| |
Bone Mineral Density and Parathyroid Hormone as Independent Risk Factors for Mortality in Community-Dwelling Older Adults: A Population-Based Prospective Cohort Study in Brazil. The São Paulo Ageing & Health (SPAH) Study
|
| |
| |
Download the PDF here
from Jules: you can see in the Geriatric Syndrome study done in SF and reported at CROI 2015 in HIV+ aged 50-74, avg age 57, 25% had at least 1 fall. In WIHS women had high rates of falls, where I think perhaps the risk for falls may be greater. Mortality/death rates in older aging HIV+ are underestimated because its impossible to predict how one will respond to a comorbidity and as HIV+ age with multiple comorbidities we cannot predict how this will affect death/mortality & quality of life.
"This study demonstrated for the first time that, in addition to the established clinical risk factors for mortality in the elderly population, low bone density at the hip and higher plasma levels of PTH were independent predictors of all-cause and cardiovascular mortality in a population-based, prospective study of community-dwelling older adults. These effects were independent of recognized cardiovascular predictors of death and risk factors associated with mineral metabolism. Moreover, higher PTH was associated with a higher risk of mortality, even in individuals with PTH in the normal range (plasma levels 50 to 65 pg/mL) without other signs of disturbed bone metabolism, such as abnormal serum calcium, renal dysfunction, or vitamin D deficiency. These findings suggest that plasma levels of PTH provide prognostic information, even in the absence of primary or secondary hyperparathyroidism.
Low proximal femoral BMD was found to be associated with an increased subsequent risk of death from all causes after controlling for age and other potential confounding factors. Several studies have found that baseline BMD is inversely related to subsequent mortality in elderly women and men.[7-9, 11, 12, 34] Further, in a large cohort of white women 65 years of age and older, a greater decline in BMD was associated with increased mortality from all causes and CVD, regardless of initial BMD.[35]
.......Analyzed as a group, the subjects with low bone mass (T-score < -1) had an increased risk of cardiovascular death compared with those with normal BMD. Consequently, further in vitro and in vivo research is needed to determine whether a biological mechanism exists for an independent association between BMD and mortality or whether BMD is a nonspecific indicator of frailty, ill health, or other comorbidities.
In conclusion, this study showed that low BMD at the hip and higher plasma levels of PTH, but not vitamin D levels per se, were predictors of all-cause and cardiovascular mortality in healthy community-dwelling older adults, independent of vitamin D status, renal function, bone mass, and comorbidities. Taken together, these findings support the notion that more attention should be paid to these factors in clinical practice to improve the management of the elderly population......Although the lowest quartile of 25OHD levels (<13 ng/mL) was associated with mortality risk in the unadjusted analyses, the multivariate model showed no significant association between 25OHD levels and the risk of all-cause mortality in this cohort, regardless of whether 25OHD was analyzed as quartiles, as clinical categories, or as a continuous variable. This finding contrasts with recent large population-based studies[42-45] in which lower 25OHD levels were related to a higher mortality risk."
Falls occur often in the elderly and may lead to serious trauma. The risk of falling established in previous research varies from 28-35% per year among people aged 65 years and older who live in the community(1-4) and from 45-70% among people who live in nursing homes.(5,6) The incidence of falls is higher in women than in men and rises with increasing age.(7,8) About 10% of falls among elderly residing in the community results in serious injuries, with 1-2% of them resulting in hip fractures, 3-5% in fractures at other sites, and 5% in soft tissue injuries and head trauma.(2,9-11) Even if no physical injury occurs, frequent falls can have a serious psychological impact by inducing fear of falling, which can lead to self-imposed restrictions in activity, decreased mobility, and increased dependency.(1,12) The costs associated with falls and fall injuries are high, especially the cost spent on hip fractures. Fractures of the hip lead often to hospital admission and necessitate long-term rehabilitation or nursing home care.
CROI: Geriatric Syndromes are Common Among Older HIV-Infected Adults- (03/16/15)
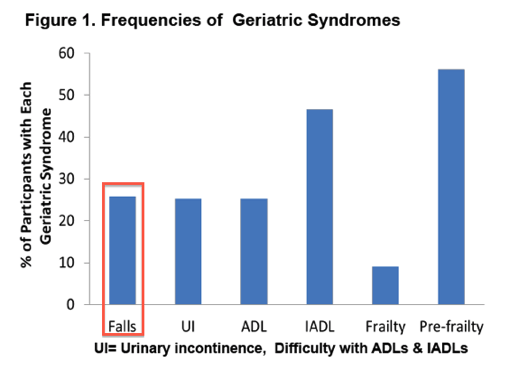
Geriatric Syndromes in Older HIV-Infected Adults...at 57 age...Participants had a median of 4 (IQR: 3-6) comorbidities & were taking a median of 9 (6-12) nonantiretroviral medications & (53.6%) had 2 or more geriatric syndromes - (05/20/15)
HIV+ Falls, Fractures & Death...."impending epidemic of bone fractures".....federal officials & others continue & persist to ignore this problem - (08/12/16)
-----------------------------------------
ABSTRACT
Previous studies have shown a relationship between osteoporosis and increased mortality risk. However, none of these studies performed a concomitant evaluation of the parathyroid hormone (PTH)-calcium-vitamin D axis and bone mass to accurately determine the contribution of each of these parameters to survival in older subjects. Thus, we sought to investigate the association between bone parameters and mortality in a longitudinal, prospective, population-based cohort of 839 elderly subjects. Clinical data (including history of fractures and cardiovascular events) were assessed using a specific questionnaire. Laboratory exams, including serum 25OHD and PTH, were also performed. Bone mineral density (BMD) at the lumbar spine and hip were evaluated using DXA. All analyses were performed at baseline (2005 to 2007). Mortality was recorded during follow-up. Multivariate Cox proportional regression was used to compute hazard ratios for all-cause and cardiovascular mortality. Over a mean 4.06 ± 1.07 years, there were 132 (15.7%) deaths. These individuals were compared to 707 subjects who were alive at the end of the coverage period for mortality data collection.
In a multivariate Cox proportional hazards model, age (HR 1.32; 95% CI, 1.13 to 1.55; p = 0.001, for each 5-year increase), male gender (HR 1.90; 95% CI, 1.30 to 2.79; p = 0.001), recurrent falls (more than two in the previous year; HR 1.65; 95% CI, 1.06 to 2.56; p = 0.026), diabetes mellitus (HR 2.17; 95% CI, 1.46 to 3.21; p < 0.001), low physical activity score (HR 1.78; 95% CI, 1.14 to 2.79; p = 0.011), prior cardiovascular event (HR 1.76; 95% CI, 1.18 to 2.63; p = 0.006), total hip BMD (HR 1.41; 95% CI, 1.15 to 1.72; p = 0.001, per each 1 SD decrease), and intact PTH (iPTH) (HR 1.06; 95% CI, 1.04 to 1.08; p < 0.001, per each 10 pg/mL increase) were independently associated with all-cause mortality. The subjects in the highest quartile of PTH (>49 pg/mL) were at a higher risk of cardiovascular death (HR 3.09; 95% CI, 1.36 to 6.99; p = 0.007) compared with the subjects in the lowest quartile (<26 pg/mL). Low BMD and higher PTH were significantly associated with mortality in community-dwelling older adults. These findings support the notion that careful screening of these bone parameters might lead to better management of older patients and improve outcomes in this population.
Introduction
Osteoporotic fractures, particularly hip and vertebral fractures, are linked with adverse outcomes[1-3] including increased mortality.[1, 4-6] Low bone mineral density (BMD) has also been associated with an increased mortality risk independent of fragility fracture.[7-10] However, the majority of studies concerning low BMD and mortality have focused on white populations[9, 11]; a smaller number have included ethnic minorities.[12, 13] Some epidemiological studies have also suggested that mineral metabolism biomarkers, especially low serum 25-hydroxyvitamin D (25OHD) concentrations and higher intact parathyroid hormone (iPTH) levels, separately or in combination, are related to mortality among institutionalized older adults[14] and community-dwelling elderly people without primary hyperparathyroidism.[15]
Several biological mechanisms have been proposed to explain a possible causal relationship between vitamin D deficiency or elevated PTH and increased mortality risk, especially cardiovascular features, including atherosclerosis,[16, 17] left ventricular hypertrophy,[18, 19] and arterial stiffness and hypertension.[20]
However, these associations between bone parameters (BMD, 25OHD, and iPTH) and mortality in the general population are still controversial.[21, 22] Moreover, previous studies of mineral metabolism and mortality risk have generally evaluated BMD, 25OHD, and iPTH concentrations separately. No study has performed a concomitant evaluation of the PTH-calcium-vitamin D axis and bone mass, and this is essential for determining the contribution of each of these parameters to survival in older subjects. Thus, in the present study, we investigated BMD, bone metabolism biomarkers, and clinical variables as potential independent predictors of all-cause and cardiovascular mortality in a population-based cohort of community-dwelling elderly men and women in Sao Paulo, Brazil.
Discussion
This study demonstrated for the first time that, in addition to the established clinical risk factors for mortality in the elderly population, low bone density at the hip and higher plasma levels of PTH were independent predictors of all-cause and cardiovascular mortality in a population-based, prospective study of community-dwelling older adults. These effects were independent of recognized cardiovascular predictors of death and risk factors associated with mineral metabolism. Moreover, higher PTH was associated with a higher risk of mortality, even in individuals with PTH in the normal range (plasma levels 50 to 65 pg/mL) without other signs of disturbed bone metabolism, such as abnormal serum calcium, renal dysfunction, or vitamin D deficiency. These findings suggest that plasma levels of PTH provide prognostic information, even in the absence of primary or secondary hyperparathyroidism.
The SPAH is a longitudinal population-based study with several strengths, including a high participation rate and a homogeneous and well-characterized community-dwelling sample that is likely representative of the Brazilian elderly population.[33] Furthermore, in previous studies, the non-exclusion of vitamin D supplementation, bisphosphonate use, primary hyperparathyroidism, and chronic renal disease may hamper interpretation and misrepresent the influence of the pivotal role of PTH and BMD in mortality in the elderly population. Finally, we investigated a more comprehensive set of risk factors, including laboratory and BMD parameters, than those examined in other cohorts.[8, 9, 12, 21, 22]
Low proximal femoral BMD was found to be associated with an increased subsequent risk of death from all causes after controlling for age and other potential confounding factors. Several studies have found that baseline BMD is inversely related to subsequent mortality in elderly women and men.[7-9, 11, 12, 34] Further, in a large cohort of white women 65 years of age and older, a greater decline in BMD was associated with increased mortality from all causes and CVD, regardless of initial BMD.[35]
The mechanism of this association is unknown. Studies in the elderly population have found a significant relationship between osteoporosis and mortality in excess, mostly caused by CVD/atherosclerotic diseases[35, 36] and frailty syndrome.[37] Beyond the potential cardiovascular confounders, when studying BMD in relation to mortality, it is important to thoroughly investigate whether a low BMD is not merely a marker of underlying illness.[11, 12] A very ill person will undertake less physical activity, resulting in a lower BMD. In the SPAH population, however, two important parameters related to frailty (low physical activity score and recurrent falls) were predictors of mortality risk, independent of BMD. Other components of the frailty syndrome, such as low appendicular muscle mass and a history of clinical fractures, were not significantly associated with death in this population, most likely because they are confounders for the association between BMD and mortality. In fact, baseline hip and humerus fractures increased the unadjusted risk of overall mortality, but this association was altered by adjustment for BMD. Nevertheless, we did not specifically evaluate the lower limb disability index or self-assessed poor health, which are more precise markers of fragility. Moreover, the relationship between BMD and all-cause mortality was stronger than the relationship between BMD and cardiovascular deaths, although the borderline significance of the risk ratios for cardiovascular death according to the stratification by T-score (osteopenia or osteoporosis) might be related to the limited size of the sample, particularly the number of cardiovascular deaths. Analyzed as a group, the subjects with low bone mass (T-score < -1) had an increased risk of cardiovascular death compared with those with normal BMD. Consequently, further in vitro and in vivo research is needed to determine whether a biological mechanism exists for an independent association between BMD and mortality or whether BMD is a nonspecific indicator of frailty, ill health, or other comorbidities.
Studies investigating the association between circulating PTH and the death risk in elderly persons living in the community are scarce.[15, 21, 22] Here, we described the prospective association between plasma PTH and all-cause and cardiovascular mortality in a community-based sample. Moreover, our data confirmed two previous studies reporting that higher plasma PTH was associated with a higher incidence of death, even in individuals with PTH in the normal range.[15, 22]
Several mechanisms have been proposed to explain the link between PTH and mortality, such as atherogenesis via vascular calcification,[38] the induction of left ventricular hypertrophy,[18, 19] renal dysfunction,[39] and noncalcemic effects such as direct effects on vascular smooth muscle cells[40] and impaired myocardial energy utilization.[41] In this study, after adjustment for established cardiovascular risk factors and factors related to mineral homeostasis (including calcium, phosphorus, vitamin D levels, and renal function) in the SPAH study, PTH remained associated with a greater risk of overall and cardiovascular mortality, suggesting that confounding from these factors seems not to explain our findings. However, although no major confounding was detected in our multivariable model, we cannot rule out the possibility of residual confounding from unmeasured factors.
To date, there is no evidence that reducing PTH levels will decrease cardiovascular risk in the general population. Because our study was observational, our results do not allow us to fully determine whether PTH per se is a causal factor or to provide a direct benefit of a reduction of PTH levels. However, the results emphasize that further interventional studies are necessary to examine whether a reduction in PTH levels corresponds to a reduction in cardiovascular events in the community.
Although the lowest quartile of 25OHD levels (<13 ng/mL) was associated with mortality risk in the unadjusted analyses, the multivariate model showed no significant association between 25OHD levels and the risk of all-cause mortality in this cohort, regardless of whether 25OHD was analyzed as quartiles, as clinical categories, or as a continuous variable. This finding contrasts with recent large population-based studies[42-45] in which lower 25OHD levels were related to a higher mortality risk. A Mendelian randomization analysis of three large Danish cohorts including a impressive number of 95,766 participants suggested that genetically low plasma 25OHD concentrations (lower than 20 nmol/L, that is equivalent to 8 ng/mL) were associated with increased all-cause mortality.[46] These divergent results might be explained by some differences between the current study and previous reports. First, if the association between vitamin D status and mortality could be driven by subjects with 25OHD levels in this extremely low range (<10 ng/mL), we may not have had the ability to detect an effect because of the limited number of individuals with very low 25OHD levels (<20%). Second, unlike some studies,[42, 47] in the SPAH population serum 25OHD was measured using a radioimmunoassay that is considered to overestimate the 25OHD level because the antibody used in the assay also binds 24,25-dihydroxyvitamin D; this limitation could influence the results toward the null hypothesis. Third, unlike some studies that included people with physical disabilities and thus reduced mobility,[14] the SPAH study included only well-functioning older subjects, many of whom still held jobs that required sun exposure, which may have been related to higher 25OHD levels. Alternatively, it may be that the limited size of the sample reduced the study's statistical power to detect the association between vitamin D status and mortality. Even though, our finding of non-association between lower 25OHD concentrations and cardiovascular mortality is similar to results of the recent Danish cohort study with sufficient statistical power to detect an association of vitamin D and cardiovascular deaths.[46]
Higher iPTH levels are most likely due to low vitamin D levels and/or impaired renal function. Nevertheless, the relationship between PTH and 25OHD levels is complex, and there is a modest association between these hormones.[22] In our data, 25OHD levels were not retained in the final multivariate models after adjustment for other confounders, including BMD and PTH. Even after excluding those individuals of lower levels of 25OHD and eGFR in this cohort, PTH remained as an independent predictor of mortality, suggesting that PTH may be a more important risk factor for mortality or may even mediate the relationship between 25OHD and mortality.
Limitations of the study include the limited sample size and the exclusion of individuals from the original SPAH cohort who were not known to be alive at the time of the censoring analysis. Moreover, plasma PTH levels and other variables involved in bone metabolism were measured only at the baseline assessment, and it is unclear how well a single measurement reflects the PTH levels during the follow-up.
In conclusion, this study showed that low BMD at the hip and higher plasma levels of PTH, but not vitamin D levels per se, were predictors of all-cause and cardiovascular mortality in healthy community-dwelling older adults, independent of vitamin D status, renal function, bone mass, and comorbidities. Taken together, these findings support the notion that more attention should be paid to these factors in clinical practice to improve the management of the elderly population.
Results
During a mean of 4.06 ± 1.07 years of follow-up, there were 132 (15.7%) deaths among the 839 participants, of which 57 (43.2%) were related to CVD. The remaining 75 non-cardiovascular deaths could not be further stratified according to other causes (eg, cancer, infection) for valid analysis because of the small number of observations for each cause.
The baseline differences between the individuals who survived the follow-up and those who died are shown in Table 1. Compared with the surviving participants, the deceased subjects were older, were more frequently men, and had a lower BMI, a higher prevalence of recurrent falls, a low physical activity score, an alcohol intake ≥3 units/day, diabetes mellitus, and a prior cardiovascular event. These individuals also had a lower eGFR, lower 25OHD, higher iPTH, and lower BMD at the femoral neck and total hip.
All-cause mortality
The association between baseline characteristics and all-cause mortality are presented in Table 2. Older age, male gender, lower BMI, low physical activity score, recurrent falls, clinical fracture (hip, humerus), alcohol consumption, diabetes mellitus, cardiovascular event, lower eGFR, higher iPTH, lower 25OHD, and lower total hip BMD were most strongly associated with all-cause mortality (all p < 0.05; Tables 2 and 3).
In the multivariate Cox proportional hazards models (Table 4), age, low physical activity score, recurrent falls, diabetes mellitus, and previous cardiovascular event maintained an independent association with all-cause mortality, even after controlling for potential confounders and intermediate variables.
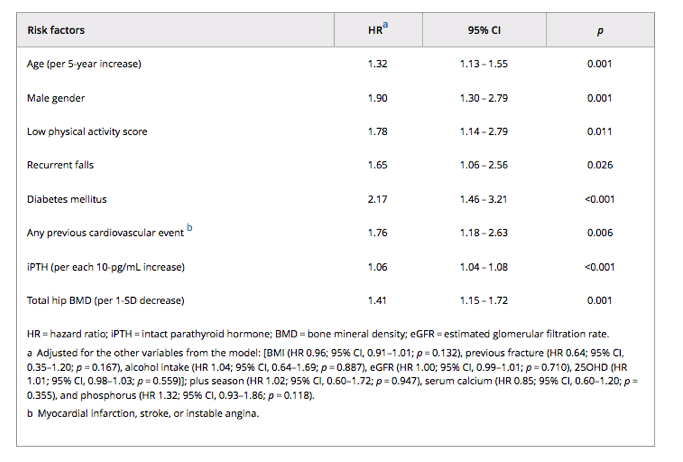
Of the bone parameters, lower baseline total hip BMD and higher iPTH levels were also predictors of all-cause mortality in the multivariate analysis (Table 4). Additionally, the adjusted Kaplan-Meier curves (Fig. 2A-D) for all-cause mortality according to hip BMD and iPTH showed a divergence of mortality risk among the strata.
Compared with the subjects in the highest quartile (Q4: >0.940 g/cm2) for total hip BMD, the participants in the lowest quartile (Q1: <0.728 g/cm2) had a higher risk of all-cause mortality (HR 2.86; 95% CI, 1.48 to 5.54; p = 0.002), as did the subjects in the second quartile (Q2: 0.728 to 0.831 g/cm2; HR 2.05; 95% CI, 1.10 to 3.81; p = 0.024). The participants in the third quartile for hip BMD (Q3: 0.832 to 0.940 g/cm2) also had a trend toward a higher risk of all-cause mortality compared with those in the highest quartile for hip BMD (HR 1.83; 95% CI, 0.99 to 3.36; p = 0.053). Analyzing total hip BMD as a continuous variable did not alter this association (HR 1.41; 95% CI, 1.15 to 1.72; p = 0.001 per 1-SD decrease), nor did the clinical category (osteoporosis [T-score ≤ -2.5] at total hip versus normal BMD [T-score ≥ -1.0]: HR 3.51; 95% CI, 1.89 to 6.52; p < 0.001; and osteopenia [T-score < -1.0 and > -2.5] versus normal BMD: HR 2.03; 95% CI, 1.28 to 3.22; p = 0.003).
The subjects in the highest quartile for iPTH (Q4: >49 pg/mL) had a significantly increased risk of all-cause mortality (HR 1.75; 95% CI, 1.04 to 2.94; p = 0.037) compared with the participants in the lowest quartile (Q1: <26 pg/mL). There was no increased mortality risk between the highest quartile and the second quartile (Q2: 26 to 35 pg/mL) (HR 1.33; 95% CI, 0.75 to 2.35; p = 0.334) or between the highest quartile and the third quartile (Q3: 36 to 49 pg/mL; HR 1.24; 95% CI, 0.71 to 2.16; p = 0.459). Similar results were found when iPTH levels were analyzed as a continuous variable (HR 1.06; 95% CI, 1.04 to 1.08; p < 0.001, per each 10-pg/mL increase) or as clinical categories (>65 pg/mL versus ≤65: HR 2.25; 95% CI, 1.40 to 3.61; p = 0.001).
Although the multivariate model showed no significant association between 25OHD levels and the risk of all-cause mortality (Table 4), an additional analysis was done in order to make sure that vitamin D levels did not influence the relationship between iPTH and mortality. Because more than 50% of subjects did have 25OHD deficiency, the risk factors for mortality were determined after excluding the individuals with 25OHD <20 ng/mL (n = 470). Of 362 subjects with 25OHD ≥20 ng/mL, 49 (13,5%) had died during the study. As a result, the multivariate Cox proportional hazards models confirmed that iPTH maintained an independent association with all-cause mortality (HR 1.12; 95% CI, 1.03 to 1.21; p = 0.007, per each 10-pg/mL increase). Furthermore, after excluding subjects with more severe vitamin D deficiency (25OHD <15 ng/mL) (n = 278), similar results were obtained (HR 1.06; 95% CI, 1.03 to 1.09; p < 0.001, per each 10-pg/mL increase). Thus, the association between iPTH and all-cause mortality was shown to be independent of vitamin D levels even in patients with only normal serum vitamin D levels. The strength of relationship between 25OHD and iPTH was very weak (Spearman's rank correlation coefficient: r = -0.187; p = 0 < 0.001).
Moreover, the same analysis was performed with the renal function. After excluding 196 subjects with low eGFR (<45 mL/min/1.73 m2), 576 individuals were analyzed, of whom 69 (12.0%) had died during follow-up. Again, iPTH was retained as predictor of all-cause mortality (HR 1.13; 95% CI, 1.05 to 1.21; p = 0.001, per each 10-pg/mL increase), independently of renal function. The strength of relationship between eGFR and iPTH was very weak (Spearman's rank correlation coefficient: r = -0.105; p = 0.005; Supporting Fig. 1).
Cardiovascular mortality
The association between baseline characteristics and cardiovascular mortality are presented in Tables 2 and 3. Older age, male gender, lower BMI, low physical activity score, clinical fracture (hip, humerus), diabetes mellitus, hypertension, any cardiovascular event, lower eGFR, higher iPTH, and low total hip BMD were most strongly associated with cardiovascular mortality (all p < 0.05).
After adjusting for potential confounders, the multivariate Cox proportional hazards models (Table 5) retained age, male gender, low physical activity score, diabetes mellitus, and previous cardiovascular event as independent risk factors for cardiovascular death.

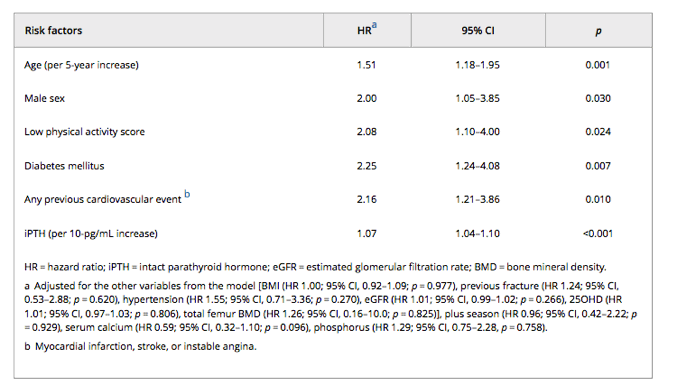
Higher iPTH levels and low BMD at the total hip were also predictors of cardiovascular mortality in multivariate Cox proportional hazards models. The adjusted Kaplan-Meier curves (Fig 3A-C) for cardiovascular mortality according to hip BMD and iPTH showed a significant difference in mortality risk among the strata.
Compared to subjects with a normal BMD at the total hip (T-score ≥ -1.0), those with osteopenia (T-score < -1.0 and > -2.5 at total hip) or osteoporosis (T-score ≤ -2.5) had an increased risk of cardiovascular mortality (HR 1.98; 95% CI, 1.00 to 3.96; p = 0.054 and HR 2.33; 95% CI, 0.87 to 6.22; p = 0.091, respectively).
The subjects in the highest quartile for iPTH (Q4: >49 pg/mL) were at a higher risk of cardiovascular death (HR 3.09; 95% CI, 1.36 to 6.99; p = 0.007) compared with the subjects in the lowest quartile (Q1: <26 pg/mL). As was the case for overall mortality, there was no significantly increased mortality risk for the individuals in the second quartile (Q2: 26 to 35 pg/mL; HR 1.51; 95% CI, 0.58 to 3.89; p = 0.399) or the third quartile (Q3: 36 to 49 pg/mL; HR 1.54; 95% CI, 0.61 to 3.89; p = 0.366) compared with those in the lowest quartile. The association between iPTH and mortality remained significant when iPTH was analyzed as a continuous variable (HR 1.07; 95% CI, 1.04 to 1.10; p < 0.001, per each 10-pg/mL increase) or when clinical categories were used (>65 pg/mL versus ≤65: HR 3.17; 95% CI, 1.63 to 6.16; p = 0.001).
Similar to overall mortality, an additional analysis was performed excluding individuals with vitamin D deficiency. Of 561 subjects with 25OHD ≥15 ng/mL, there were 57 (10.2%) deaths from cardiovascular causes during the study. As a result, the multivariate Cox proportional hazards models retained iPTH as an independent predictor of cardiovascular mortality (HR 1.11; 95% CI, 1.02 to 1.21; p = 0.017, per each 10-pg/mL increase).
Likewise, after excluding individuals with renal dysfunction (eGFR <45 mL/min/1.73 m2), 26 (4.6%) of 576 individuals with eGFR ≥45 mL/min/1.73 m2 died during follow-up. In this subsample, iPTH was also retained as risk factor for cardiovascular mortality (HR 1.15; 95% CI, 1.03 to 1.28; p = 0.01, per each 10-pg/mL increase) in the multivariate Cox proportional hazards models.
Hence, the association between iPTH and cardiovascular mortality was shown to be independent of vitamin D levels and renal dysfunction in this population.
|
|
| |
| |
|
|
|