nonfrailty as limitations in no domain.
Researchers used weighted kappa statistics to determine agreement between IADLs (0, 1, or 2 or more categories) and frailty (nonfrail, prefrail, or frail). They used logistic regression modeling to pinpoint independent predictors of IADL impairment.
Study population: The study group included 1015 people with HIV, 44% of them 40 to 49 years old, 41% of them 50 to 59, and 15% 60 or older. Most participants, 81%, were men, 48% white, 29% black, and 20% Hispanic. Two thirds of participants had a CD4 count above 500 at baseline, and 94% had a viral load below 200 copies/mL.
Results: Sixty-two participants (6%) were frail 2 or more impairments in IADLs and 377 (37%) were prefrail. IADL assessment determined that 63 people (6%) had 2 or more impairments and 115 (11%) had 1 impairment. Among people with impairments, the most to least frequent were difficulties with housekeeping (48%), transportation (36%), shopping (28%), laundry (20%), money management (14%), cooking (15%), phone use (12%), and medication use (5%).
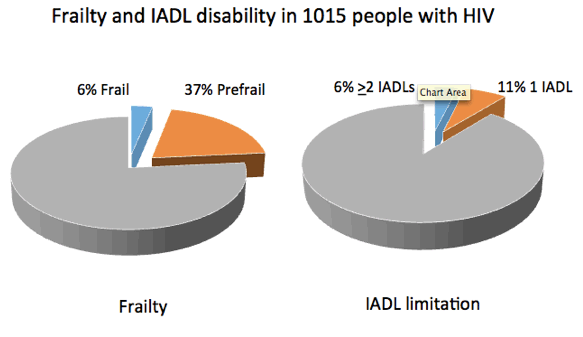
Figure 1. Frequency of any disability in instrumental activities of daily living among categories of non-frail, pre-frail, and frail participants in the Human Immunodeficiency Virus Infection, Aging, and Immune Function Long-Term Observational Study.
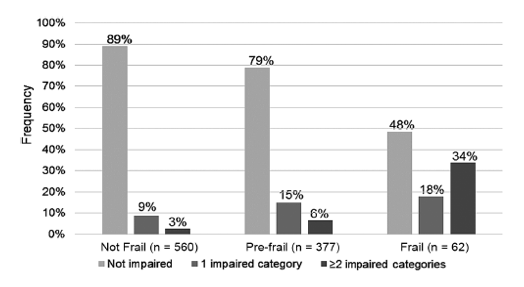
Among study participants assessed for both frailty and IADLs, 32 frail people (52%) and 80 prefrail people (21%) had at least 1 IADL. Only 62 nonfrail people (11%) had at least 1 IADL. But statistical agreement between frailty and IADLs was limited, with a weighted kappa below 0.18 (95% confidence interval [CI] 0.13 to 0.23).
Five variables--none of them specifically related to HIV infection--independently raised or lowered odds of disability indicated by IADL impairment, at the following adjusted odds ratios (aOR) and 95% CIs:
Lowered odds:
⋅ Private insurance (vs none): aOR 0.45 (0.27 to 0.75), P = 0.002
Raised odds:
⋅ High-school education or less (vs more): aOR 2.16 (1.38 to 3.4), P < 0.001
⋅ Current smoking (vs never): aOR 2.55 (1.54 to 4.24), P < 0.001
⋅ Under 3 days vigorous/moderate activity per week (vs more): aOR 1.95 (1.28 to 2.97), P = 0.002
⋅ Neurocognitive impairment at baseline: aOR 2.29 (1.4 to 3.76), P = 0.001
Prior smoking was not associated with IADL impairment.
Discussion/implications: The higher-than-expected disability rate as measured by IADLs, 18%, exceeded a recent US general-population estimate of 6% to 8% with disability.
Neurocognitive impairment was the only comorbidity linked to IADL disability. Unrelated comorbidities included cardiovascular disease, liver disease, HCV positivity, and diabetes. The researchers believe "neurocognitive function should be considered routinely when evaluating disability in older HIV-infected persons."
Two factors tied to IADL disability are modifiable--current smoking and low physical activity.
Another HAILO study reviewed above linked frailty and prefrailty to higher risk of recurrent falls.
Because of the cross-sectional nature of this analysis, one cannot assume that identified predictors of IADL disability caused disability.
Study participants were enrolled in antiretroviral treatment trials and observational studies for a median of 7.8 years and thus probably do not represent the broader HIV population in the United States.
Key take-home: Frailty and disability are not the same thing. They are distinct, "each with unique contributing factors and interventions for treatment and prevention."
"A recent publication using National Health and Nutrition Examination Survey (NHANES) data found a markedly lower (6%-8%) prevalence of IADL disability among participants aged 65-74 years compared to nearly 20% in our HIV-infected population aged ≥50 years. The prevalence of disability in HAILO is similar or slightly lower than what was found in 2 recent publications that reported on HIV-infected persons aged ≥50 years [16, 17]. One of these 2 studies, from Mexico City, Mexico, showed overall rates of disability similar to those seen in our cohort, with IADL impairment in 18% of participants aged ≥50 years. In contrast, the second study reported on a San Francisco, California cohort with IADL impairment of 39%, but included a higher proportion of participants aged >60 years. The San Francisco study population was drawn largely from a safety-net clinic where nearly all patients were publicly insured, while 41% of our population had private insurance. We postulate that the different disability rates among cohorts reflect differing perceptions of disability, differences in the family unit, and variable access to senior resources. Our findings complement these other studies of older HIV-infected persons and highlight the 2 to 3 times higher rate of IADL impairment observed across cohorts compared to the general population, especially among middle-aged adults".
"As in prior studies, neurocognitive impairment was a strong predictor of IADL; in our study it was the comorbidity most strongly associated with risk of IADL disability. Although earlier initiation of ART decreases the development of neurocognitive impairment among HIV-infected persons, several factors including ART toxicity, prior opportunistic infections, cerebrovascular disease, and age-associated cognitive decline may further adversely impact neurocognitive function among older, HIV-infected adults in the current ART era. Regardless of the causes, even mild cognitive impairment may result in a loss of function that impairs daily living and quality of life. Given these factors, neurocognitive function should be considered routinely when evaluating disability in older HIV-infected persons."
"....in the general older adult population there is evidence that socioeconomic disadvantages may exert cumulative adverse effects over an individuals lifetime. Thus, when considering the social and environmental aspects of disability, it is perhaps not surprising that these socioeconomic and lifestyle factors were more significant than other health or physical function-related measures..... Consistent with the concept of disability as a social phenomenon, we found a strong association between disability and socioeconomic (education, health insurance) and lifestyle factors (smoking, physical activity). While these socioeconomic variables may merely serve as a proxy for other key factors not readily ascertainable in our cohort, including financial resources, employment, and housing access, the prominence of social and economic factors as important factors in an individuals experience of disability is notable. The effects of socioeconomic factors on prevalent disability may be explained by the inability to mobilize resources. Aging HIV-infected adults may face unique challenges with use of services, including ageism at HIV-specific resource centers and HIV or gender identity/sexual identity stigma from other resource centers that cater to older adults in the general population, such as senior centers or churches. Many older, HIV-infected adults have tenuous relationships with family and therefore rely on other, often inconsistent, forms of social support when it comes to day-to-day needs. Economic challenges may be exacerbated by periods of temporary disability or participation restrictions, which in turn have an effect on the ability to return to work."
Additional information:
⋅ Fried LP, Ferrucci L, Darer J, Williamson JD, Anderson G. Untangling the concepts of disability, frailty, and comorbidity: implications for improved targeting and care. J Gerontol A Biol Sci. 2004;59:M255-M63.
⋅ Onen NF, Overton ET. A review of premature frailty in HIV-infected persons: another manifestation of HIV-related accelerated aging. Curr Aging Sci. 2011;4. doi 10.2174/1874609811104010033.
⋅ Centers for Disease Control and Prevention. Morbidity and Mortality Weekly Report. January 15, 2016/65(1):14. QuickStats: Percentage of adults with activity limitations, by age group and type of limitation--National Health Interview Survey, United States, 2014.
------------------------------------------------
Higher Risk of Heart Failure in Aging Vets With HIV, Regardless of Ejection Fraction
Report: Freiberg MS, Chang CH, Skanderson M, et al. Association between HIV infection and the risk of heart failure with reduced ejection fraction and preserved ejection fraction in the antiretroviral therapy era: results from the Veterans Aging Cohort Study. JAMA Cardiol. 2017;2:536-546.
Background: As people with HIV live longer thanks to antiretroviral therapy (ART), cardiovascular disease has become a worrisome contributor to morbidity and mortality. HIV populations run a higher risk of myocardial infarction and heart failure than the general population, but research has not determined which types of heart failure are more likely with HIV.
"Studies have shown that HIV infection increases the risk of HF independent of AMI9,10 and that the increased risk is higher among older people, individuals of black race/ethnicity, and those with obesity, hypertension, diabetes, current smoking, alcohol abuse or dependence, elevated HIV-1 RNA viral loads, or a history of AMI.....many HIV-infected individuals will survive with a damaged heart, and their health care professionals will have the challenge of preventing and managing HF in this high-risk population."
Objective: This 7-year study compared rates of four types of heart failure in veterans with versus without HIV in the Veterans Aging Cohort Study (VACS).
Methods: The VACS matches HIV-positive and negative veterans by age, sex, race/ethnicity, and VACS site. This analysis included VACS participants enrolled by April 1, 2003. The analysis excluded veterans who had cardiovascular disease before baseline.
Researchers analyzed heart failure rates from October 2015 to November 2016. They defined heart failure as failure with preserved ejection fraction (HRpEF, EF >50%), borderline HRpEF (EF 40% to 49%), heart failure with reduced EF (HRrEF, EF <40%), and heart failure of unknown type (EF missing).
The VACS team used Cox proportional hazards regression to estimate the hazard ratio for the association between HIV and each type of heart failure, adjusting for age, sex, race/ethnicity, and other relevant variables.
Study population: The analysis involved 98,015 veterans, 31,523 (32.2%) with HIV infection. The entire group averaged 48.3 years in age, and 97% were men. The largest proportion of vets, 48%, were black, while 38% were white and 8% Hispanic. Among veterans with HIV, 74% were taking ART, baseline median viral load stood at 1357 copies/mL, and baseline median CD4 count measured 382.
Results: Through a median follow-up of 7.1 years, 2636 heart failures occurred, 35.7% in veterans with HIV. Similar proportions of heart failures were HFpEF (34.6%) and HFrEF (37.1%), while 15.5% were borderline HFpEF and 12.8% HF of unknown type.
In the analysis adjusted for potential confounders, compared with HIV-negative veterans, those with HIV ran a 21% increased risk of heart failure with preserved ejection fraction (ejection fraction ≥50%) [aHR] 1.21, 95% confidence interval [CI] 1.03 to 1.41), a 37% higher risk of borderline heart failure with preserved ejection fraction (ejection fraction 40%-49%) (aHR 1.37, 95% CI 1.09 to 1.72), and a 61% higher risk of heart failure with reduced ejection fraction (ejection fraction <40%) (aHR 1.61, 95% CI 1.40 to 1.86).
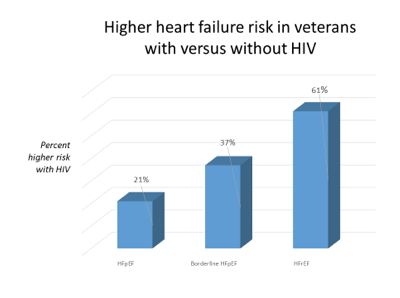
HIV remained an independent predictor of heart failure when the researchers restricted the analysis to veterans without hypertension (aHR 1.32, 95% CI 1.08 to 1.61), veterans without alcohol or cocaine abuse or dependence (aHR 1.43, 95% CI 1.25 to 1.65), veterans who never smoked (aHR 1.33, 95% CI 1.05 to 1.70), and veterans who had acute myocardial infarction during follow-up (aHR 1.58, 95% CI 1.37 to 1.82).
Higher risk of total HF with HIV held true with the analysis limited to whites (aHR 1.31, 95% CI 1.12 to 1.52), blacks (aHR 1.41, 95% CI 1.26 to 1.59), or veterans younger than 40 (aHR 2.41, 95% CI 1.60 to 3.63).
Analysis limited to veterans with HIV determined that time-updated CD4 count below 200 (versus 500 or higher) approximately doubled the risk of total HF (aHR 2.09, 95% CI 1.71 to 2.55), HFpEF (aHR 1.87, 95% CI 1.28 to 2.73), borderline HFpEF (aHR 2.10, 95% CI 1.30 to 3.39), HFrEF (aHR 1.87, 95% CI 1.36 to 2.57), and EF missing (aHR 3.37, 95% CI 1.95 to 5.84). Time-updated viral load above (versus below) 500 copies/mL raised the risk of total HF (aHR 1.31, 95% CI 1.12 to 1.53) and HFrEF (aHR 1.63, 95% CI 1.28 to 2.08).
Discussion/implications: Men with HIV run a higher risk of heart failure with preserved or reduced ejection fraction than demographically and behaviorally similar HIV-negative men.
Heart failure with reduced ejection fraction can occur in relatively young HIV-positive men, "decades earlier than might be expected among uninfected individuals."
The researchers believe the association between low time-updated CD4 count and heart failure "suggests that duration of HIV infection and, by extension, chronic inflammation, T-cell activation, and loss of adaptive immunity likely all have important roles" in development of heart failure.
"Most important, our data also suggest that even HIV-infected individuals with high CD4 cell counts are likely still at risk for HF compared with uninfected individuals, in part because HIV-infected individuals with high CD4 cell counts who are rapidly diagnosed, treated, and virally suppressed do not return to their pre-HIV levels of inflammation.
Moreover, this residual inflammation is associated with an increased risk of future non-AIDS diseases. In contrast, time-updated elevated HIV-1 RNA viral load was only significantly associated with HFrEF. These findings are consistent with reports before the ART era in which unsuppressed HIV viremia, perhaps through direct infection of cardiac myocytes or cardiac autoantibodies results in a cardiomyopathy consistent with HFrEF."
Key take-home: HIV providers "should focus on guideline-recommended HIV treatment and HF risk factor prevention (including diabetes, hypertension, renal disease, smoking, alcohol abuse and dependence, and obesity), as well as screening for HIV in individuals with new-onset HF where appropriate."
EDITORIAL: "...both young and old appear to be at risk and HFrEF is the major HF manifestation among those younger than 40 years old. These individuals, now into their fifth and sixth decades, represent a vulnerable group for which HFrEF has received scant attention. From a public health standpoint, more attention should be paid to heighten awareness of the comorbidity of HIV and HFrEF in the aging HIV-positive population and to design approaches to incorporate HF screening, risk estimation, and treatment.
Of the 2636 HF events, 941 were in PLWH and HFrEF was the single most common type (40%). Human immunodeficiency virus infection conferred a 41% increased risk (95% CI, 29%-54%) for total HF, with a stronger risk for developing HFrEF (HR, 1.61; 95% CI, 1.40-1.86) than HFpEF (HR, 1.21; 95% CI, 1.03-1.41). Risk for HF was greater among PLWH despite having a lower burden of traditional HF risk factors, including hypertension, diabetes, high low-density lipoprotein cholesterol level, and obesity. In subgroup analyses adjusted for numerous clinical variables, the HF risk was driven mainly by HFrEF for white, black, and young (<40 years of age) veterans."
"The elephant in the room, which this study was not designed to address, is the mechanism by which HIV infection and its treatment impact HF risk. The HF risk identified in this study appears to be independent of hypertension and myocardial infarction, which are the 2 leading determinants of HF risk in the general population. The extent to which this risk is driven by direct effects on the myocardium vs more systemic effects (such as inflammation or vascular injury) are uncertain. Can preclinical abnormalities be detected in PLWH using imaging (eg, myocardial strain imaging and diastolic dysfunction) or circulating biomarkers that predict incident HF? Which ART regimens are most "cardiac friendly"? Is the HFrEF seen in the group younger than 40 years old reversible with appropriate HF and HIV therapy? Last, treatment patterns for HF among PLWH remain poor.13 Effective approaches to improve HF medication use patterns for this group have also not been demonstrated. On these issues, there is great need and opportunity for further knowledge generation."
Additional information:
⋅ National Heart, Lung and Blood Institute. Heart failure.
⋅ American Heart Association. What is heart failure?
⋅ American Heart Association. Warning signs of heart failure.
⋅ Mayo Clinic. Heart failure.
⋅ Butt AA, Chang CC, Kuller L, et al. Risk of heart failure with human immunodeficiency virus in the absence of prior diagnosis of coronary heart disease. Arch Intern Med. 2011;171:737-743.
----------------------------------
Greater Brain Atrophy With HIV Over Age 60 Despite Viral Suppression - "older age >60 may be tipping point for faster atrophy"
Report: Clifford KM, Samboju V, Cobigo Y, et al. Progressive brain atrophy despite persistent viral suppression in HIV patients older than 60 years. J Acquir Immune Defic Syndr. 2017;76:289-297.
Background: As people live longer with HIV infection, longer exposure to the virus and to antiretroviral therapy (ART) pose growing risks to the central nervous system (CNS), including faster brain atrophy. ART-induced viral suppression in plasma may not control CNS inflammation and so may not prevent progressive brain injury.
"HIV-related brain injury prior to the cART era is well documented and includes global brain atrophy, subcortical volumetric reductions, and cognitive impairment.4-7 Despite increased access to cART, cognitive manifestations of HIV, classified in research settings as HIV-associated Neurocognitive Disorder (HAND), remain a substantial concern with frequency increasing with age. Some studies demonstrate CNS inflammation persisting despite suppressive cART, suggesting that cART alone is insufficient in preventing progressive brain injury. Autopsy studies further note persistent HIV DNA in brain tissue of virally suppressed individuals on stable cART.12 Detectable HIV DNA in the peripheral blood despite viral suppression is linked to smaller subcortical and cerebellar gray matter volumes. The literature is replete with cross-sectional analyses describing brain volumetric reductions associated with HIV serostatus, but do not typically examine rates of change and tend to include individuals not on cART or without plasma viral suppression."
Objective: This longitudinal study aimed to compare rates of brain volume changes in virally suppressed people 60 or older and age- and sex-matched HIV-negative people without neurocognitive impairment.
Methods: From May 2008 through January 2016, researchers recruited HIV-positive adults from the University of California, San Francisco HIV Over 60 Cohort. They excluded people with conditions that may confound an HIV-related cognitive diagnosis, including major neurological or psychological conditions, current or past brain infection, stroke, traumatic brain injury, active cancer, and current substance abuse or dependence. Everyone with HIV had taken ART for at least 12 months and had a viral load persistently below 400 copies/mL.
HIV-negative controls came from a longitudinal cohort assembled at the same center to study healthy brain aging. All met criteria indicating normal cognitive function. The investigators matched healthy controls to the HIV group by age and sex.
Everyone in both groups had at least 2 longitudinal magnetic resonance imaging (MRI) scans to measure changes in brain volume. For longitudinal comparisons, the researchers used two approaches: region of interest (ROI) and tensor-based morphometry (TBM).
Study population: The study focused on 38 people with HIV and 24 HIV-negative healthy controls. There were 2 women in each group. Age averaged 64.2 in the HIV group (range 60 to 76) and 65.4 in the control group (range 59 to 70).
Most people in the HIV group (89%) and the control group (96%) were white. Among people with HIV, estimated median duration of infection was 22.5 years, CD4 count averaged 574, and average viral load lay below 100 copies/mL. Median HIV suppression duration was 3.2 years.
Results: Fifteen of 38 people with HIV (39%) had HIV-associated neurocognitive disorder (HAND), 4 (11%) with asymptomatic neurocognitive impairment and 11 (29%) with mild neurocognitive disorder. Baseline cross-sectional ROI analysis determined that HIV-positive people with versus without HAND had significantly reduced volume in the cerebellum, nucleus accumbens, and brainstem. Compared with healthy controls, the HIV HAND group had significantly reduced volumes in the same 3 regions.
Through an average 3.4 years of follow-up in both study groups, longitudinal ROI analysis adjusted for age and sex documented a faster annualized rate of progressive brain atrophy in the HIV group in the cerebellum (0.42% vs 0.02%, P = 0.016), caudate (0.74% vs 0.03%, P = 0.012), frontal lobe (0.48% vs 0.01%, P = 0.034), brainstem (0.31% vs 0.01%, P = 0.026), and pallidum (0.73% vs 0.39%, P = 0.046), and in total cortical gray matter (0.65% vs 0.16%, P = 0.027).
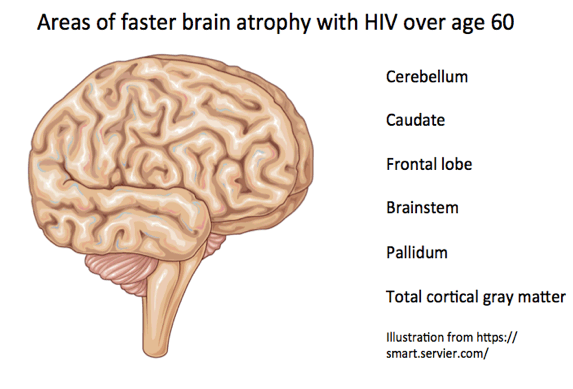
Within the HIV group, people with HAND did not have faster atrophy progression than people without HAND. But compared with HIV-negative controls, HIV-positive people with HAND had faster progressive atrophy in the cerebellum (0.50% vs 0.02%, P = 0.006), caudate (1.07% vs 0.03%, P = 0.015), temporal lobes (0.83% vs 0.18%, P = 0.049), brainstem (0.52% vs 0.01%, P = 0.005), pallidum (0.81% vs 0.39%, P = 0.047), and thalamus (0.82% vs 0.44%, P = 0.018), and in total cortical gray matter (0.87% vs 0.16%, P = 0.039).
Longitudinal TBM analysis indicated a faster atrophy rate in the right lateral cerebellum in the HIV group with HAND than in controls (P < 0.001).
Discussion/implications: The study documented "broad and sizably different longitudinal [brain] atrophy rates" in virally suppressed HIV-positive people 60 or older versus age- and sex-matched cognitively normal healthy adults.
The longitudinal nature of this analysis, the researcher believe, clarifies "that reduction in brain volumes over time in HIV are not simply archival damage from the pre-cART era." But it remains possible that pre-cART changes "set the stage for later amplified brain changes."
At the same time, the authors note, HIV-independent factors including smoking, cognitive reserve, and alcohol use could contribute to faster atrophy in people with HIV.
The researchers stress that most of the HIV patients in this study became infected in the 1980s, before combination ART became available, and "thus may differ from younger cohorts in brain vulnerability or survivor tendencies."
Key take-home: The faster brain atrophy documented in older people with HIV suggests "a potential gap in neuroprotection, despite adherence to cART with long-term maximal viral suppression in plasma."
"Our cross-sectional analyses confirm limited volumetric reductions in HIV-infected participants with cognitive impairment compared to healthy controls, including regions previously noted to be affected in the setting of HIV, such as the cerebellum, nucleus accumbens and brainstem".
"By virtue of the older age of our study participants compared to other studies, they may be particularly vulnerable to faster changes in brain atrophy as age-associated brain atrophy rates do not appear to have linear slopes in healthy aging. Furthermore, our data examined participants in age over 60 years, surpassing age set points thought to represent the 'tipping point' from linear to faster atrophy in presumed healthy aging. For example, a study of 1100 healthy elders noted nonlinear atrophy trajectories dominate in most subcortical regions after age 60 years."
"Better executive functioning performance was associated with larger total baseline volume of deep gray structures, particularly, the caudate (p=0.005) and nucleus accumbens (p=0.001). Similarly, we found associations between executive function performance and frontal (p=0.017), temporal (p=0.006) and total cortical gray matter volumes (p =0.010) and a positive association between motor performance and both cerebellum volume (p=0.001) and thalamus (p=0.003).
Over the duration of follow-up , cognitive status remained stable for 66% of participants (n=25), whereas 5/15 participants with HAND no longer met HAND criteria at follow-up and 8/23 without HAND at baseline met criteria at follow-up, consistent with the fluctuating pattern described by others. As a group, neuropsychological performance, measured as a summary z-score of all tests administered, did not change over time (p=0.585)."
"Yet, other factors could have influenced these faster rates of compared to healthy controls, including HIV-independent factors such as smoking, cognitive reserve, noted by lower total educational attainment in table 1, and alcohol use as well as HIV-associated factors, including chronic inflammation, medication effects, and small vessel ischemic disease (SVID). A history of alcohol abuse was reported in 7/38 (18%) HIV-infected participants with 6/38 (16%) reporting past abuse in substances besides alcohol (e.g. marijuana, cocaine, methamphetamine). Within the HIV-infected group, two participants
(5.3%) reported having a history of both alcohol and other substance abuse. In comparison, only 2/24 (8.3%) controls reported past substance abuse with none attributed to alcohol. Given that participants in each group were excluded for current substance use disorders, these differences are more likely to inform baseline volumes than longitudinal change...... Our findings in both grey and white matter suggest SVID alone is insufficient to explain our findings."
"In models adjusting for age and sex, HIV serostatus was associated with more rapid average annualized rates of atrophy in the cerebellum (0.42% vs. 0.02%, p=0.016), caudate (0.74% vs. 0.03%, p=0.012), frontal lobe (0.48% vs. 0.01%, p=0.034), total cortical gray matter (0.65% vs. 0.16%, p=0.027), brain stem (0.31% vs. 0.01%, p=0.026), and pallidum (0.73% vs. 0.39%, p=0.046). Among those with HIV, atrophy rates did not differ statistically by cognitive status. Despite persistent control of plasma viremia, these older HIV-infected participants demonstrate more rapid progressive brain atrophy when compared to healthy aging. Either HIV or other factors that differ between older HIV-infected participants and healthy controls could be responsible for these differences. Most of our participants were infected with HIV in the 1980s, before cART was accessible, thus may differ from younger cohorts in brain vulnerability or survivor tendencies. A previous study conducting similar cross-sectional analysis confers HIV-infection in conjunction with aging is related to cortical
volume loss .48 Our findings of no cross-sectional differences by serostatus alone was unexpected, suggesting the uniqueness of this sample of long-term survivors, although, differences emerged when examining those with HAND compared to controls. Nevertheless, we note progressive atrophy despite minimal baseline cross-sectional findings and can conclude that these changes are not limited to those with past injury. A lack of difference between HAND and non-HAND individuals further supports this contention. Our findings complement past work in this cohort noting Diffusion Tensor Imaging (DTI) deficits in white matter fiber integrity in older HIV participants compared to age-matched controls.24
Thus, we have identified multi-modal evidence of detrimental outcomes despite cART."
Additional information:
⋅ National Institute of Neurological Disorders and Stroke. Cerebral atrophy information page.
⋅ Valcour Lab. UCSF HIV Over 60 Cohort Study.
⋅ CHARTER. CNS HIV Antiretroviral Therapy Effects Research. A prospective observational cohort study.
⋅ Heaton RK, Franklin DR Jr, Deutsch R, et al. Neurocognitive change in the era of HIV combination antiretroviral therapy: the longitudinal CHARTER study. Clin Infect Dis. 2015;60:473-480.
-----------------------------
HIV Tied to Worse Health Quality of Life and Higher Chance of Depression
Report: Langebeek N, Kooij KW, Wit FW, et al. Impact of comorbidity and ageing on health-related quality of life in HIV-positive and HIV-negative individuals. AIDS. 2017;31:1471-1481.
Background: Despite virologic suppression with antiretroviral therapy, recent research documents worse health-related quality of life (HRQL) in people with HIV than in the general population. Reasons for this difference are potentially diverse yet poorly defined. HIV populations often include higher proportions of sexual and ethnic minorities than the general population, and they may differ in numerous behavioral and lifestyle factors. In addition, age-related comorbidities are often more prevalent in people with HIV. All these variables may affect HRQL.
"Currently, approximately 50% of individuals in care with HIV in The Netherlands are 45 years or older....even among well treated HIV-infected individuals without comorbidity or AIDS-defining events, life expectancy remains lower than in the general population. Differences in lifestyle factors, notably substance use, have also been demonstrated between HIV-infected and HIV-uninfected populations. Both minority status and lifestyle factors have previously been associated with a lower level of HRQL. Previous research has demonstrated that HIV infection and ageing also negatively impact HRQL".
"Previous research has demonstrated that HIV infection and ageing also negatively impact HRQL both in general terms and concerning the physical dimension of HRQL. Older HIV-infected individuals were previously shown to have a worse performance on physical function tests....depression is more prevalent among HIV-infected individuals than among HIV-uninfected individuals. The inflammatory pathway is a possible contributor to the high incidence of depression in HIV-positive individuals."
Objective: In this cross-sectional study AGEhIV Cohort investigators aimed to compare HRQL and depression in cohort members with versus without HIV and to assess potential contributions of comorbidity, age, and HIV infection to HRQL and depression.
Methods: AGEhIV recruited HIV-positive and negative participants at least 45 years old between 2010 and 2012. HIV-positive participants come from an Amsterdam HIV outpatient clinic and behaviorally similar HIV-negative people come from an Amsterdam sexual health clinic. For each person with HIV, researchers selected an HIV-negative person of the same sex and about the same age.
Researchers screened participants for common age-related comorbidities. Participants completed the Medical Outcomes Study Short Form 36-item survey (SF-36) to measure HRQL and the 9-item Patient Health Questionnaire (PHQ-9) and the 20-item Centers for Epidemiologic Studies depression scale (CES-D) to assess depressive symptoms.
The investigators used multiple linear regression analysis to identify factors independently associated with HRQL. They used multiple logistic regression analysis to identify factors independently associated with depression.
Study population: The analysis included 541 people with HIV and 524 without HIV. At enrollment age was similar in the two groups (about 52 years) and about 87% were men. The HIV group included a lower proportion of Dutch people (72% versus 81%, P = 0.001) and a lower proportion who attained a high educational level (42% versus 56%, P < 0.001). The proportion of heavy drinkers was similar in the two groups (about 6%), but the HIV group had a higher proportion of current smokers (32% versus 25%, P = 0.009) and a higher proportion who ever injected drugs (3.6% versus 1.1%, P = 0.017). Most people with HIV, 92%, had a viral load below 200 copies/mL before enrollment.
Results: In unadjusted analyses people with HIV reported significantly worse HRQL than HIV-negative people on 8 of 10 SF-36 scales, and they reported significantly more depression on the PHQ-9. After adjustment for all variables considered, HRQL on 6 of 10 SF-36 scales and depression remained significantly worse in people with HIV.
After adjustment in the comorbidity and age analysis, HIV infection remained independently associated with worse physical HRQL, worse mental HRQL, and greater likelihood of depression. A higher number of comorbidities remained independently associated with worse physical HRQL but not with mental HRQL or depression.
Younger age was significantly associated greater likelihood of depression and worse mental (but not physical) HRQL. Differences in HRQL between people with versus without HIV did not become greater with older age or higher number of comorbidities. "we cannot rule out survival bias, that is persons with poorest HRQL may not have lived long enough to be part of the oldest age categories".
Several variables were independently associated with worse physical HRQL, worse mental HRQL, or depression, as outlined in the following table.
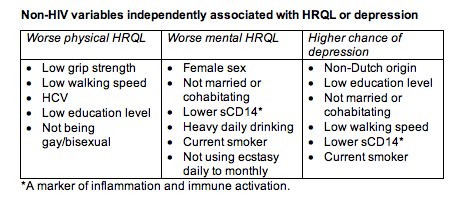
Discussion/implications: Compared with HIV-negative people the same age, this 45-or-older group with largely suppressed HIV infection had worse physical and mental HRQL and a higher likelihood of depression, even after adjustment for an array of potential confounders.
These HRQL differences did not become greater with age or with more comorbidities, but more comorbidities independently predicted worse physical HRQL.
Multivariate analysis linked younger age to worse mental HRQL and higher likelihood of depression.
The unexpected association between higher sCD14 (an inflammation/immune activation marker) and (1) better mental HRQL and (2) lower depression risk may have two explanations: (1) People with better mental health may take on more physiologically demanding occupational or social roles, which may increase systemic inflammation. (2) The cross-sectional nature of this analysis may miss a longitudinal link between inflammation/immune activation and mental HRQL or depression.
Key take-home: Prevention and management of comorbidities, including HCV infection, "may contribute to enhancing patients' HRQL." But the negative impact of HIV may remain even with optimal comorbidity prevention and management.
"HIV-positive status, a higher number of comorbidities, low walking speed, low grip strength and being HCV-infected were all significantly and independently associated with a reduced physical HRQL. To the extent that these factors are modifiable, our findings suggest that optimizing the prevention and management of comorbidities and treatment of HCV coinfection may contribute to enhancing patients' HRQL. However, our findings also suggest that a negative impact of HIV infection on HRQL will remain even in the presence of optimized prevention and management of comorbidities and HCV coinfection, because the impact of HIV infection was statistically independent of all other variables."
"Mental HRQL and depression were independently associated with HIV-positive status, lifestyle factors, that is substance use, and demographic characteristics. Substance use can be both a cause and a consequence of a low mental health or depression. Given the cross-sectional nature of our analysis, we cannot make inferences about the direction of this relationship. Female sex, non-Dutch origin, a low educational level and living alone were all associated with an increased risk of having a low mental HRQL and/or depression. HIV healthcare providers need to be alert to individuals with one or more of these characteristics as they may require more additional support or referral to mental health services."
"Our finding that HIV-positive status was significantly and independently associated with lower mental HRQL and more depression is in line with a large body of evidence showing a high prevalence of depression in HIV. Negative affect in HIV infection has previously been linked to stressors such as HIV stigma and discrimination and also to poverty, unemployment and social isolation, which are disproportionately prevalent in the HIV-infected population".
Additional information:
⋅ Amsterdam Institute for Global Health & Development. AGEhIV: Comorbidity and Aging With HIV.
⋅ Miners A, Phillips A, Kreif N, et al. Health-related quality-of-life of people with HIV in the era of combination antiretroviral treatment: a cross-sectional comparison with the general population. Lancet HIV. 2014;1:e32-40.
⋅ Schouten J, Wit FW, Stolte IG, et al. Cross-sectional comparison of the prevalence of age-associated comorbidities and their risk factors between HIV-infected and uninfected individuals: the AGEhIV cohort study. Clin Infect Dis. 2014;59:1787-1797.
⋅ Cholera R, Pence BW, Bengtson AM, et al. Mind the gap: gaps in antidepressant treatment, treatment adjustments, and outcomes among patients in routine HIV care in a multisite U.S. clinical cohort. PLoS One. 2017;12:e0166435.
----------------------------------------------
More Psychiatric Symptoms in People Older Than 60 With vs Without HIV
"....substantial neurobehavioral burden in a sample of HIV-infected participants aged 60 and older, regardless of cognitive status, compared to historical healthy controls."
Report: Milanini B, Catella S, Perkovich B, et al. Psychiatric symptom burden in older people living with HIV with and without cognitive impairment: the UCSF HIV over 60 cohort study. AIDS Care. 2017;29:1178-1185.
Background: Cognitive impairment, already a frequent complication of HIV infection, seems likely to become more common and pronounced as the HIV population continues to age. Mental health problems also affect a high proportion of people with HIV and can contribute to poor medication adherence, difficulties in everyday living, and worse quality of life. But potential interactions between HIV-associated neurocognitive disorder (HAND) and mental health conditions remain poorly understood in older people with HIV.
"The frequency of cognitive symptoms is substantially higher among older PLWH, a population that is rapidly growing due to widespread use of cART. Mental health comorbidities are common in HIV and mood symptoms such as depression and apathy. Neuropsychological impairment in the setting of HIV is known to include cognitive, motor and behavioral attributes; thus, it is likely that individuals with HAND in the late cART era have the highest psychiatric symptom burden."
Objective: This cross-sectional study aimed to compare neuropsychiatric symptom burden in older HIV-positive people with versus without HAND and to compare frequency of those symptoms in the HIV population and healthy elderly controls.
Methods: HIV-positive participants came from the University of California, San Francisco (UCSF) HIV Over 60 Cohort, a prospective study of cognition and function requiring once-yearly visits. Participants completed a standard 90-minute neuropsychological testing battery allowing a consensus panel to determine whether a person had HAND.
The study group also completed the Neuropsychiatric Inventory Questionnaire (NPI-Q) and the Geriatric Depression Scale (GDS). Participants provided contact information for a spouse, close friend, or family member. Interviews of this proxy using the Clinical Dementia Rating scale provided a global rating of functional capability. The proxy could also complete the NPI-Q.
A 62- to 79-year-old HIV-negative control group came from a study of healthy aging at UCSF's Memory and Aging Center. All control participants reported normal everyday function and denied cognitive symptoms.
"At the time of study entry, all HIV-infected participants reported being free of current substance abuse".
Study population: The HIV group consisted of 39 people with HAND and 35 without HAND. Among HIV-positive people with HAND, 16 (41%) had asymptomatic neurocognitive impairment and 23 (59%) had mild neurocognitive disorder.
Median age was 67 in both HIV groups, compared with 72 in 236 HIV-negative controls (P < 0.001). About 95% of people with HIV were men, compared with 39% of controls (P < 0.001). Years of education averaged 15.7 in the HAND group, 16.7 in the non-HAND group (P = 0.096), and 17.1 in controls (P = 0.008 versus HIV group). Respective proportions of whites were 87%, 94%, and 92% (no significant differences). The HAND and non-HAND groups did not differ in clinical factors that might affect psychiatric symptom burden.
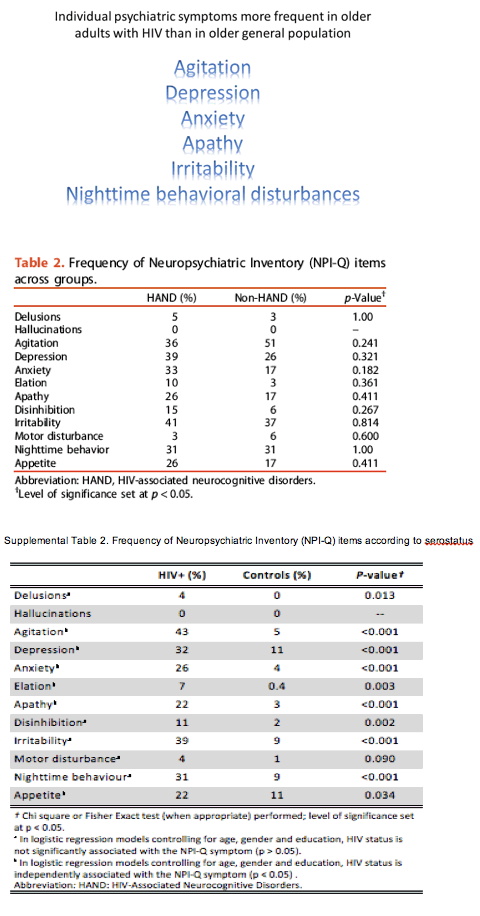
Results: HIV-positive people with HAND did not differ from those without HAND in psychiatric symptoms as defined by total NPI-Q score (mean 2.64 versus 2.14, P = 0.319) or individual symptoms on the NPI-Q. Nor did the HAND group differ significantly from the non-HAND group in NPI-Q clusters (mood, agitation, psychosis, or restlessness).
"The frequency of total scores at or above the standard GDS clinical cutoff of 10 (range = 10-30) varied by group, with 46% of the HAND group having mild-to-severe depressive symptoms compared to 30% for the non-HAND group (Figure 2). HIV-infected participants endorsed a higher number of depressive symptoms compared to historical controls (mean = 2.67 (± 2.85); p < 0.001), of which 3% endorsed mild-to severe depressive symptoms."
Compared with elderly HIV-negative controls, both HIV groups had significantly higher frequency of 6 individual psychiatric symptoms: agitation, depression, anxiety, apathy, irritability, and nighttime behavior disturbances (P < 0.05). The HIV groups also had significantly higher rates of three NPI-Q clusters than the control group: mood, agitation, and restlessness (P < 0.001).
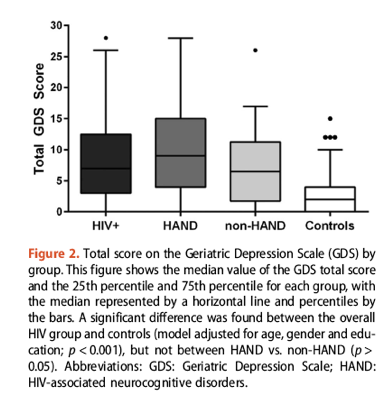
Average total GDS score did not differ significantly between the HAND group and the non-HAND group (9.72 and 6.68, P = 0.064). But a higher proportion of the HAND group than the non-HAND group had a total GDS score above a standard clinical cutoff of 10 (46% versus 30%). In a model adjusted for age, gender, and education, the combined HIV group had a significantly higher total GDS score than did HIV-negative controls (P < 0.001).
Discussion/implications: HIV-positive people 60 or older--with or without HAND--had significantly greater psychiatric symptom burden than elderly HIV-negative people at the same center. This finding provides "evidence that neuropsychiatric symptoms were not associated with cognitive functioning in older persons living with HIV."
Use of both self-report and proxy-report to assess neuropsychiatric burden lowered chances that lack of self-insight by cognitively impaired people with HIV affected findings.
"The frequency and severity of neuropsychiatric symptoms were significantly higher in HIV compared to healthy elders:"
The higher psychiatric symptom burden with HIV is even more worrisome because older people with HIV are less likely to receive mental health care than younger people with HIV.
Key take-home: Regular psychiatric symptom screening of elderly people with HIV is essential and may be more effective if a caregiver or proxy is engaged.
Additional information:
⋅ Valcour Lab. UCSF HIV Over 60 Cohort Study.
⋅ Fazeli PL, Crowe M, Ross LA, et al. Cognitive functioning in adults aging with HIV: a cross-sectional analysis of cognitive subtypes and influential factors. J Clin Res HIV AIDS Prev. 2014;1:155-169.
⋅ Castellon SA, Hardy DJ, Hinkin CH, et al. Components of depression in HIV-1 infection: their differential relationship to neurocognitive performance. J Clin Exp Neuropsychol. 2006;28:420-437.
⋅ Cysique LA, Deutsch R, Atkinson JH, et al. Incident major depression does not affect neuropsychological functioning in HIV-infected men. J Int Neuropsychol Soc. 2007;13:1-11.
⋅ Cholera R, Pence BW, Bengtson AM, et al. Mind the gap: gaps in antidepressant treatment, treatment adjustments, and outcomes among patients in routine HIV care in a multisite U.S. clinical cohort. PLoS One. 2017;12:e0166435.
-------------------------------------------
Almost 40% of Aging HIV/HCV Group Have Potential DAA Interaction - 77% ≥60 Have Potential DAA Interaction
Report: Smolders EJ, Smit C, de Kanter CTMM, et al. High need to switch cART or comedication with the initiation of DAAs in elderly HIV/HCV-coinfected patients. J Acquir Immune Defic Syndr. 2017;76:193-199.
Background: An estimated 2.3 million people worldwide have HIV/HCV coinfection. Antiretroviral therapy (ART) made HIV infection a chronic disease, and direct-acting antivirals (DAAs) made HCV infection a usually curable disease. Both antiretrovirals and DAAs may be "victims" or "perpetrators" of possibly dangerous drug-drug interactions. In the aging HIV/HCV population, many people also rely on medications for somatic and psychiatric comorbidities, and DAAs may also interact with these medications.
Objective: This retrospective cross-sectional study set out to identify comedication use in people with HIV/HCV coinfection and to predict potential drug-drug interactions.
Methods: The study group came from ATHENA, a nationwide observational cohort in the Netherlands. This analysis included all ATHENA participants known to be HIV/HCV-positive on January 1, 2015. None had been treated with DAAs at that point.
Researchers determined which antiretrovirals and other comedications each person took. Then they used the University of Liverpool hepatitis drug-interaction tool to identify potential drug-drug interactions between these agents and DAAs they might start. The investigators classified comedication-DAA pairs as (1) no interaction expected, (2) possible interaction expected, (3) contraindication, or (4) no information available in Liverpool database. The antiretroviral interaction analysis included only patients taking two nucleos(t)ide reverse transcriptase inhibitors plus one additional boosted or unboosted agent.
Study population: ATHENA included 777 HIV/HCV-coinfected people on January 1, 2015, 86% of them men. Median age stood at 49.3 years (range 23 to 80). Eighty-eight people (11%) were 60 or older. More than half of participants, 56%, had a METAVIR score of F0-2, and 23% were F3-4. Almost two thirds of coinfected people had HCV genotype 1 or 4.
Results: Among the 777 people studied, 488 (63%) used 156 unique nonantiretroviral comedications, with the number of prescriptions ranging from 1 to 14. The most frequently prescribed comedications were drugs for opioid dependence (11% of prescriptions), proton pump inhibitors (9%), calcium supplements (6%), and selective serotonin reuptake inhibitors, platelet aggregation inhibitors, vitamin D, and statins (4% each).
Among the 488 people taking a nonantiretroviral comedication, 299 (61%) had a potential type 2 or 3 interaction with DDAs (see Methods for types). Among all 777 study participants, 38% had a potential type 2 or 3 comedication-DAA interaction.
While 40% of people younger than 60 risked such an interaction, 77% who were 60 or older faced a potential type 2 or 3 interaction. Whereas 34% of people without cirrhosis risked a type 2 or 3 comedication-DDA interaction, 55% with cirrhosis faced such a risk. Sofosbuvir/simeprevir and ombitasvir, paritaprevir, and ritonavir with or without dasabuvir (PrO+/-D) accounted for the highest number of potential interactions, while grazoprevir/elbasvir and sofosbuvir/velpatasvir accounted for the lowest number.
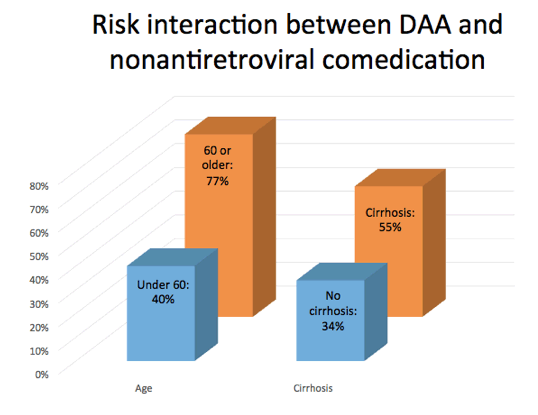
Among all 777 study participants, 762 (98%) took antiretrovirals. Almost three quarters of participants, 70%, took tenofovir/emtricitabine, while 14% took abacavir/lamivudine. The most frequently used additional antiretroviral classes were nonnucleosides (46%), boosted protease inhibitors (37%), and integrase inhibitors (17%).
Grazoprevir/elbasvir would cause the most type 3 drug interactions and would require changing either the DAA regimen or the ART regimen. Two other DAA combinations had potential type 3 interactions--sofosbuvir with velpatasvir/simeprevir and PrO+/-D.
No one with genotype 1 or 4 treated with sofosbuvir/daclatasvir would have to change their ART regimen. But the dose of daclatasvir would have to be adjusted with some antiretrovirals. Ledipasvir and velpatasvir combined with sofosbuvir could be used safely with all third antiretrovirals. But 199 people (31%) with genotype 1 or 4 used tenofovir with a boosted protease inhibitor and would have to switch either tenofovir or the protease inhibitor.
Discussion/implications: "Comedication use in the aging HIV/HCV population is frequent and diverse [and] there is a high potential for drug-drug interactions between DAAs plus comedications/cART."
People 60 or older had a higher potential risk of DAA-comedication interactions than did younger people. And people with cirrhosis ran a higher interaction risk than people without cirrhosis.
Sofosbuvir/simeprevir and PrO+/-D had the highest number of potential interactions with nonantiretroviral comedications. Both simeprevir and ritonavir (part of PrO) are CYP3A4 inhibitors.
Grazoprevir/elbasvir had the fewest potential interactions with nonantiretroviral comedications and the most potential interactions with antiretrovirals. Grazoprevir/elbasvir and simeprevir are contraindicated with boosted HIV protease inhibitors, nonnucleosides (except rilpivirine), and elvitegravir/cobicistat.
Key take-home: Clinicians prescribing DAAs with other medications should plan treatment with a pharmacologist versed in use of all relevant agents and with reliable drug-drug interaction tools such as the University of Liverpool HIV and Hepatitis Drug Interaction Checkers.
Additional information:
⋅ University of Liverpool: Hep Drug Interaction Checker.
⋅ University of Liverpool: HIV Drug Interaction Checker.
⋅ El-Sherif O, Khoo S, Solas C. Key drug-drug interactions with direct acting antiviral in HIV-HCV coinfection. Curr Opin HIV AIDS. 2015;10:348-354.
⋅ Poizot-Martin I, Naqvi A, Obry-Roguet V, et al. Potential for drug-drug interactions between antiretrovirals and HCV direct acting antivirals in a large cohort of HIV/HCV coinfected patients. PLoS One. 2015;10:e0141164
|
|
| |
| |
|
|
|